ABSTRACT
Experiments were conducted to determine the role of epicuticular wax cover on the rate of water loss of sorghum plants. Relative Water Content (RWC), water potential (Ψ), transpiration rate, stomatal conductance and stomatal density were measured in 3 near-isogenic lines of sorghum variety ROKY62, bloom (BmBm), sparse-bloom (hh) and bloomless (bmbm) under different water conditions. Under drought conditions, bloomless sorghum exhibited more negative Ψ values than the bloom type, -1.70 Mpa as compared to -1.43 Mpa for the bloomless and bloom lines, respectively. Under normal water conditions, similar trend was observed in Ψ, where the values were -1.62 and -1.21 Mpa for the bloomless and bloom lines, respectively. Data indicated that there is a positive relationship between wax cover and the internal water content of plants. The bloomless plants had higher stomatal conductance and transpiration rates than the bloom or sparse-bloom plants. Microscopic examinations revealed higher stomatal densities on the flag leaf than on the third leaf down in all 3 isogenic lines. We conclude that wax cover on sorghum leaves reduces the transpirational water loss and prevents a rapid decrease in plant water potential.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.742.746
URL: https://scialert.net/abstract/?doi=ajps.2004.742.746
INTRODUCTION
Sorghum bicolor (L.) Moench is one of the most important sources of carbohydrate in many arid countries. It is grown in the African sub-Sahara and in the semi-arid regions of India, Pakistan and Indonesia as source of stable food. These regions are characterized by scanty rainfall and high evaporation rates, making agricultural practices challenging. Sorghum is also grown in the semi-arid portion of the Great Plains of the United States.
The vegetative body of sorghum plants is normally covered with a powdery wax. The wax cover may range from a heavy covering termed “bloom” to a light covering known as “sparse bloom.” While working with the World Collection of Sorghum in Coimbatore, Ayyangar and Ponnaiya[1] discovered that an African variety from the group sorghum elegans did not exhibit visible wax covering. They termed the condition “bloomless” and determined through back crossing that wax cover on sorghum leaves is controlled by a single dominant gene BmBm. Peterson et al.[2] reported the occurrence of two independent genes bm1bm1 and bm2bm2 controlling bloomlessness and three independent genes h1h1, h2h2 and h3h3 controlling sparse-bloom condition. The presence or absence of wax cover in sorghum is believed to play important roles in the plant’s drought resistance mechanism, in plant-insect interactions[3], plant digestibility and yield[4,5].
Wax belongs to a class of organic substances exuded from plant cells and that may appear in combination with cutin and suberin[6]. Wax exudates are most often deposited on the cuticular layer enveloping the epidermis at the interface of the plant and the atmospheric environments, playing a defense mechanism against desiccation and insect herbivores. Wilkinson and Cummins[7] analyzed the composition and roles of wax on sorghum and concluded that the presence of epicuticular wax might explain the resistance of sorghum to water loss. They determined that plant surface wax prevents water loss by reducing solar energy load on the plant through increased reflectance, an avoidance of reduced water potential and maintenance of a more complete stomatal control over transpiration[8]. The objective of this research was to investigate some physiological parameters associated with water loss on three isogenic lines of sorghum, variety ROKY62, the bloom, the sparse-bloom and the bloomless.
MATERIALS AND METHODS
Experiments were conducted at the Oklahoma State University, Stillwater, OK (USA), College of Agriculture’s greenhouse facilities to measure several physiological parameters related to plant-water relation of sorghum. The experimental design consisted of 6 treatments in form of rows. Seeds of three isogenic lines of sorghum, variety ROKY62, bloom (carrying the gene BmBm), sparse-bloom (carrying the gene h1h1) and bloomless (carrying the gene bmbm) were sown in rows, 1.0 meter long separated by 0.75 m. Germinated seedlings were thinned to allow 10 cm spacing between consecutive plants within rows. The total number of plants per row was 11, but one plant at each extremity of the rows was excluded from the test. The remaining 9 plants per row were labeled and used for all data collection. The 6 treatments were replicated 3 times and randomized. The experimental rows were watered every other day and fertilized once during the experimental period. At boot stage and continuing for 2 weeks, drought stress was imposed by withholding water. Control treatments continued to receive water every other day until the experiment was terminated.
Seven days following the onset of imposed drought and continuing for 7 consecutive days, wax exudation on leaves were recorded daily on control and drought imposed plants by visual observations. The amount of surface wax deposited was ranked on all the test plants. Because of the lack of published guideline for visual observations, we set and followed the following scale: 1 = glossy leaf surface, no wax even when leaves are rubbed against; 2 = glossy but light scattering, trace of wax only after rubbing the leaves; 3 = the amount of surface covered by wax equal to the amount of surface not covered; 4 = plant body mostly covered by wax and 5 = flaky or powdery wax over the entire plant body.
Two sets of leaf discs were taken every day from each of the test plants. One set was used for water potential determination as described by Kanematsu[6] and the second set was used for Relative Water Content (RWC) determination as described by Turner[9]. Water potential data were statistically analyzed using the analysis of variance (ANOVA) for a Randomized Block Design[10].
Stomatal density was determined according to modified techniques of Sinclair and Dun[5], Witham et al.[11]. The modification involved replacing the liquid plastic with finger nail polish. Both ad-axial and ab-axial surfaces of the leaves were investigated microscopically and the numbers of stomata per view field were recorded. All measurements were performed on the flag leaves and the third leaves down. The data were analyzed statistically using ANOVA for a Randomized Block Design.
Stomatal conductance and transpiration rates were determined over the 7 days on all experimental plants, using a steady state porometer Li-Cor 6000, according to Kanematsu[6]. Measurements were performed on the flag leaves and the third leaves down. The data were statistically analyzed using an analysis of variance (ANOVA) for a Randomized Block Design.
RESULTS
Wax deposition on the plant surface was determined using our grading scale described above. Wax covering was higher in the bloom plants than in the sparse bloom plants. We observed more intense wax deposition under imposed drought conditions than under well-watered conditions (Fig. 1a). The occurrence of wax increased linearly as the number of days without water increased, in both bloom and sparse-bloom lines (Fig. 1b). Over the entire experimental period, no surface wax was detectable with our scaling method on the bloomless plants under drought or well-watered conditions.
Plants Relative Water Content (RWC) were calculated as percent of [(fresh weight-dry weight)/(turgid weight-dry weight)]. Under imposed drought, the bloom and sparse-bloom lines had higher relative water content than the bloomless line (Fig. 2b). Under well-watered conditions however, Fig. 2a, the RWC values of the 3 isogenic lines were similar, with daily averages of 91.9, 90.3 and 91.4% for the bloom, the sparse-bloom and the bloomless, respectively. As the umber of days without water was prolonged, the RWC values of the bloomless line decreased at faster rates than those of the bloom and sparse-bloom lines. At day 7 of imposed drought, the RWC values were 76.0, 60.2 and 53.3% for the bloom (waxy), the sparse-bloom and the bloomless (non-waxy), respectively.
Plants water potential (Ψ) values were more negative under imposed drought than under well watered conditions (Table 1). Under drought conditions, the Ψ values of the flag leaves were -1.43 and -1.53 Mpa in the bloom and sparse-bloom, respectively, as compared to -1.21 and -1.40 Mpa under well-watered conditions.
| Table 1: | Averages water potential (Mpa) of ROKY 62 bloom, sparse-bloom and bloomless sorghum lines under drought and well watered conditions |
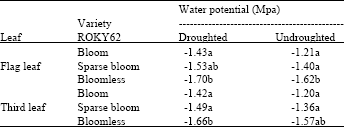 | |
| Numbers, within column, followed by the same letter(s) are not statistically different at 5% probability level | |
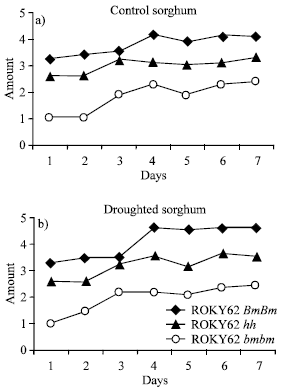 | |
| Fig. 1: | Progression of wax cover on sorghum leaves over 7 days, under well watered conditions (1a) and under imposed drought (1b.) Plants were water stressed for seven days and the amount of wax deposition recorded on the scale of 1 to 5 with 1 having no visible wax and 5 being very waxy for 7 more consecutive days |
| Table 2: | Average stomatal density (number of stomata cm-2) of the flag leaf and the third leaf down of ROKY62 sorghum isogenic lines |
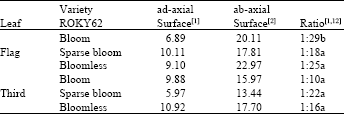 | |
| Numbers followed by the same letter are not significant at 5% probability level | |
| Table 3: | Stomatal conductance and transpiration rates of the flag leaf and third leaf down of ROKY62 |
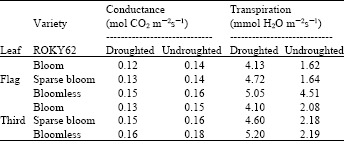 | |
| Each number is a mean of 81 individual measurements | |
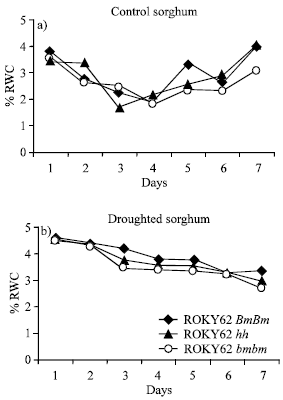 | |
| Fig. 2: | Changes in relative water content of sorghum over 7 days, under well watered conditions (2a) and under imposed drought (2b.) Plants were water stressed for seven days and the change in RWC were determined over 7 days for 7 more consecutive days |
The bloomless line had Ψ values of -1.70 Mpa under drought conditions as compared to -1.62 under well-watered conditions. Similar trends were observed with the third leaves down under drought conditions with Ψ values of -1.42 and -1.49 Mpa as compared to -1.20 and -1.36 Mpa for the bloom and the sparse-bloom lines, respectively. For the bloomless line, the values were -1.66 and -1.57 Mpa under droughted and well-watered conditions, respectively. Statistical analyses at 5% probability level indicated a significant difference exists between the water potential of the bloom and the bloomless line on both flag leaf and the third leaf down.
Stomatal densities of the flag leaf and the third leaf down of all test plants were determined microscopically and the stomatal ratios of ad-axial to ab-axial surfaces were calculated and presented in Table 2. Higher stomatal densities were observed on the lower surface of sorghum leaf than on the upper surface, with an average of ratio of 2.2 to 1. Statistical analyses were performed on the ratio between ad-axial and ab-axial stomatal density to help understand the phenomenon of leaf rolling. The flag leaf ratio of the bloom lines statistically surpassed the sparse-bloom and the bloomless lines, explaining the physiological importance of the flag leaf to the emergence of the panicle, seed setting and maturity. Most of the stomatal aperture were closed under drought conditions but remained opened under normal water conditions.
Two other parameters of photosynthetic activities, the stomatal conductance and transpiration rates, were determined using a steady state porometer, Li-Cor 6000. Measurements were performed on the flag leaf and third leaf down. The transpiration rates of the flag leaves were higher than those of the third leaves down under drought conditions (Table 3). In the bloom and sparse-bloom lines, respectively, flag leaf photosynthetic values of 4.13 and 4.72 mmol H2Om-2s-1 were observed as compared to 4.10 and 4.60 mmol H2Om-2s-1 for their third leaves down. The stomatal conductance values did not exhibit a conclusive trend, but values of the third leaves were higher than those of the flag leaves.
DISCUSSION
Plant surface wax is an anatomical feature under the control of a family of genes among which the BmBm, bmbm, HH, h1h1, h2h2 and h3h3. The phenotypic expressions of these genes appear to be enhanced by plant age as well as the plant water status as supported by this research. Wax deposition on sorghum leaf surface was recorded over the growing cycle of the plant. Present data indicated that wax cover reaches its maximum 25 to 30 days after germination, corresponding to boot stage or panicle emergence, when water balance is crucial for setting healthy and viable progeny seeds. The amount of wax cover not only increased with plant age but also with increasing water stress. The leaf rolling observed in sorghum plants was longitudinal to the main axis of the leaves, so as to expose the ab-axial surfaces high in wax cover but also high in stomatal density. It was demonstrated that wax plays an important role in preserving water status by reducing the solar energy load on the plant surface through reflectance and reduction the solar energy load on the leaf surface[12]. When water becomes a limiting factor for growth and reproduction, leaf rolling and exposing the high stomatal density surface but high in wax cover to reflect solar radiation appears to be one physiological strategy used by plants to maintain a steady water status. This is of course a disadvantage for the bloomless sorghum lacking the epicuticular wax cover.
Present data agreed with published data of Wilkinson and Cummins[7], who observed a relationship between the presence of wax on the plant surface and water loss in sorghum. Under limiting water conditions, higher RWC and less negative Ψ values were recorded in the bloom and sparse-bloom sorghum than in the bloomless. This is an indication that preserving plant internal water to avoid a more reduced water potential is among one of the effects of the epicuticular wax deposit on plant body. Under well-watered conditions, there was no difference in RWC of the isogenic lines. The Ψ values were less negative when plants have some amount of wax cover on their leaf surface, indicating that wax cover plays an important role in preventing water loss.
Transpiration rate was measured as the amount of water vapor exiting the stomatal openings/leaf area/second. The bloomless line had higher transpiration rate than the bloom line or the sparse-bloom line, indicating that the lack of epicuticular wax may favor desiccation under severe water stress. This is in agreement with published research by Jordan et al.[13] who found that plants with lower amount of wax had a higher transpiration rates but were more digestible. It is evident that plants having the dominant alleles for surface wax can be important breeding materials for the improvement of food supply in many parts of the world.
Sorghum plants possess genetic and morphological mechanisms of adaptation to low rainfall environments. These adaptation mechanisms play important roles for the third world inhabitants who rely heavily on sorghum for food. The role of epicuticular wax in preventing water loss was investigated under greenhouse conditions. Based on this research, we conclude that wax cover on sorghum leaves reduces the transpirational water loss by preventing rapid decrease water potential and relative water content during periods of water stress. We found the existence of an inverse relationship between the amount of plant epicuticular wax and its rate of water loss, which led us to conclude that wax cover on the sorghum leaf surface is a better trade than the stomatal density in controlling water loss.
ACKNOWLEDGMENTS
Special thanks to Dr. Roland Dute, Department of Biology, Auburn University, Auburn, Alabama, USA and Dr. James Rayburn, Department of Biology, Jacksonville State University, Jacksonville, Alabama, USA for reviewing this manuscript.
REFERENCES
- Turner, N.C., 1981. Techniques and experimental approaches for the measurement of plant water status. Plant Soil, 58: 339-366.
CrossRefDirect Link








