Research Article
Effects of Explant Type and Growth Regulators on Organogenesis in a Medicinal Tree, Phellodendron amurense Rupr.
Not Available
S. Yokota
Not Available
S. Yahara
Not Available
N. Yoshizawa
Not Available
Phellodendron amurense Rupr. (syn. name: Kihada, Huang Bai, Amur Corktree), which belongs to the family Rutaceae, is a dioecious tree native to Eastern Asia (northern China, Manchuria, Korea, Ussuri, Amur, and Japan), and has remarkable medicinal properties. The bark of this tree contains medicinally important compounds, such as anti-inflammatory, antipyretic, cholagogue, and antibacterial medicine. It is also effective for purging heat, detoxifying, clearing damp heat, and lowering blood sugar. It can be said that P. amurense is an effective herb used topically for sores, and damp heat conditions of the skin[1].
The wood of P. amurense has a beautiful colour, and high quality grains, and is very easy to process. For this reason, the wood of this species has been used for making furniture or industrial arts. This tree also provides industrial cork, and yellow dying[2]. In Japan, however, at least 80% current consumption of Phellodendri cortex is import-dependent. To increase the cultivation of the cork tree therefore, more productive use of the wood must be sought for, seeing that the sale of Phellodendri cortex (endodermis) is economically disadvantageous[3].
Although this tree is conventionally propagated by seeds, the germination rate is very low[4]. In addition, propagation by stem cutting is very difficult, because of its poor rooting ability[5]. Kamiya et al.[5] also reported that about 35% of plants were regenerated successfully by grafting. On the other hand, the in vitro micro propagation technique holds a potential, and could offer solutions to these problems. Micro propagation techniques would produce large quantities of uniform plants from selected plant species with proven qualities.
Although there are a few reports on micro propagation through axillary buds of P. amurense[6], so far there is no report of micro propagation on this valuable medicinal plant through hypocotyl, cotyledon, leaf, and internodal explants. The present paper reports a new micro propagation protocol through various explants of P. amurense, and also reports the comparative response of selected explants, and effects of growth regulators on the explants.
Fruits of Phellodendron amurense Rupr. were collected from a 50-year-old tree growing at the Medicinal Plant Garden of Kumamoto University, Japan. After removing flesh from fruits, seeds were recovered. They were washed with detergent-containing water for 15 min, and then rinsed thoroughly with running tap water for 20 min. After these seeds were surface-sterilized with 70% ethanol for 3 min, they were moved to a laminar-air-flow cabinet, and transferred to a sterilized conical flask. Subsequent surface disinfection was done with 3% sodium hypochlorite solution for 20 min. To remove any trace of the sterilants, the seeds were then washed with at least three changes of sterile distilled water. After that, sterilized seeds were germinated on 10 mL of MS medium[7] surface in culture tube (120x25 mm, Asahi Techno Glass, Japan) supplemented with 0.44, 0.89, 2.22, and 4.44 μM BAP, and without growth regulator. Among these treatments, 2.22 μM BAP containing medium showed the maximum number of seed germination, and the frequency was 80%, whereas other treatments showed less performance than 2.22 μM BAP. Five to six weeks old in vitro grown seedlings were used as the source of explants for different experiments. Subsequently hypocotyl , and cotyledon explants were prepared from 6-week-old aseptically grown seedlings, and leaf, and internode explants were prepared from in vitro grown shoots, which were developed on node cultures of aseptically grown seedlings. Then the explants were cultured on MS media supplemented with different concentrations, and combinations of a cytokinin, and auxins for inducing adventitious shoots. These explants were cultured on MS media containing different concentrations (0.9-4.4 μM) of BAP in combination with 0.5-2.5 μM NAA or IBA for the induction of callus with adventitious shoot regeneration. For the regeneration of complete plantlets, the microcuttings (2-4 cm long) obtained from micro propagated shoots were rooted on MS media supplemented with different concentrations (0.5-4.0 μM) of IBA, NAA, or IAA. pH of both proliferation, and rooting media were adjusted to 5.7±0.1. The media were fortified with 3% sucrose (w/v), and gelled with 0.2% gellan gum. All cultures were initiated in 120x25 mm glass tube containing 10-15 mL of medium. The cultures (callus with 4-6 small shoots) were regularly subcultured on fresh medium at 4 weeks intervals in 200 mL conical flasks. The cultures were grown at 25±1°C under illumination for 16-h photoperiod with a light intensity of 50-60 μ mol m-2 s-1.
After one month of rooting culture, rooted plantlets were removed from the culture media, and the roots were washed in tap water to remove all traces of gellan gum. Plantlets were then transferred to plastic pots (9 cm diameter) containing Kanuma soil (Kanuma Bonsai Potting Medium Co., Japan), covered with a transparent plastic cup to ensure high humidity during the acclimatization period of 20 days. They were maintained under culture room conditions. The potted plants were irrigated with MS basal salt solutions (¼ strength) devoid of sucrose, and myo-inositol every 4 days for 3 weeks. Plastic cups were removed after three weeks in order to acclimatize plants to laboratory room conditions. Acclimatized plants were then transferred to larger pots (24 cm diameter), and maintained in greenhouse.
The experiment had 4 replicates, each replicate consisted of 20 culture flasks (200 mL). Results were recorded at a regular interval of 4 weeks of culture, and analysed by analysis of variance using randomized block design (RBD) method. It was recorded that the percentage of calli producing shoots, total number of shoots arising from each callus, usable shoots (shoot length above 2 cm), average length of shoots, percentage of rooted cuttings, number of roots per rooted cuttings, and average length of roots. The effects of different treatments were compared to detect the significance differences among the treatment means using a three-factor nested design (ANOVA), and Duncan’s multiple range test at a 5% probability level according to Gomez, and Gomez[8].
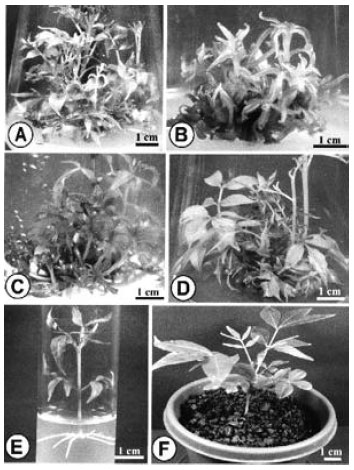
The effects of different concentrations, and combinations of cytokinin-auxin on multiple shoot induction were studied with using hypocotyl, cotyledon, leaf, and internode explants. These explants were cultured on MS media supplemented with different concentrations of BAP in combinations with two auxins, NAA, and IBA, for regenerating adventitious shoots. After 4 weeks of culture, all the cultured explants showed growth of callus with adventitious shoot buds from the cut ends (Fig. 1A-D). Morphology (colour, structure), and amount of callus varied with different concentrations, and combinations of BAP, and auxins. Among the different cytokinin-auxin combinations tested, BAP-NAA formulation showed a better performance than the other combination of BAP-IBA. Among nine plant growth regulator (PGR) combinations used in this experiment, the cultured explants produced adventitious shoots in eight combinations. Although the combination of 0.9 μM BAP, and 2.5 μM NAA failed to produce any adventitious shoot, but produced large amount of callus.
 | |
| Fig. 1: | Adventitious shoot formation from in vitro grown hypocotyl, internode, cotyledon, and leaf explants of Phellodendron amurense |
| A-D: | Proliferation of calli with development of adventitious shoots at the cut margins of hypocotyl (A), and internode (B) explants after four weeks of culture; cotyledon (C), and leaf (D) explants after six weeks of culture |
 | |
| Fig. 2: | Regeneration of plants from in vitro grown hypocotyl, internode, cotyledon, and leaf explants of Phellodendron amurense |
| A-D: | Multiplication, and elongation of adventitious shoots on hypocotyl (A), internode (B), cotyledon (C), and leaf (D) explants after nine weeks of culture in the medium containing 2.2 μM BAP plus 0.5 μM NAA |
It was recorded after 4 weeks interval of culture that percentage of shoot formation, total number of shoots per culture, number of usable shoots per culture, and average length of shoots from different treatment. Among various combinations of BAP-NAA, the highest regeneration response achieved with each explant was recorded in the medium containing 2.2 μM BAP plus 0.5 μM NAA after 6 weeks of incubation, and the maximum frequency of shoot bud differentiation was 97.5% for hypocotyl, 77.5% for cotyledon, 72.5% for leaf, and 62.5% for internode explants. Besides, another combination of 4.4 μM BAP plus 0.54 μM NAA also produced considerable percentage of shoots from four explants, 92.5, 72.5, 62.5, and 57.5%, respectively.
The maximum number of shoots per culture was 45.0 for hypocotyl, 36.0 for cotyledon, 24.4 for leaf, and 19.3 for internode explants, which were recorded with 2.2 μM BAP plus 0.5 μ NAA (Fig. 2A-D). Maximum of 31.1, 22.5, 6.1, and 7.5 usable shoots per culture were recorded from hypocotyl, cotyledon, leaf, and internode explants, respectively, with the same PGR combination. Highest average length of the elongated shoots were 50.2 mm for hypocotyl, 65.1 mm for cotyledon, 70.4 mm for leaf, and 47.5 mm for internode explants, which were observed on the medium containing 2.2 μM BAP plus 0.5 μM NAA.
Among BAP-IBA formulations, maximum of 75.0% explants produced adventitious shoots for hypocotyl, 71.3% for cotyledon, 51.2% for leaf, and 48.1 for internode explants, when they were cultured on the medium containing 2.2 μM BAP plus 0.5 μM IBA. Maximum number of shoots per culture was 32.4, 25.4, 18.1, and 15.3 for hypocotyl, cotyledon, leaf, and internode explants, respectively, on medium with 2.2 μM BAP plus 0.5 μM IBA. The number of usable shoots per culture was 22.4 for hypocotyl, 14.6 for cotyledon, 7.5 for leaf, and 5.6 for internode explants with the same PGR combination. In addition, average length of the elongated shoots were 38.6 mm, 48.0 mm, 58.7 mm, and 32.6 mm for hypocotyl, cotyledon, leaf, and internode explants, respectively, with 2.2 μM BAP plus 0.5 μM IBA.
Analysis of variance revealed significant differences among different explants, plant growth regulators, and their concentrations, which influenced the differences on percentage of adventitious shoot formation, total number of shoots, number of usable shoots, and average length of shoots per explant.
The analysis of variance indicated that the PGR, concentrations (C), explants (E), PGRxconcentration (PGRxC), PGRxexplant (PGRxE), concentrationsxexplant (CxE) were highly significant (P≤0.01) for the character of number of total shoots per culture, number of usable shoots per culture, and average length of shoots (mm). On the other hand, the interactions with PGRxCxE for all the characters were not statistically significant (Table 1). The highest mean value indicates that in respect of total number of shoots per culture, number of usable shoots per culture, and average length of shoots, all explants, and plant growth regulator combinations were statistically significant at 5% level according to Duncan’s multiple range test (DMRT) (Table 2). BAP (2.2 μM) plus NAA (0.5 μM) combination was significantly different from 2.2 μM BAP plus 0.5 μM IBA combination for all the characters. The regeneration efficiency significantly depended on explant type. High percentage of shoot formation, total number of shoots per culture, and usable shoots per culture were obtained in hypocotyl, followed by cotyledon, leaf, and internode explants in both plant growth regulator combinations, and differences among them were statistically significant. However, highest average length of shoots was obtained in leaf explant in both combinations of plant growth regulators, followed by cotyledon, hypocotyl, and internode explants, and also differences among them were statistically significant (Table 2).
Proliferated shoots were excised, and cultured on MS media containing different concentrations of IBA, NAA, and IAA for rooting. The percentage of root formation, number of roots per shoot, and length of the longest root were recorded after 5 weeks of the culture.
| Table 1: | Analysis of variance for percentage of shoot formation, number of total shoots per culture, number of usable shoots per culture, and average length of shoots in the hypocotyl, cotyledon, leaf,, and internode explants |
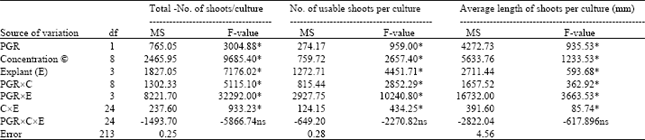 | |
| Asterisk indicates significance at 1% level, and ‘ns’ indicates non-significance. df = degree of freedom; MS = mean square. | |
| Table 2: | Comparison by DMRT among different plant growth regulators within different explants for highest mean number of adventitious shoot proliferation of P. amurense |
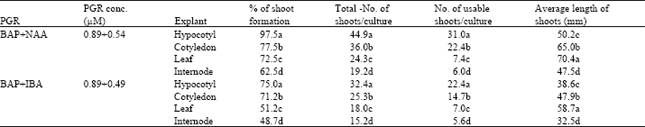 | |
| Each mean is based on four replicates. Values with different letters are significantly different from each other at 5% level according to DMRT | |
| Table 3: | Analysis of variance for percentage of root formation, number of total roots per shoot,, and average length of roots in microshoots cultured on media containing different concentrations of auxins for root induction |
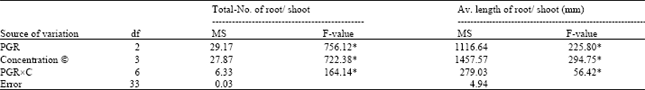 | |
| Asterisks indicate significance at 1% level. df = degree of freedom; MS = mean square. | |
| Table 4: | Effects of auxins on rooting of in vitro regenerated shoots of P. amurense on MS medium after incubation for 3 weeks |
 | |
| Means with different letters are significantly different from each other (P<0.05) according to DMRT | |
The rooting response to different auxins is shown in Table 4. Among three types of auxin, IBA was found to be comparatively more effective than the other two auxins, NAA, and IAA, for rooting. Among different concentrations, the maximal 97.5% of the culture produced roots when the micro-cuttings were cultured on the medium containing 2.0 μM IBA (Table 4) (Fig. 2E). Media containing NAA, and IAA also resulted in root formation whereas the rooting response was not as good as in the IBA-containing media. Highest number of 7.2 roots per rooted cutting, and the highest length of the longest root (50 mm) were recorded on 2.0 μM IBA-containing medium. These results indicate that percentage of root formation, and number of roots per shoot was highly influenced by concentrations, and type of auxin. Poor rooting was obtained with IAA at the concentrations tested compared to IBA, and NAA, though 4.0 μM IBA gave lower results on root-formation frequency, and number of roots per shoot (Table 4).
The results on analysis in variance of the two characters, total number of roots per shoot, and average length of the longest root are presented in Table 3. Rooting responses of explants were highly significant (P≤0.01), suggesting that they were largely influenced by PGR, concentrations (C), and their interaction (PGRxC). The significant differences among the means of different types of PGRs were evaluated with the help of DMRT. Among three types of PGRs, the highest values for all the characters were obtained with IBA, followed by NAA, and IAA, and the differences among them were significant (Table 4).
To acclimatize these in vitro developed plantlets, twenty of them were removed from culture tubes, and the gellan gum was washed away from the roots thoroughly with running tap water, then they were transferred to small plastic pot (9x8 cm) containing Kanuma soil for hardening under diffuse light (16h photoperiod). Normal growth of the potted plants was observed after 10-15 days of transfer. After 2 months, they were moved to another larger pot (25x22 cm) containing same soil, and transferred to greenhouse. The in vitro grown plantlets were gradually acclimatized, and successfully established on the Kanuma soil under greenhouse condition, with a survival rate of 90% (Fig. 2F), where they showed no morphological variation with mother plants.
PGR concentrations gave a marked effect on shoot regeneration from different explants of P. amurense. In general, percentage of shoot formation, number of total shoots, and average length of shoot per explant increase up to a certain concentration depending on the kind of PGR, and the explants[9]. A higher concentration of cytokinin gives profuse callusing, and reduction of shoot bud induction[10]. Hypocotyl of P. amurense is considered to be the best explant for adventitious shoot bud regeneration, which is in agreement with the findings of Mundhara, and Rashid[11]. Furthermore, hypocotyl explant has the advantages of easy manipulation, and of that large number of explants can be obtained from a single plant.
Shoot regeneration from cotyledon has also been achieved in a wide range of tree species using a BAP plus NAA combination[12]. Superior effect of the BAP plus NAA combination on adventitious bud proliferation from cotyledon has also been reported by Singh et al.[13] for Dalbergia sissoo. In the present study, it is found that BAP with NAA or IBA successfully produced adventitious shoots from cotyledon with the high percentage 77.5, and 71.3%, respectively, although cotyledon explant showed less shoot formation, and proliferation rates than hypocotyl explant. Similar results were reported in Panax ginseng[14].
Plant regeneration was also achieved from leaf explant of P. amurense on MS medium containing BAP with NAA or IBA whereas BAP plus IBA showed less effective than BAP plus NAA. Similar results were obtained on leaf segment culture of, and Platanus acerifolia[15].
Among various concentrations, and combinations of BAP, and NAA, internode explants of P. amurense showed the best result on the medium containing 2.2 μM BAP, and 0.5 μM NAA. Similar observations were reported on internode segment cultures of Adhatoda vasica[16]. It was also reported that BAP alone produced adventitious shoots from internode segments of Withania somnifera[17]. Kn-IBA, and Zn-IAA combination produced shoot buds from internode segments of Coccinia grandis[18], and beech[19], respectively. These results indicate that suitability of auxin-cytokinin combination for regeneration of adventitious shoots in vitro depends on the genotypic variation of the experimental explants.
In this study, the rooting response of microcuttings on an IBA containing medium was better than that in either NAA or IAA containing medium. Being of a stable nature, IBA is the preferred auxin for adventitious root initiation in many species e.g. Syzygium cuminii[20], Adhatoda vasica[16]. In addition, Hutchinson[21], and Litz, and Jaiswal[22] found IBA to be a superior auxin compared with IAA or NAA for the in vitro rooting of apple shoots. Success of auxin-free basal medium for efficient root induction is also reported by Mao et al.[23] In contrast to the present investigation, it has been reported that auxin is needed for the induction of roots in P. amurense[6]. The incidence of highly efficient root formation on auxin-free medium may be due to the availability of higher quantity of endogenous auxin in in vitro raised shootlets[24]. In the case of P. amurense, IBA is considered to be the best auxin for rooting from microcuttings.
The four explants used in the present study showed a wide, and pronounced range of variation for all the studied four characters. The mean, and standard error greatly varied among explants. In this experiment, it was confirmed that hypocotyl is best for shoot buds regeneration, followed by cotyledon, leaf, and internode. It was also found that leaf is best for producing the highest length shoots. The different results observed here may be depend on the different responses of explants to in vitro conditions, especially to plant growth regulators. The interaction between explant, and growth medium has been reported in many plant species[10].
In conclusion, an efficient protocol for adventitious shoot morphogenesis, and in vitro hardening of P. amurense regenerants has been described. The protocol was optimized by manipulations of different explants from axenic plants, and explants placement on the induction medium containing various concentrations and combination of BAP with NAA or IBA. Protocols described here provide a rapid plant regeneration system which could be used for the large scale micro propagation, conservation, and producing transgenic plants via an Agrobacterium-mediated system of genetic transformation of this species.