ABSTRACT
Microbial resistance, a world health-hazard, is dramatically increasing. Evaluation of natural products to access new, safe and effective principles to rotate or substitute with the invalidated ones is one of the scientific strategies to combat drug-resistant pathogens. With this perspective, methanolic extracts of fifty plant species of 33 families which had documented uses in Iranian Traditional Medicine, were screened for antibacterial activity against five strains of each of gram negative (G-) and gram positive (G+) bacteria. Thirty samples from 28 species in 21 families had antibacterial activity at least against one of the bacterial strains. Bioactivities were evaluated by measuring Diameter of inhibition zones in Agar well diffusion assays. Among the active plants, 32.6% were active against G-, 62% against G+ and 47.3% against both G- and G+ bacteria. Dianthus coryophyllus was active against all tested G- and G+ bacteria except Micrococcus luteus. Most susceptible G- bacteria were Klebsiella pneumoniae and Bordetella bronchiseptica and least susceptible G- bacterium was Escherichia coli. In G+ bacteria, most and least susceptible were Staphylococcus aureus and M. luteus, respectively. The extracts having more traditional usages and fairly broad spectra of activity, were used to determine Minimum Inhibitory Concentrations (MIC). The least MIC, as 0.062 mg ml-1 belonged to Myrtus communis seeds against S. aureus, Bacillus cereus and B. bronchiseptica and to Terminalia chebula ripe seeds against S. aureus. The bioactive extracts were well stable at room temperature up to 18 months. Concepts including Percent Activity, Bacterial Susceptibility Index, Average Percent of Bacterial Susceptibility and Spectral Intensity Index are proposed as new approaches for interpretation of the results.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.55.60
URL: https://scialert.net/abstract/?doi=ajps.2004.55.60
INTRODUCTION
According to WHO reports[1] increase of antibacterial resistance is a growing problem in many countries. This problem undoubtedly will persist as long as antimicrobial agents are employed. Since the advent of semi synthetic penicillins in the 1960s it was not before staphylococcal resistance emerged and for the 15 years methicillin resistant Staphylococcus aureus (MRSA) has been troubling hospital services all over the world[2,3]. Isolation of microbial agents less susceptible to regular antibiotics and recovery of increasing resistant isolates during antibacterial therapy is rising throughout the world[4-10]. One of the measures to minimize the increasing rate of resistance in the long run, is to have continuous in depth investigation for new, safe and effective antimicrobials as alternative agents to substitute with no effective ones. Natural resources specially plants and microorganisms are potent candidates for this aim. Many workers have reported antibacterial activities of plant extracts on this subject. McCutcheon et al.[11] tested 100 methanolic extracts of the plants, used by British Colombian Native people, against 11 bacterial isolates. They found 85% of the plants were active at least against one of the bacteria. They also reported that 75% of active extracts were active against methicillin-resistant S. aureus. Basile et al.[12] evaluated the action of acetone extract of Lunularia cruciata (Bryophyta) against 13 bacterial strains and noticed it had high antibacterial potency. Digrak et al.[13] examined antibacterial activity of several plants and found Mimosa bark extract had the greatest antibacterial activity against several species of Bacillus spp. Golan-Goldhirsh et al.[14] tested extracts of 66 desert plants for antimalarial activity. They found 12 extracts demonstrated strong growth inhibition (>96%) of the malaria parasite, Plasmodium falciparum. Mansouri[15] tested ethanolic extracts of 11 plants against 489 samples of S. aureus and noticed Myrtus communis leaves had the greatest activity, inhibiting the growth of 99% of the isolates. Adedayo et al.[17] reported antibacterial potency of Senna alata flower extract against S. aureus, S. faecalis, Micrococcus luteus, Bacillus subtilis and Pseudomonas putida. Mansouri et al.[18] evaluated antibacterial activity of crude extract of M. communis against 10 laboratory strains of bacteria. They noticed that the crude extract inhibited the growth of all tested bacteria except Campylobacter jejuni. Yoshikazu et al.[19] evaluated inhibitory effect of 21 plant samples on production of Verotoxin by enterohemorrhagic E. coli 0157:H7, a food-borne human pathogen and found 4 plants well active.
In a two-year study, a procedure was set to screen and evaluate antibacterial activity of some plants used by Iranian Traditional People (ITP) as antiseptic, anti-inflammatory and in treatment of infectious diseases and dermatophytes. Information gathered from ethnopharmacologists, herbal-drug sellers and rural native-healers. Based upon the collected information, plant organs used in this study are as used by ITP.
MATERIALS AND METHODS
Plant material and extraction procedure: Fifty plant species belonging to 33 families used by ITP, collected from South regions of Iran from June 1997 through September 1999 and identified by Mrs. P. Rashid Farrokhi in the Herbarium of Plant Systematic Laboratory of the College of Agricultural Sciences, Bahonar University of Kerman, Iran, where voucher specimens of plants were deposited. Plant organs of few species as Alpinia officinarum, Amomum subulatum, Cinnamomum zeylanicum, Helictres isora, Semicarpus anacardium, Smilax china, Terminalia chebula and Zingiber officinale used by ITP but not grown in Iran, were prepared through the mentioned Herbarium. The fine powder of air dried specimens were extracted three times with methanol at 70°C for 4 h and the extracts were then concentrated under reduced pressure to yield a dense residue. Each sample transferred to glass vials and lyophilized overnight before use.
Test organisms and agar well diffusion bioassay: Ten registered bacterial strains including E. coli (PTCC No. 1330), Pseudomonas aeruginosa (PTCC No. 1074), P. fluorescens (PTCC No. 1181), Klebsiella pneumoniae (PTCC No. 1053), Bordetella bronchiseptica (PTCC No. 1025), Staphylococcus aureus (PTCC No. 1112), S. epidermidis (PTCC No. 1114), M. luteus (PTCC No. 1170), Bacillus cereus (PTCC No. 1015) and B. pumilis (PTCC No. 1319) obtained from Persian Type Culture Collection, Tehran, Iran (PTCC). The bacteria rejuvenated on Mueller-Hinton-Agar medium (MHM, E. Merk, Germany) and subcultured as needed. For bioassays, suspension of approximately 1.5x108 cells ml-1 in sterile normal saline prepared as described by Forbes et al.[20] and about 1.5 ml of it was uniformly seeded on MHM in 12x1.2 cm glass Petri dishes, left aside for 15 min and excess of suspension was then drained and discarded properly. Wells of 6 mm in diameter and about 2 cm apart punctured in the culture media using sterile cork borers. As a precaution not to miss trace amounts of antimicrobials, a relatively high concentration of 20 mg ml-1 of each extract was prepared in dimethyl sulfoxide (DMSO): methanol (1:1, v/v) (DM solvent) and administered to fullness in each well. Culture plates, were incubated at 37°C for 48 h and then bioactivity was determined by measuring Diameter of Inhibition Zones (DIZ) in mm. Solvent controls were included, although no antibacterial activity has been noted in the solvent employed. All samples tested in triplicate and average results recorded.
Determination of minimum inhibitory concentration: Minimum inhibitory concentrations (MIC) of the most active methanol extracts (DIZ = 15 mm) were determined using two-fold serial dilutions of 20 to 0.31 mg ml-1 in DM solvent against the three most sensitive bacterial strains of S. aureus, B. cereus and B. bronchiseptica and tested as mentioned.
Determination of shelf life or stability of the activity: To measure the stability of the bioactive extracts in both solubilized and dry states, 20 mg ml-1 of each was prepared in DM solvent and 20 mg dry samples in small vials were kept at room temperature and were tested for antibacterial activity against S. aureus (the most sensitive to all active extracts) at 14 days intervals up to 18 months.
New approach in evaluation of antimicrobial screening results: The following terminologies proposed to present comparative numerical scales for evaluation of the efficiency of active extracts and compare susceptibility of bacterial strains in screening studies.
Percent activity: Percent Activity of each extract is calculated as 100 X No. of susceptible bacterial strains to a specific extract ÷ total No. of tested bacterial strains. This term is expressed as % G- for percent activity against G- bacteria, % G+ for percent activity against G+ bacteria and %T for percent total activity against both G- and G+ bacteria.
Bacterial susceptibility index: Bacterial Susceptibility Index (BSI) is calculated as 100 X No. of extracts effective against each bacterial strain ÷ No. of total samples. BSI is expressed as % value and is used to compare the relative susceptibilities among the bacterial strains. BSI ranges from zero (resistant to all samples) to 100 (susceptible to all samples).
Average percent of bacterial susceptibility: Average Percent of Bacterial Susceptibility (APBS) is calculated as sum of % activities (%G-, %G+ or %T) ÷ No. of total samples. APBS represents overall susceptibility of each group of bacterial strain.
Spectral intensity index: Spectral Intensity Index (SII) is calculated as Mean of DIZs (mm) of all sensitive bacterial strains to a specific sample X %T ÷ 100.
RESULTS
From 50 plant species in 33 families used by ITP, 28 species in 21 families showed antibacterial activities at 20 mg ml-1 as shown in Table 1. All other 22 plant species did not show any inhibitory effects, hence are not listed in the table. The largest DIZ belonged to Myrtus communis leaf extract against B. bronchiseptica, S. epidermidis and B. cereus, as 26 mm. The broadest spectra of activity belonged to Dianthus coryophyllus, all bacterial strains were sensitive to it except M. luteus. M. communis (leaves) and Zingiber officinale (rhizome) were effective against all of the G+ bacterial strains. Considering BSI, K. pneumoniae and B. bronchiseptica were most susceptible G- bacteria, having BSI equal to 40 and most susceptible G+ bacterium was S. aureus having BSI equal to 96.6. Average BSI for G- bacteria was 26.6 and for G+ bacteria was 61.3, showing that the active plants are more effective against G+ than G- bacteria. The average BSI for both G- and G+ bacteria was calculated as 43.9. The APBS values show that 32.6% of the samples were active against some G-, 62% were active against some G+ and 47.3% were active against some of both G- and G+ bacteria. The highest SII values belong to T. chebula (13.6), M. communis leaves (13.4) and D. coryophyllus (13.4), respectively. Foeniculum vulgare, Cuminum cyminum and Borago officinalis were poorly effective only against G- bacteria and none of G+ bacteria were sensitive to them, their SII values were low as 2.4, 2.4 and 1, respectively. The MIC values determined against S. aureus, B. cereus and B. bronchiseptica via two fold serial dilutions are presented in Table 2. The lowest MIC values were measured in T. chebula (ripe seeds) as 0.62 mg ml-1 against S. aureus with 10 mm DIZ and in M. communis seeds as 0.62 mg ml-1 against all three tested bacteria with 10, 9 and 9 mm DIZs, respectively. The highest MIC values were measured as 10 mg ml-1 in Thymus vulgaris against B. cereus and Trachysermum copticum against B. bronchiseptica, with 9 mm DIZs.
All of the active extracts were well stable at room temperature in both DM solvent and dry state up to 18 months and did not show any reduction of activity against S. aureus, as compared to the activities of the starting day.
DISCUSSION
The potencies of traditional antibiotics are decreasing steadily since drug-resistant bacteria are globally increasing,. Some workers express the situation very dramatic, Smith et al.[3] expresses that "The emergence of S. aureus resistance threatens to return us to the era before the development of antibiotics". The hazard of increasing prevalence of drug-resistant bacteria is a major health problem throughout the world. Beside all preventive measures, Health organizations should put emphasis on research for new and safe antimicrobials, preferably from natural origins like plants and microorganisms, to substitute with impotent traditional antimicrobials or to be used in antibiotic rotation programs [21-23].
Although the nature and number of active antibacterial principles involved in each extract of the present research are not clear, but the broad spectra of activity of several plant extracts specially on S. aureus, is promising. It is wise to consider the most active plant extracts of Table 2 for evaluation against methicillin-resistant S. aureus[10,24] and vancomycin-resistant enterococci[4,25] two of the most problematic bacteria. The results of such studies form the basis for further investigation to isolate pure compounds, elucidate the structures and evaluate them against wide range of drug-resistant bacterial strains with the goal to find new therapeutic principles.
In the proposed method for evaluation of the results, the followings have to be considered properly. A) Percent Activity of samples is used when several bacterial strains are employed. B) Bacterial Susceptibility Index (BSI), defined as percent value, is applicable in comparisons of the relative susceptibilities among different tested bacterial strains. It varies from zero (the bacteria resistant to all samples) to 100 (bacteria are susceptible to all samples). C) Average Percent of Bacterial Susceptibility (APBS) representing overall susceptibility of each group of bacteria is a means of overall comparison among different groups of tested bacteria. D) Spectral Intensity Index (SII) is used as a means of expressing combination of both spectral range and magnitude of effectiveness (DIZs) of a specific sample against all sensitive bacteria. Since the intensity of effectiveness of samples at a constant concentration (e.g. 20 mg ml-1 at this study) can not be compared clearly among similar %Ts, SII can be used as a means of comparison or ranking between the effective extracts especially among equal %Ts.
| Table 1: | In vitro evaluation of antibacterial activity of plants used in Iranian Traditional Medicine against 5 G- and 5 G+ strains of bacteria, indicated by Diameter of Inhibition Zones (DIZ, mm). Plant species, families, organs used, bacterial species, are listed. Percent activity, Spectral Intensity Index, Bacterial Susceptibility Index and Average Percent of Bacterial Susceptibility represent comparative numerical scales for evaluation of antibacterial efficiency of extracts and susceptibility of bacterial isolates (refer to text for details). In this table, the plants are sorted according to Spectral Intensity Index values. Blanks represent no inhibition and are equal to zero |
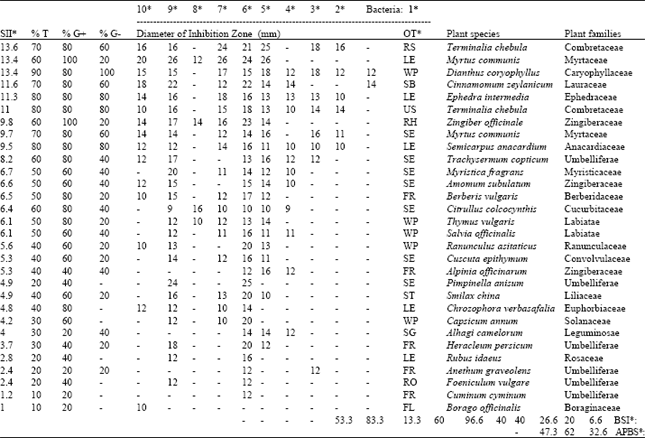 | |
| OT*: Organs tested as FL: Flower, FR: Fruit, LE: Leaves, RH: Rhizome, RO: Roots, RS: Ripe Seeds, SB: Stem Bark, SE: Seeds, SG: Stem Gum, ST: Stem, US: Unripe Seeds and WP: Whole Plant. 1*: E. coli, 2*: P. aeruginosa, 3*: P. fluorescens, 4*: K. pneumoniae, 5*: B. bronchiseptica, 6*: S. aureus, 7*: S. epidermidis, 8*: M. luteus, 9*: B. cereus, 10*: B. pumilis. % G-: Percent Activity on G- bacteria, % G+: Percent Activity on G+ bacteria, %T: Percent Total Activity on both G- and G+ bacteria, SII*: Spectral Intensity Index, BSI*: Bacterial susceptibility index (%), APBS*: Average % of bacterial susceptibility | |
| Table 2: | Minimum inhibitory concentration (MIC) of the most active plant extracts against three susceptible bacterial strains of S. aureus, B. cereus and B. bronchiseptica. Concentrations at which Diameter of inhibition zones (DIZ, mm) are indicated in bold represent the MIC of the extract on the respective bacterium. Blanks represent no inhibition and are equal to zero |
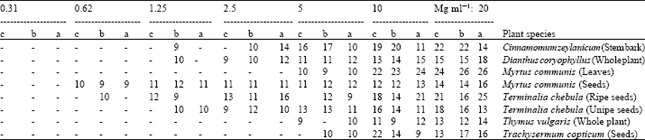 | |
| a, DIZ on S. aureus, b, DIZ on B. cereus, c, DIZ on B. bronchiseptica | |
SII does not have an upper limit, but its lower limit is 0. It is applicable only in experiments performed under similar conditions but not applicable to compare the results of different workers, except under identical conditions. It is applicable when more than one bacterial strain is used in screening programs. In extensive surveys, where many bacterial strains are employed, SII can be expressed for each tested bacterial group like MRSA or VRE strains, etc. If SII is being used to compare results of different studies, experimental conditions should be set similar, e.g. bacterial strains, bioassay method, reagents and culture media, incubation period and temperature, plant variety and the collection region, sample preparation and extraction methods.
Since S. aureus (PTCC No. 1112) had the highest BSI among other bacterial strains (96.6) it can be used as a very susceptible strain in preliminary screening programs. Finally, since Iran possesses wide range of medicinal plants[16] in-depth biological and phytochemical studies of them would provide valuable information to the world media of knowledge.
ACKNOWLEDGMENTS
Financial support of Research Affairs Office of Bahonar Univ. of Kerman is appreciated. Thanks to Mrs. P. Rashid Farrokhi from Herbarium of Plant Systematic Laboratory of the College of Agricultural Sciences, Bahonar University of Kerman. The spiritual result of this research is dedicated to Mr. A. Afzalipour the founder of Universities at Kerman, God may bless him.
REFERENCES
- Archibald, L., L. Phillips and D. Monnet, 1997. Antimicrobial resistance in isolates from inpatients and outpatients in the united states: Increasing importance of the intensive care unit. Clin. Infect. Dis., 24: 211-215.
PubMed - Smith, T.L., M.L. Pearson, K.R. Wilcox, C. Cruz and M.V. Lancaster et al., 1999. Emergence of vancomycin resistance in Staphylococcus aureus. N. Engl. J. Med., 340: 493-501.
Direct Link - Gold, H.S. and R.C. Moellering Jr., 1996. Antimicrobial-drug resistance. N. Engl. J. Med., 335: 1445-1453.
CrossRefDirect Link - Chen, D.K., A. McGeer, J.C. de Azavedo and D.E. Low, 1999. Decreased susceptibility of Streptococcus pneumoniae to fluoroquinolones in Canada. N. Engl. J. Med., 341: 233-239.
CrossRefPubMedDirect Link - Lelievre, H., G. Lina, M.E. Jones, C. Olive, F. Forey and M. Roussel-Delvallez, 1999. Emergence and spread in French hospitals of methicillin-resistant Staphylococcus aureus with increasing susceptibility to gentamicin and other antibiotics. J. Clin. Microbiol., 37: 3452-3457.
Direct Link - Shopsin, B., B. Mathema, J. Martinez, E. Ha and M.L. Campo et al., 2000. Prevalence of methicillin-resistant and methicillin-susceptible Staphylococcus aureus in the community. J. Infect. Dis., 182: 359-362.
PubMedDirect Link - McCutcheon, A.R., S.M. Ellis, R.E. Hancock and G.H. Towers, 1992. Antibiotic screening of medicinal plants of the British Colombian native people. J. Ethnopharmacol., 37: 212-223.
Direct Link - Golan-Goldhirsh, A., H. Lugasi-Evgi, P. Schlesinger, I. Kedar, J. Gopas and Y. Pollack, 1999. Screening for cytotoxic and antimicrobial activities in desert plants of the Negev and Bedouin market plant products. Pharma. Biol., 37: 188-195.
Direct Link - Shahidi, G.H., M.R. Moein, A.R. Foroumadi and F. Rokhbakhsh-Zamin, 2002. Cytotoxic activity of medicinal plants used in Iranian traditional medicine on two strains of Saccharomyces cerevisiae. DARU, 10: 162-164.
Direct Link - Adedayo, O., W.A. Anderson, M. Young, V. Snieckus, P.A. Patil and D.O. Kolawole, 2001. Phytochemistry and antibacterial activity of Senna alata flower. J. Pharm. Biol., 39: 408-412.
CrossRefDirect Link - Mansouri, S., A. Foroumadi, T. Ghanei and A.G. Najar, 2001. Antibacterial activity of the crude extracts and fractionated constituents of Myrtus communis. Pharm. Biol., 39: 399-401.
Direct Link - Yoshikazu, S., H. Murata, T. Nakanishi and Y. Inatomi, 2001. Inhibitory effect of plant extracts on production of verotoxin by enterohemorrhagic Escherichia coli O157: H7. J. Health Sci., 47: 473-477.
Direct Link - Gerding, D.N., T.A. Larson and R.A. Hughes, 1991. Aminoglycoside resistance and aminoglycoside usage: Ten years of experience in one hospital. Antimicrob. Agents Chemother., 35: 1284-1290.
PubMedDirect Link - Quale, J., D. Landman and E. Atwood, 1996. Experience with a hospital-wide outbreak of vancomycin-resistant enterococci. Am. J. Infect. Control, 24: 372-379.
PubMedDirect Link - Abi-Hanna, P., A.L. Frank and J.P. Quiinn, 2000. Clonal features of community-acquired methicillin-resistant Saphylococcus aureus in children. Clin. Infect. Dis., 30: 630-631.
Direct Link








