Research Article
Assessment over Different Extraction Periods on the Amount of Zinc Extracted in Different Soils from Bangladesh
Not Available
Katsumi Ishikawa
Not Available
A.K. Chowdhury
Not Available
Naima Islam
Not Available
Zinc is a widely distributed element that occurs in small amounts in most soils and plants, but obviously plays a vital role as micronutrient for plant growth. It is a dominant heavy metal found in many organic waste materials[1]. It is an essential trace element for plant growth and human nutrition, but it can be toxic at high concentrations, depending on pH and organic C concentrations[2-5]. Zinc availability in soils is dependent on various chemical forms, particularly as it is affected by soil pH, because the different Zn fractions differ in their solubility[6-9]. Now a days the deficiency of zinc exhibits acutely in many regions through out the world as well as in the most parts of Bangladesh also.
There has been speculation regarding the observed decrease in Zn availability to plants in flooded soils. Dutta et al.[10] reported that the decrease in availability of Zn in acidic to near-neutral soils upon flooding was caused by Zn by precipitation as hydroxides, carbonates and sulfide compounds and by co-precipitation with Fe2+. The prime objective of a zinc soil test is to determine whether a given soil will give rise to zinc deficiency in a particular crop. Many early investigators used Aspergillus niger for estimating the zinc supplying power of soils[11]. Zinc deficiency was first observed in orchard soils in the USA in 1927. Now, zinc deficiency is probably the most wide spread micronutrient disorder of food crops the world over. After recognition of zinc deficiency as a field problem in rice in Asian countries, it has been reported over extensive areas in China, India, Bangladesh, Burma, Pakistan, Phillippines, Indonesia, Japan, Taiwan and Thailand[12].
Unfortunately, when new soil extraction methods were first published, there was often little or no information presented regarding the choice of extraction conditions or the effects of varying the conditions. Exceptions were in the case of Tucker and Kutrz[13], studied on HCl-extractable zinc. They reported that zinc removal during a six-week extraction period by successive extractions with 0.1N HCl and by Aspergillus niger were approximately equal and the removal of zinc by these procedures constituted about one-fifth of the total soil zinc. Some other studies have also been carried out in regarding to extract Zn with various conditions[14-18]. In this study, extractants and extraction periods are shown to have significant effects on the amount of zinc extracted from different soils of Bangladesh.
Bangladesh is situated between 20.34°-26.38° north latitude and 88.01°-92.40° east longitude. Except for the hilly regions in the north-east and south-east, some areas of high lands in the north and northwestern part, the country consists of low, flat and fertile land. In Bangladesh, 30 Agro-Ecological Zones (AEZ) have been defined. These zones can however be grouped into 20 major physiographic units. Each of the zones has specific characteristics which are related mainly to topography and soil type. Samples of unfertilized top soils (0-150 mm depth) from six different parts of Bangladesh were collected to use in this study. Brief descriptions of the soil samples are as follows:
Tea soil: Location: Manipur tea garden, Sylhet; AEZ: Eastern Surma-Kusiyara Floodplain; Soil type: Non-calcareous soil; Organic carbon: 1.12%; pH: 5.8; EDTA extractable Zn: 0.48 ppm; Olsen P: 11 ppm; Sand: 1%; Loam/Silt: 25%; Clay: 74%.
Madhupur soil: Location: Madhupur, Tangail; AEZ: Madhupur tract; Soil type: Shallow grey terrace soil; Organic carbon: 1.00%; pH: 6.3; EDTA extractable Zn: 0.51 ppm; Olsen P: 13 ppm; Sand: 0%; Loam/Silt: 12%; Clay: 87%.
Ishurdi soil: Location: Ishurdi, Pabna; AEZ: Low Ganges River Floodplain; Soil type: Calcareous dark grey soil; Organic carbon: 0.80%; pH: 7.7; EDTA extractable Zn: 0.69 ppm; Olsen P: 6 ppm; Sand: 0%; Loam/Silt: 48%; Clay: 52%.
Saline soil: Location: Khulna; AEZ: Ganges Taidal Floodplain; Soil type: Non-calcareous grey soil; Organic carbon: 0.76%; pH: 8.1; EDTA extractable Zn: 0.89 ppm; Olsen P: 9 ppm; Sand: 0%; Loam/Silt: 17%; Clay: 83%.
Farm soil: Location: Bangladesh Agricultural University (BAU), Mymensingh; AEZ: Old Brahmaputra Floodplain; Soil type: Non-calcareous dark grey soil; Organic carbon: 1.17%; pH: 6.6; EDTA extractable Zn: 0.55 ppm; Olsen P: 14 ppm; Sand: 2%; Loam/Silt: 51%; Clay: 47%.
Poorly drained soil: Location: Maskanda, Mymensingh; AEZ: Old Brahmaputra Floodplain; Soil type: Non-calcareous soil; Organic carbon: 1.25%; pH: 7.0; EDTA extractable Zn: 1.91 ppm; Olsen P: 13 ppm; Sand: 1%; Loam/Silt: 51%; Clay: 48%.
The soils were selected to represent a range of parent materials, states of soil development and drainage status. The experiment was arranged with above six soils and six different extraction periods. All treatments were replicated three times. Bulk samples of the supplied soils were collected, air-dried and ground to pass through a 2 mm stainless steel sieve, homogenized by mixing and stored at 25° in plastic packets. The soils were analyzed by the centrifuge method[19], for pH by the 1:2.5 soil/solution.
Measurement of some parameters
Soil pH: By using a combined glass and reference electrode (saturated with KCl) method the pH of the each soil sample was measured.
Organic carbon in soils: According to Walkey and Black[20] method, organic carbon in soil samples was determined. The percentage of organic carbon was calculated by using the empirical correction factor of 1.3 as suggested by Kalembasa and Jekinson[21],
| Here, | ||
| V1 | = | Volume of the K2Cr2O7 |
| V2 | = | Volume of the Fe(NH4)2(SO4)2.6H2O |
| N | = | Normality of FeSO4 |
| W | = | Wt. of soil |
Olsen phosphate in soil: For extracting Olsen phosphate, 1 g of soil (air-dried, 2 mm sieved) sample was taken and added to it 20 mL of 0.05 N NaHCO3 (adjusted to pH 8.5 with NaOH). After that the soil solution was shaked for 30 min on an end-over-end shaker. Then the sample was centrifuged for 20 min followed by filtration through (Whatman No. 42) filter papers. Finally, the concentration of P in the filtrate was measured by phospho-molybdate method[22].
Procedure for extracting zinc: The entire soil core was mixed thoroughly for the extraction of Zn by an operationally defined sequential extraction procedure. Extractions were taken place to define Zn concentration in water-soluble and exchangeable forms. For extracting Zn, we equilibrated the samples containing 10 g of air-dried equivalent soil and 25 mL of appropriate reagents in polypropylene centrifuge tubes on an end-over-end shaker at 20 for 1, 2, 4, 8, 16 and 24 h. The four reagents used were 0.05 M HCl, 1 M NH4COOCH3 (pH 7), 0.04 M EDTA, (Disodium salt of ethylenediamine tetra acetic acid, pH adjusted to 6 with NaOH (7), ASI (Agro Service International which contains 0.25 N NaHCO3-0.01M EDTA-0.0N NH4F).
Extraction periods varied from 1-24 h in six segments. Shaking of soil solutions with above reagents for 1, 2, 4, 8, 16 and 24 h were taken place in each soil sample to remove Zn in exchangeable forms. After completion of shaking, the samples were removed to centrifuge for 10 min and then filtered through filter paper (Whatman No. 42). Concentrations of Zn in all supernatant liquids obtained by the sequential extraction series were analyzed by flame atomic absorption spectrometry.
The trends in extraction of zinc with extracting periods observed the similar activities with EDTA, HCl, ASI and CH3COONH4 to those reported by Tucker and Kurtz[13], Chowdhury et al.[23]. Extraction of zinc from soils by these reagents appeared to take place quite rapidly being essentially completed in 1 to 4 h of extraction periods. The large concentration of zinc were extracted in 4 h of extraction period by HCl, EDTA, CH3COONH4 and ASI through a very small gradual increase in the following 23 h of extraction time.
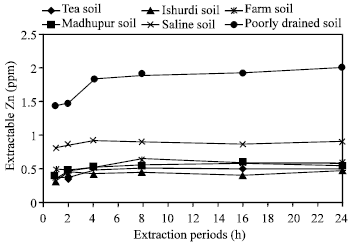 | |
| Fig. 1: | Effect of time on the extraction of Zn in different soils by CH3COONH4 |
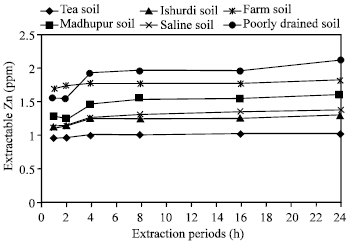 | |
| Fig. 2: | Effect of time on the extraction of Zn in different soils by EDTA |
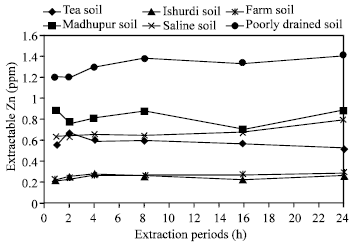 | |
| Fig. 3: | Effect of time on the extraction of Zn in different soils by ASI |
An exception was found to occur with the poorly drained soil, where this soil with HCl and to a lesser extent with EDTA and CH3COONH4, there were significant increase in the amounts of zinc extracted between 1 to 8 h (Fig. 1-4).
Effects of extraction periods with CH3COONH4: With CH3COONH4 as an extractant, all six soils showed continuous increasing release of zinc during 1, 2 and 4 h of extraction periods. It was observed for all the soils that the amount of extracted zinc had been increased gradually for 1, 2 and 4 h period and then did not change distinctly with 8, 16 and 24 h (Fig. 1 and 5-10). By 4 h period, all six soils showed greater amount of released zinc. Here, highest amount of Zn was released in Poorly drained soil than the others while Ishurdi soil showed the lowest concentration of Zn. The decreasing order of releasing zinc in different times with CH3COONH4 in different soils followed as poorly drained >Saline>Farm>Madhupur>Tea>Ishurdi soil. Zinc is more strongly absorbed by soils as the pH rise up in Ishurdi soil and thus, extractability was likely to be reduced in this soil.
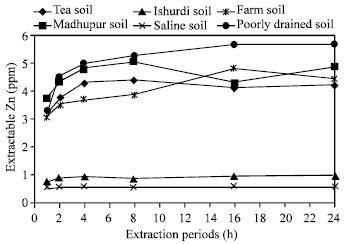 | |
| Fig. 4: | Effect of time on the extraction of Zn in different soils by HCl |
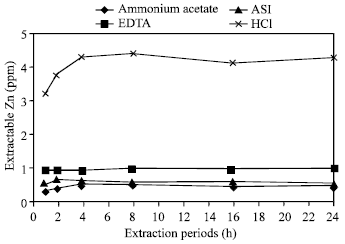 | |
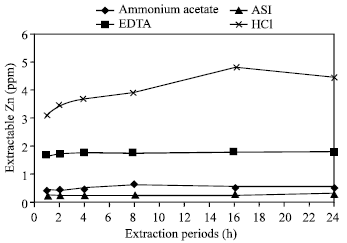
| Fig. 5: | Effect of time on the extraction of Zn in Tea soil by different extractants |
The combination of high pH relatively weak displacing power of the NH4+ ion for cations such as Zn2+ was the most likely reason for the relatively slow attainment of the equilibrium with the CH3COONH4 extractant. Barrow[24] reported the same tendency and accorded with the result of this same reagent, an extraction time of at least 4 h was required to ensure the maximum possible extraction of soil zinc.
Effects of extraction periods with EDTA: Disodium salt of EDTA usually used at its different strength as an extractant. In this study, by using 0.04 M EDTA all six experimental soils responded to remove zinc in various quantity with different extraction periods. Results showed from the Fig. 2 that in using EDTA, all soils released Zn linearly in an increasing manner up to all experimental periods.
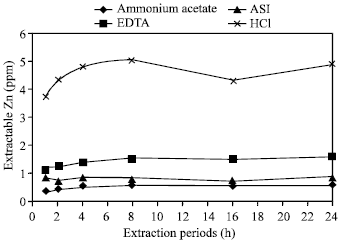 | |
| Fig. 6: | Effect of time on the extraction of Zn in Madhupur soil by different extractants |
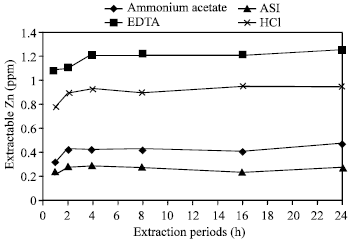 | |
| Fig. 7: | Effect of time on the extraction of Zn in Ishurdi soil by different extractants |
At the end of 1, 2 and 4 h of extraction periods the increasing rate of extracted Zn were significantly higher, but after then with the periods of 8, 16 and 24 h the increasing rate were not differentiated significantly. The extraction period of 4 h would be suggested as the critical time to allow higher amount of extracting zinc in different soils. The differences of released Zn in Tea and Farm soils with all the extraction periods were not identically different i.e. there were no effects observed in these two soils upon extraction periods. In Tea soil, zinc was extracted in a poorest amount; alternatively, in the case of Farm and Poorly drained soils, zinc was extracted in higher amounts than the others. According to Chowdhury et al.[25] the extraction of Zn in poorly drained soil has anomalous behavior due to Fe/Mn concretionary material containing occluded Zn in the soil, the graduation solution of such material by EDTA could have been responsible for the continuing release of zinc from this soil with longer extraction period.
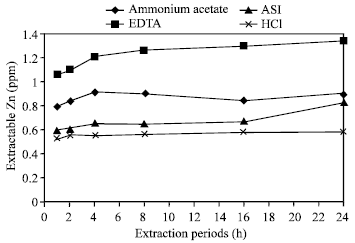 | |
| Fig. 8: | Effect of time on the extraction of Zn in Saline soil by different extractants |
Effects of extraction periods with ASI (Agro Services International): A little variation of extractable zinc was found from all the soils (six) in different times by using the extractant ASI (Fig. 3). Only poorly drained soil showed the significant increase of releasing zinc than the others in various periods. Very small amount of total Zn and negligible differences in the increment of Zn were extracted in Ishurdi and Farm soils after each experimental time. The amount of extracted zinc increased gradually up to 4 h and then not changed identically in the samples of Ishurdi, Saline, Farm and Poorly drained soils; few exceptions occurred in Tea and Madhupur soils, these two soils showed some anomalous results. Though the increments were not remarkable, the 4 h extraction period allowed for releasing higher amount of Zn in maximum soil samples. Here, 4 h period might be assessed as the critical time for extracting Zn.
Effects of extraction periods with HCl: By using HCl as an extractant, Saline soil showed no differences after elapsing all experimental periods, Ishurdi soil also exposed more or less the same results in extracting Zn and in both cases the amount of extractable Zn were less than 1 ppm that was distinctly lower than the others (Fig. 4). In the rest four soils, the amount of extracted zinc increased significantly for 1, 2, 4 and 8 h and then did not change (16 and 24 h) significantly. Here, 8 h period was the critical time. With HCl, four soils i.e. Tea, Madhupur, Farm and Poorly drained soils released zinc in greater amounts by all periods than the other two soils (Ishurdi and Saline soils). Ishurdi and Saline soils responded poorly, may be due to neutralization of acids by the soils. Tucker and Kurtz[13] examined extractability of Zn with HCl where showed the amount of extracted Zn increased steadily with time of contact up to around 40 min with only slightly more being extracted between 40 and 120 min.
 | |
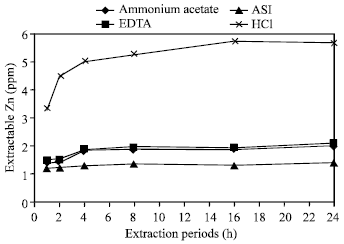
| Fig. 9: | Effect of time on the extraction of Zn in Farm (BAU) soil by different extractants |
Extraction of zinc in one hour period: For extracting zinc with one hour extraction period, ASI and CH3COONH4 exposed their lower capability while HCl showed its maximum capability (Fig. 1-10). Lesser amount of zinc was extracted in Ishurdi soil with CH3COONH4, Tea soil with EDTA, Farm and Ishurdi soil with ASI and Saline soil with HCl. On the other hand, higher amount was obtained by one hour period in Poorly drained soil with CH3COONH4 and ASI, Farm soil with EDTA and Madhupur soil with HCl. Takkar and Mann[26] worked with 0.01 N HCl and showed 1 h period reasonable for extracting zinc.
Extraction of zinc in two hour period: Small increments shown in extracting Zn with two hour period in Madhupur, Ishurdi, Saline and Poorly drained soil by CH3COONH4 (Fig. 1); Ishurdi, Saline and Farm soils by EDTA (Fig. 2); Tea, Ishurdi, Saline and Farm soils by ASI (Fig. 3) and only Ishurdi soil by HCl (Fig. 4). Alternatively, significant increments of extractable Zn released in Tea, Madhupur, Farm and Poorly drained soils by HCl (Fig. 4). Slightly decrements exposed in Madhupur soil by EDTA and ASI compare to one hour extraction period. No significant changes occurred in releasing zinc for Tea soil by EDTA, Saline soil by HCl, Farm soil by CH3COONH4 and Poorly drained soil by EDTA and ASI in between one and two hours of extracting periods. Excepts in some soils with HCl, all other reagents did not allow significant changes in extracting Zn from different soils by 2 h extraction period compare to 1 h. Maclaren et al.[27] used 0.04 M EDTA for extracting Zn with 2 h period in their experiment.
Extraction of zinc in 4, 8, 16 and 24 h periods: The extraction period within 4 to 24 h showed more or less the same tendency in releasing zinc from all soils with every extractant.
 | |
| Fig. 10: | Effect of time on the extraction of Zn in Poorly drained soil by different extractants |
Zinc was extracted in a lowest quantity from Ishurdi soil, while the highest concentration was extracted from Poorly drained soil by CH3COONH4 in 4, 8, 16 and 24 h of extraction periods (Fig. 1). With the same period conditions, zinc was released in a lower and higher amount in Tea and Poorly drained soils respectively by the reagent EDTA. The similar trend were also observed in Farm and Poorly drained soil by ASI, Saline and Poorly drained soil by HCl. The common phenomena in the amount of releasing zinc in most of the soils with most of reagents by extracting periods from 4 to 24 h, exhibited the insignificant increments of extractable zinc (Fig. 5-10). The increments were negligible in compare to those of the longer expiration of extracted periods (8, 16 and 24 h). In the cases of few soil samples the values were also little bit decreased scatteredly after expanding these periods. As the concentrations of released Zn were not differentiated significantly in between the extraction periods from 4 to 24 h, it could be suggested as 4 h period for routine work. Haldar and Mandal[28] examined with 1 N CH3COONH4 to extract the amount of Zn by 6 h extraction period.
Comparative effects of different periods: In comparing the amount of extracted zinc in all supplied soils with all the reagents, smaller amount of zinc was released in 1 h extraction period among the six periods (Fig. 1-10). The concentration of extractable zinc was found little bit higher in all soil samples with most of the reagents by 2 h of extraction period than by 1 h. For extracting Zn in different soils with all the reagents in this study, the 1 and 2 h periods might not enough to come in contact for complete reaction with soil solutions. The concentration of released zinc in 4 h extraction period was obviously and significantly greater than those of extracted by 1 and 2 h periods. From the Fig. 1-4, it is observed that Zn was released in a greater amount significantly in Poorly drained soils by 8 h with all the reagents and the highest amount of Zn was determined by HCl. The obtained concentration of zinc was not as much different significantly extracted with the periods in 4, 8, 16 and 24 h as compared with the large variable span of these periods in each of the soil sample by all the reagents. By considering the elongation of different treatment times, it might be assessed 4 h as to convenient for extracting zinc compare to other periods.
Chowdhury et al.[23] examined the effects of extraction period on the extraction of zinc from some New Zealand soils by 0.04 M EDTA, 0.05 M HCl and 1M CH3COONH4. A high proportion of the zinc was extracted with EDTA and HCl within the first 0.5 h followed by small increase over the next 23 h. An exception occurred with a soil containing iron manganese concretionary material. In this soil with EDTA, there were significant increases in the amount of zinc extracted between 1 and 8 h. The amounts of Zn extracted by CH3COONH4 increased gradually with the time of extraction up to approximately 4 h.
Results showed that the effects of length of extraction period on the extraction of exchangeable zinc from soils vary between extractants. It should also be noted that in some cases the effect of altering the extraction conditions of a single reagent varied between soils. For instance, Tea and Ishurdi soil gave anomalous results with ASI extraction in changing the length of extraction period. Though the reagents CH3COONH4, EDTA, ASI and HCl have different extractable capacity, but these all showed an increasing tendency of extracting zinc with the elapse of time up to 8 h. An eight hour extraction period is considered to be too long the routine determination of zinc in soil. Eventually, this study has shown that for some soils at least, a shorter extraction period of four hours using all the extractants may be adequate.