Research Article
In vitro Micropropagation of Some Important Sugarcane Varieties of Bangladesh
Not Available
M.B.H. Sikdar
Not Available
Dipak Kumar Paul
Not Available
M. Mizanur Rahman
Not Available
Md. Rezuanul Islam
Not Available
Using conventional method 10 to 15 years of work is needed to complete a selection cycle and an improved variety can be planted commercially several years later when seedcanes will have been produced in Bangladesh, sugarcane is propagated by settlings. Every year a lot of sugarcane is required as sets. Thus, farmers loss their production and sugar industries also loss sugar. Moreover, due to flood, farmers need to wait for "Jow condition" (optimum soil moisture condition for ploughing) for set sowing. Sugarcane is a major agricultural crop in the tropical and subtropical regions of the world. If the seedlings are produced in a Biotechnology Laboratory as commercial basis, farmers can sow them immediately after the run-off of the floodwater[1] On the other hand, sets use for sowing can be sent to the sugar industries for sugar production time requirement and continued infection by systematic disease are serious problems to multiply an elite genotype of sugarcane in the open field[2]. The technique of plant tissue culture is being routinely used for producing large number of clonal plants by in vitro culture of explants from wide range of species throughout the world. It has become now a viable alternative to the conventional clonal propagation methods, therefore, the present experiment was conducted to investigate the following objective: Determination of the most suitable medium compositions for rapid response on callus induction, selection and standardization of medium compositions, growth regulators and other additives for maximizing shoot differentiation, proliferation and growth, selection of suitable auxin, it’s concentration and combination for inducing roots in vitro, establishment of plantlets and to transfer in the soil for commercial crop production, to observe the variation of in vitro grown sugarcane plant for breeding program.
The young leaf-sheath of two sugarcane varieties viz., Isd-28 and Isd-29 were used as an experimental material. The explants were collected from 2 months old field grown plants, raised from the stem cutting. These explants were surface sterilized with various percentages of mercuric chloride (HgCl2) and inoculated aseptically for in vitro regeneration on MS or modified MS medium with different concentrations and combinations of growth regulators.
Different culture media used: Media for callus induction and there multiplication, different strengths of MS media supplemented with mesoinositol as organic compound as well as different concentration of auxin (2,4-D) and coconut milk were used, shoot induction media/Media for shoot differentiation from callus tissue, MS medium supplemented with auxins (NAA, IBA), cytokinins (BA) and coconut water were used for the purpose, media for root formation in vitro growing shoot, modified medium supplemented with different concentrations and combinations of auxins (NAA, IBA) and Activated Charcoal (AC) were used for the rooting of the shoots.
Sterilization and culture establishment: The leaf sheath from varieties Isd-28and Isd-29 were treated with HgCl2 for raising aseptic cultures. The treatment with different concentration of mercuric chloride (HgCl2) at different duration was applied. It was observed that cent percent contamination free cultures could be obtained when the leaf sheath explant were treated with 0.1% HgCl2 for 8 to 10 min.
Callus induction: After sterilization, the leaf sheath explants were cultured on MS medium supplemented with different growth regulators. Satisfactory callus formation was noticed only with few treatments. 2,4-D was used in different concentration viz., 0.5, 1.0, 2.5, 3.0, 4.5. Among these treatment, the modified MS medium with only inositol as the organic compound and supplemented with 3 mg L-1, 2,4-D and 10% coconut milk produce maximum amount of regenerative callus from the leaf sheath (Table 1). On this media the explant produced creamy white calli and the callus showed rapid growth and gradually spread on the surface of media after 4-5 weeks of callus induction. The calli were then subcultured of on the freshly prepared media of same composition to increase their volume for conducting experiment on shoot differentiation[3].
Shoot differentiation: The growth regulators only the BA as cytokinin and IBA and NAA as auxins were used. Various concentration of BA viz., 0.1, 0.5, 1.0, 2.5, 3.5 mg L-1 and BA+NAA or BA+IBA viz., 0.5+0.1, 0.5+0.25, 0.5+0.5, 1.5+0.1, 1.5+0.25, 1.5+0.5, 2.5+0.1, 2.5+0.25 and 2.5+0.5 mg L-1 were used indifferent replication.
| Table 1: | Effect of 2, 4-D on MS basal and modified media on callus induction |
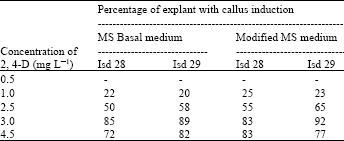 | |
| Isd 28 and 29 = Sugarcane variety (Ishardi 28 and Ishardi 29) | |
Different concentrations and combinations of growth regulators in MS media only the combination of 1.5 mg L-1 BA+0.5 mg L-1 NAA or 1.5 mg L-1 BA+0.5 mg L-1 IBA were found suitable for regeneration of shoots from the callus tissue (Table 2).
Rooting of the shoots: Usable shoots were isolated from in vitro proliferating cultures. Shoots were placed on modified MS medium supplemented with 0.1-2.0 mg L-1 of three different auxins viz., IAA, NAA and IBA. Three types of auxins, IBA and are found to be more effective. Microshoot of Isd-28 and Isd-29 showed better rooting performance. The quinces of root formation for the two varieties were 90 and 87% for NAA; 93 and 95% for IBA, respectively. On the medium with 0.5 mg L-1 all the auxins produced the highest number, of roots per shoot (3.12±0.8 and 3.4±0.3 for the auxins NAA+IBA and 3.5±0.1, 3.5±0.03 for the auxins IAA+IBA in Var. Isd-28 and Isd-29, respectively) as shown in Table 3.
Effect of activated charcoal on root: Use of activate charcoal with 0.5 mg L-1 IBA+0.5 mg L-1 NAA supplemented modified MS rooting medium showed better performance than those rooted on activated charcoal omitted medium.
Establishment of the plantlets: After rooting of the in vitro regenerated shoots they were transferred to soil. Before transplantation, the individual plantlets were taken out from the culture media and the roots of the plantlet were washed gel free by continuous flowing of tap water. Then the plantlets were transferred on to the sun-dried soil containing sand in pots. The potted plants were then watered adequately, covered with perforated polythene bag and kept in the growth chamber for 10-12 days. Among the transplanted plants 60-75% were survived and acclimatized successfully on the soil. The rest of the transplants could not survive under the ex in vitro due to desiccation and microbial overgrowth. The survival of the plantlets under the ex in vitro condition on the soil also influenced by state of root growth and general health of the plantlets.
| Table 2: | Combined effect of auxin and cytokinin on shoot differentiation and proliferation |
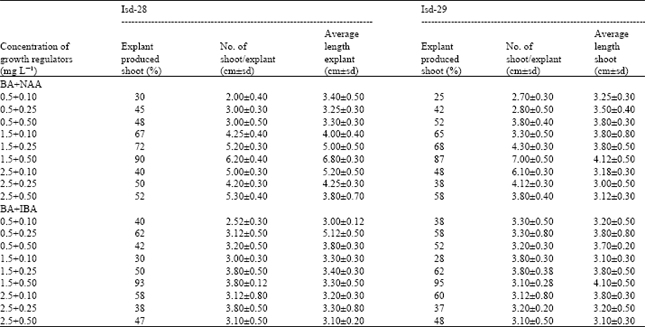 | |
| BA=Cytokinin ; NAA, IBA= Auxin; Isd 28 and 29 = Sugarcane variety (Ishardi 28 and Ishardi 29) | |
| Table 3: | Combined effect of auxins on root induction of sugarcane varieties |
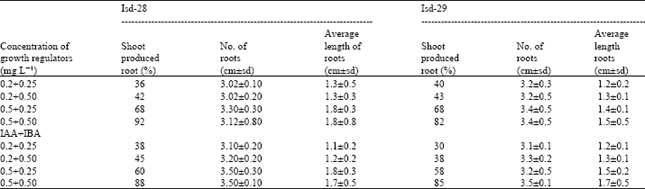 | |
| NAA, IBA, IAA= Auxin; Isd 28 and 29 = Sugarcane variety (Ishardi 28 and Ishardi 29) | |
The survival rate of healthy plantlets having 1-2 cm root length was more than those of plantlets having elongated roots.
Plant tissue culture is an essential plant biotechnology offers novel approach to plant propagation, in vitro micromanipulation and biodiversity and germplasm conservation. It has become now an important alternative to conventional propagation and breeding programs for wide range of plant science[4,5]. After surface sterilization with HgCl2 leaf sheath explants were cultured on modified MS medium containing several concentrations of auxins (2,4-D, NAA IBA) viz., 0.5, 2.5, 3.0, 4.5 mg L-1 to investigate that the medium callus induction frequency of the leaf sheath explant obtained callus from leaf explant on medium containing 31.7 μm of 2,4-D after 4 weeks[6,7]. Established callus on explant tissue taken from a range of 18 genetically diverse sugarcane cultivars on MS medium containing 13.4 μm of 2,4-D found suitable of 2,4-D (3-5 mg L-1) for callus in Bangladeshi sugarcane varieties[8]. All these studies indicate that sugarcane explant requires higher concentration of 2,4-D for callus induction. In this regard results of the present investigation are consistent with high level of cytokine and low level of auxin was essential for differentiation of adventitious shoot in sugarcane leaf sheath callus[9]. Shoots of in vitro proliferating culture were isolated and individually cultured on MS medium supplemented with auxins viz., IBA and NAA for root induction. During the investigation the number of roots per shoot were highly influenced by concentration and type of auxin used. Among the different auxin supplemented media lowest rooting was obtained on half strength MS medium and highest was obtained on modified half strength MS media. NAA was found to be comparatively more effective than IBA at different concentrations tested, The various concentrations tested 0.5 mg L-1 of all the auxin produce highest frequency of root formation. It is also showed that 0.5 mg L-1 of auxins with activated charcoal the best result. Sugarcane roots can be developed from plantlets when they are transplanted to modify MS media 0.5 mg L-1 IBA or 0.5 mg L-1 NAA+0.5 mg L-1 IBA[10]. The best root induction was observed MS medium supplemented with 7.4 μm IAA and 4% of sucrose[11] MS or modified MS with 1 mg L-1 IBA was suitable for rooting in the experiment.