ABSTRACT
Sixteen different Helianthus annuus L. hybrids growing in Turkish Trakya region were included in this study. Three different methods were employed. While germination occurred in the first method following surface sterilization which was done without removing the testa, in the second one surface sterilization was done by removing the testa. Unlike these, however, in the third one, germination occurred in moist cotton media without removing the testa. Culture experiments were done in various media with varying hormone concentrations by using different explants of the obtained samples. Productive results were obtained in cultures in which cotyledon and hypocotyl explants were used. When sections of these explants were investigated, regeneration and callus formation were found to differ between native and hybrid types.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.747.751
URL: https://scialert.net/abstract/?doi=ajps.2004.747.751
INTRODUCTION
Helianthus annuus L. that grown most in Turkey, England, Russia, India and Egypt, also grown in other Europe countries too. In our country, the big part of sunflower grown areas are in the Marmara region, especially Trakya.
The studies of determine the samples cell speciality that produce with tissue culture method from hybrid seeds are plant. In the study of this subject, the productivity research of most culture type was made in different medias and different hormone concentration. The regeneration ratio that became in culture was examined in different medias and different hormone concentration, in Helianthus annuus L.[1]. Callus and root development was examined in leaf explants of Beta vulgaris L.[2]. The regeneration ratio was studied to find in cotyledon explants of Brassica napus L.[3]. The regeneration in callus culture was studied[4] and organogenesis in cotyledon explants was examined in Helianthus annuus L.[5]. Also regeneration in most explants was studied too. In other study cell culture study in somatic embryo was made[6]. Plant grown in callus and meristematic cell culture was studied in Helianthus annuus L. In one of the new study in Beta vulgaris L., suspension cell culture was formed from callus and plant regeneration from callus origin from suspension was studied[7]. In other new study somatic mutations was studied to form with radiation practice in haploid Beta vulgaris L.[8]. In the study that make cell level, callus formation and regeneration was studied and the number of chromosomes was designate in Helianthus annuus L.[9]. Which culture method gives the best result, which part of plant gives the good result in culture or which medias and hormone concentrations are suitable was seen in these studies. In these studies Helianthus annuus L. or other plant samples was used but above hybrids they wasn’t studied more was seen. In hybrid produce, as known as, new plant was produced with a different genetic features and also anatomic and cytological features from main plant group. Besides, in production of hybrid seed, some genetic features of main plant are increase or decrease. This differences are positive about farming production but it cause the plant genetic form away from its main plant group and also cause to lost ability of productivity of generations. It is unavoidable to become differences between this hybrids main plant group and other hybrids. In general this changes became external morphologic changes sometimes it can’t possible to observe changes from external morphologies. In that case anatomic or cytological examinations must be done. In these examinations, material assurances are important too. Materials can be assured from nature or farming product. But this method is depending on appropriateness of season conditions. Also there are some difficulties in these conditions too. Because the chances of environmental factors, chances of soil features, effect of other organisms and long repeatable times are some of difficulties. In general method of grow in laboratory use to remove this effects. It is possible to get the necessary factors in laboratory condition like light, heat, moisture that necessary for the plant grow but also the difficulties appears like to get soil that content suitable media or to prepare the environment of bringing up. They think that to grow the samples in laboratory condition is minimize the problem. Because in lab conditions, the factors like heat, light, moisture was obtained all the samples in equality. Besides repeatability of studies and continuously observation obtain an advantage. Other organism can give damage in outside so laboratory condition removes these problems.
This research plan to designate Helianthus annuus L. plant that is a industry plant and important in feeding for our country and Trakya region, improvement study for farming production with hybrid kinds that produce with regeneration that gets from native plants in vitro conditions and differences between callus formation.
MATERIALS AND METHODS
In this study 15 kind of hybrids and one wild types sunflower that use in farming in Edirne and Trakya region was used. Native mature sunflower’s seeds were used for control group. Helianthus annuus L. seeds were obtained from Trakya Agricultural Research Institute.
This aim, culture was tried to get from plant’s explants in primer culture tests. Medium that prepared with Murashige and Skoog (MS) Basal salt micronutrient solution (Sigma) medium[10] different concentration of plant hormone Benzylaminpürin (BAP) and Naphthalene Acetic Acid (NAA) (Sigma) was used. Used medium sterilized in autoclave (1 Atmosphere pressure, 121°C 20 min) after dissolved in sterile distilled water[11].
In first experimental method seed with it’s shells that made surface sterilized was sowed. A seed was waited 2 h (3 times chanced) in distilled water in this method. Later they were treated with 70% ethanol for 10 min and they were treated with 5% Sodium hypoclorid for 2 min. The seeds were then rinsed 3 times with sterile water for 10 min and water change every time. Seeds were aseptically dry into Laminar flow. Later the sowing was made in petri dishes (every dishes contains 10 seed) and MS salts were put in some petri dishes. The seeds were incubated in darkness that regulated at 25-27°C. They were checked everyday. They sprout between 3-7 days. And their development was observed. When the first features and roots start to formed it was taken from nutrient medium in aseptically and explants was taken from the varied parts of seedling. Explants were taken from cotyledon’s leaves, hypocotyls, root and youth leaves. Ten explants in each petri dishes and MS medium that contains 0.1 mg L-1 NAA and 0.5 mg L-1 BAP hormones concentrations was put into petri dishes. These were incubated at 20-25°C day/light temperature in 16 h photoperiod under white fluorescent light in growth chamber. The results were evaluated after 20 days in culture by scoring regenerated, callus, root and shoot formation.
In second experimental method seed without shells made surface sterilized was sowed in medium. A seed was waited 2 h (3 times chanced) in distilled water in this method. Later they were treated with 70% ethanol for 5 min and they were treated with 5% Sodium hypoclorid for 2 min[12,13]. The seeds were then rinsed 3 times with sterile water for 10 min. Seeds were aseptically dry into Laminar Flow. Later the sowing was made in petri dishes (every dishes contains 10 seed) and MS salts were put in some petri dishes. The seeds were incubated in darkness that regulated at 25-27°C. They were checked everyday. They sprout between 3-7 days. And their development was observed. When the first features and roots start to formed it was taken from nutrient medium in aseptically and explants was taken from the varied parts of seedling. Explants were taken from cotyledon’s leaves, hypocotyls, root and youth leaves. These explants were put different petri dishes and MS medium that contains 0.1 mg L-1 NAA and 0.5 mg L-1 BAP hormones concentrations was put into petri dishes. These were incubated at 20-25°C day/light temperature in 16 h photoperiod under white fluorescent light in growth chamber. The results were evaluated after 20 days in culture by scoring regenerated, callus, root and shoot formation.
In third experiment, the seeds were been sterile like the first method and then they were sprout in moist cotton. The seeds were incubated in darkness that regulated at 25-27°C. The seed was taken to grass over in dark When the first features start to form, the seedlings were aseptically divided parts in dishes. Cotyledon’s leaves, hypocotyls, root part and leaves were taken to the different dishes. Ten explants in each petri dishes and MS medium that contains 0.1 mg L-1 NAA and 0.5 mg L-1 BAP hormones concentrations was put into petri dishes. These were incubated at 20-25°C day/light temperature in 16 h photoperiod under white fluorescent light in growth chamber. The results were evaluated after 20 days in culture by scoring regenerated, callus, root and shoot formation. They were watched everyday at the cultivars experiments and photos were taken at different times (Fig. 1).
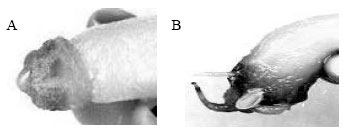 | |
| Fig. 1A: | Regeneration and callus formation from hypocotyl explants. B) Root growth from hypocotyl explants (x5) |
RESULTS AND DISCUSSION
First, various surface sterilization techniques and media were tested in order to choose the appropriate method. Of these tests, the best results from the point of view of sterilization were taken from the method in which explants, obtained from seedlings grown up in humid cotton media from seeds used after their surface sterilizations were done, were used. Unsuccessful results were obtained from those tests in which specimens with their sterilizations done without removing the test were used. During these tests, a low degree of germination was seen. It was determined that contamination rate was high and the media rapidly spoiled in cultures done with explants taken from seedlings of above-mentioned germinations. Germination rate was very low in tests performed by using seeds with their sterilizations done after removing their testa. In explants cultures of seedlings that were few in number development was rather slow and regeneration was low. Because of all these reasons we continued the culture test studies in which cotyledon leaves, hypocotyls, leaf and root explants obtained from pregerminated seedlings were used. Below findings were got with culture studies of these explants.
After all these tests, in order to determine the media and hormone concentrations that would give the most appropriate result, explants taken from seedling in sterile conditions, all which were pregerminated as this method led to high germination and low contamination rates, were placed in petri plates, which contained varying media and hormone concentrations, for cultivation. Of these different media the MS medium and culture that contained 0.1 mg L-1 NAA and 0.5 mg L-1 BAP gave the best results.
Explants of different parts of plants germinated in MS medium and in cultures involving 0.1 mg L-1 NAA and 0.5 mg L-1 BAP were obtained and cultures were monitored. For general purposes cotyledons cut from hypocotyls, explants taken from cross and longitudinal sections from cotyledon leaves, explants taken from hypocotyls, explants taken from leaves and root explants were used.
It can be concluded from the present results that although roots lived long in cultures prepared from root explants their regeneration and formation activities remained in low levels. In another test we did using the whole root that we took from seedling the root also remained same for a long time and new (secondary) root formations didn’t occur. These tests (t-test) were also performed for hybrids but no significant difference was observed[14].
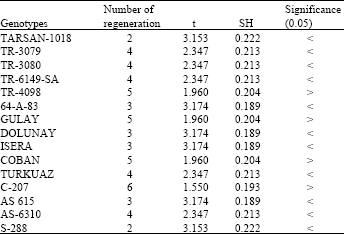
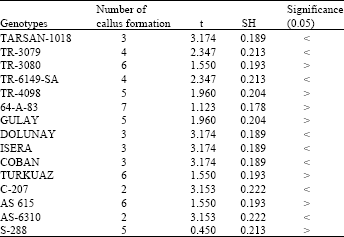
In cultures done by using hypocotyls explants regeneration was very fast and explants covered the cut surface rapidly and they increased their total volume. After a while, however, small roots and shoots appeared on these sites. But seedlings taken out of the cultures following this root and shoot appearance couldn’t live more since they didn’t adapt themselves to environmental conditions. Regeneration activities were low in same applications done for hybrids. On the other hand, this activity was high in cultures prepared by using native genotypes. In comparisons of formations, specimens of the native genotype showed more formation. The mean regeneration in the native genotype was 9 whereas statistical differences were determined in 11 of 15 hybrid genotypes. In 4, on the other hand, no significant difference was found. Mean callus formation was 9 in native genotype and statistical differences were found in 8 of compared 15 hybrid genotypes. Again, no significant difference was found in 7 (Table 1 and 2).
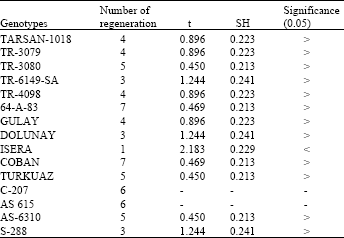
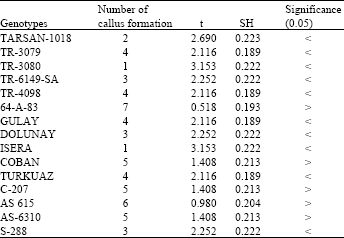
In cultures prepared by using explants of cotyledon leaves the surfaces of the cut parts of the explants recover themselves in about 2-3 days. A dark green colour occurred which we thought to be due to increased chlorophyll amount. The volumes of these explants increased in great amounts. Despite rapid regeneration, in several of the explants that showed no change for a long time shot formation was seen about 20 days later but this didn’t result in an useful productivity. In formation comparisons, native genotypes showed more formation than others. The comparisons done with this respect revealed that the mean regeneration of native genotypes was 6. While a statistical difference was found in 1 of the compared 15 hybrid genotypes no significant difference was found in 14. Mean callus formation was 8 in native genotype. Statistical differences were found in 10 of compared 15 hybrid genotypes and no significant difference was found in 5 (Table 3 and 4).
| Table 1: | Regenerated of hypocotyls explants in which contained 0.1 mg L-1 NAA and 0.5 mg L-1 BAP MS medium (Used with 10 explants for each genotypes) |
 | |
| WILD | 9 |
| Table 2: | Callus formation of hypocotyls explants in which contained 0.1 mg L-1 NAA and 0.5 mg L-1 BAP MS medium (Used with 10 explants for each genotypes) |
 | |
| WILD | 9 |
| Table 3: | Regenerated of leaf cotyledon explants in which contained 0.1 mg L-1 NAA and 0.5 mg L-1 BAP MS medium (Used with 10 explants for each genotypes) |
 | |
| WILD | 6 |
| Table 4: | Callus formation of leaf cotyledon explants in which contained 0.1 mg L-1 NAA and 0.5 mg L-1 BAP MS medium (Used with 10 explants for each genotypes) |
 | |
| WILD | 8 |
Many studies revealed that regeneration, root formation and germination abilities were much better in cultures of wild Helianthus annnus L. species compared to those of ones used for commercial production[15]. It was reported that it was possible to form callose with direct embriogenesis and to obtain a number of new plants from this callose in a very short time[16]. Same authors also found a high variation in callose formation in diploid types. Regeneraion rate was found to be low in hybrid Helianthus annuus L. types[17,18]. On the other hand, this rate is higher in wild types. In another study, it was determined that the regeneration rates of the rooting obtained from root meristems of hybrids types were higher than those of wild types[19]. In the recent study, regeneration and callose formation rates were found to be statistically high. These rates also differed among hybrid types.
ACKNOWLEDGMENTS
I would like to thank Ass. Prof. Dr. Selçuk Yurtsever for helping with statistical analaysis, Volkan Aksoy for carefully reviewing the English manusctript, in advance to Trakya Agricultural Research Institute for providing the material. The study is supported by the Scientific Research Fund of Trakya University (Project No: TUBAP-312).
REFERENCES
- Gurel, S. and E. Gurel, 2002. Establishment of cell suspension cultures and plant regeneration in sugar Beet (Beta vulgaris L.). Turk. J. Bot., 126: 197-205.
Direct Link - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link - Todorova, M., P. Ivanov, P. Shindrova, M. Christov and I. Ivanova, 1997. Doubled haploid production of sunflower (Helianthus annuus L.) through irradiated pollen-induced parthenogenesis. Euphytica, 97: 249-254.
CrossRefDirect Link








