ABSTRACT
Effects of partial and complete ringing in the trunk on overall nutrient status of peach (Prunus persica Batsch. cv. `Hikawahakuho`) trees were examined by using one-year-old trees. The partial ringing comprised removal of 2 cm length bark and remaining 2 mm strip whereas complete ringing no remaining strip. Organic (sugar and starch) and inorganic nutrient content in leaves, stems and roots were monitored for eight weeks after ringing. The treatment reduced overall reduction in sugar and starch content in all plant parts. Each sugar component also showed similar trends. The N and Ca content in the above ground parts were lower in the ringed plants than control but the reverse was true in the roots. On the other hand, P, K and Mg content in all plant parts were declined by the treatment. Partial ringing prevented the translocation of 13C-photosynthates from the above ground parts to roots although entire inhibition was apparent in the complete ringing. Such reduction in energy sources of the roots seemed to affect overall plant nutrition which leads to small sized trees.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.498.507
URL: https://scialert.net/abstract/?doi=ajps.2004.498.507
INTRODUCTION
Small, compact, dwarfed or size controlled fruit trees provide easier pruning, thinning, spraying and harvesting, high production of high-grade fruit and lower cost of production[1]. The primary factor limiting the use of size controlling rootstocks in stone fruit production is the lack of suitable rootstocks with a wide range of compatibility among cultivars[2]. Jose[3] found lower vegetative growth in all the girdling treatments in relation to control in mango trees.
Arakawa et al.[4] reported that girdling increased and reduced growth of trunk girth both above and below the treatments in Fuji apple. Sitton[5] reported that the increase of trunk’s girth above the girdling might be caused by swelling of the trunk with accumulation of carbohydrates, while the reason for different responses among cultivars is not known. Schneider[6] stated that girdling blocks the translocation of sucrose from leaves to the root zone through phloem bundles. The block decreases starch content in root system and accumulation of sucrose in the leaves[7]. Martin[8] reported complete girdling can kill the plants. Rose and Smith[9] stated that complete girdling of apple stem killed the plants and partial girdling made weakening the plant itself.
Johnson[10] reported that photosynthates produced in the leaves of shrub trees are partially and completely stopped from reaching the roots that rely upon them by girdling. The roots are still supplying water and nutrients to the twigs, buds and leaves but are getting little to nothing in return. Girdling influences not only tree growth but also fruit quality[11,12]. Davie et al.[13] stated that girdling resulted in diminished N, P and Ca levels in the leaf in the girdled branches compared with ungirdled branches. This study was undertaken to know overall plant nutrition as affected by complete and partial ringing in the trunk of young peach trees.
MATERIALS AND METHODS
Experiment 1: The experiment was carried out in an orchard in the Ehime University Farm located in southern Japan, 33°57 N, 132°47 E at an elevation of about 20 m above sea level. There is mild temperate climate characterized by hot humid summer and cold dry winter. The soil is sandy loam with a pH of 5.7.
One-year-old peach (Prunus persica Batsch. var. ‘Hikawahakuho’) trees grafted on wild peach seedling rootstocks were used in this experiment on mid April 2001. The trees were spaced at 0.75x1.0 m in a completely randomized design. Weeding and irrigation were done at 7 day-interval and insecticides were applied when necessary. There were total 48 trees used in the experiment. The bark was cut 2 cm x 2 mm and 2 cm x complete cut (bark length removed x bark width remaining) after two months of planting. The treatments were control, 2 mm x 2 cm and complete ringing. Samples were collected 1st, 2nd, 4th and 8th weeks (June 20, June 27, July 11 and August 10) after bark cutting. Twelve (3x4) trees were uprooted and washed. New shoot, two-year-old shoot, leaves and roots were separated and oven dried (heated at 90°C for one h to terminate enzyme activity and dried at 60°C for three days), ground and used for analysis of carbohydrates (fructose, glucose, sorbitol, sucrose and starch) and mineral content.
Sugar content analysis: Five-hundred-milligram samples were taken in the test tubes and 20 mL of 80% ethanol was added to each sample. The test tubes were boiled at 80°C in a water bath for 2 h. Then the samples were filtrated and extracts were stored in the conical flask. This procedure was followed three times and extracts were mixed. The extracts were evaporated to dryness using a rotary evaporator. The residues were taken up in 10 mL of 80% ethanol and stored in the freezer until analysis. An aliquot of 20 μL sample was taken into the vial and dried. Then, 40 μL pyridine including TPB (1, 3, 5, tri-phenyl benzene) 1 mg mL-1 as an internal standard, 40 μL HMDS (hexamethyldisilazane) and 40 μL TMCS (chlorotrimethylsilane) were added to the dried samples. The vials were incubated at 60°C for 30 min. One μL of the trimethylsilated samples was injected into a gas chromatograph (GC). The GC conditions were as follows: column temperature was programmed from 150-265°C at the increment rate of 10°C min-1. The GC was equipped with a glass column (2.6 mm x 2 m) packed with 1.5% SE-30 coated on Chromosorb WAW DMCS (80-100 mesh) and N2 was used as carrier gas at the flow rate of 30 mL min-1.
Starch content analysis: Fifty-milligram units from the residues remaining after alcoholic extraction for sugar analysis sugar were taken in a test tube and 5 mL distilled water added to each sample. The tubes were heated at 90°C for 5 min in a boiling water bath to ensure that the samples were properly wetted. Then the samples were cooled to room temperature and 1 mL of amyloglucosidase solution (15 mg amyloglucosidase in 20 mL of Na-acetate buffer solution, 0.012 g mL-1) was added to each sample. The solutions were incubated in a shaking water bath at 40°C for one hour and an aliquot of 0.1 mL of each sample was taken in a clean test tube and mixed with 4.9 mL of anthrone solution (0.1 g of anthrone reagent in 76% H2SO4 to a volume of 100 mL). All tubes were placed in a boiling water bath for 15 min to accelerate color development. After cooling the samples to room temperature, absorbance was recorded at 620 nm using a Hitachi U-2001 spectrophotometer.
Mineral content analysis: One-gram of ground shoot and leaf samples placed into crucible and dry-ashed by heating in a muffle furnace for 5 h at 550°C. One ml of 20% HCl solution was added with ash and rinsed two times by using 2 mL (1+1 mL) distilled water. Finally 17 mL distilled water was added to make 20 mL solution. This stock solution was used to measure K, Ca and Mg by using an atomic absorption spectrophotometer (Shimadzu AA-6200, Kyoto, Japan). Phosphorus (as molybdate-reactive P) was estimated by colorimetric method at 620 nm (Hitachi U-2001, Tokyo, Japan). For N analysis 20 mg of samples were taken and analyzed by means of a CN coder (Sumitomo NC-80, Tokyo, Japan).
Experiment 2: The experiment was carried out in the same site.
Plant materials: Potted (φ 30 cm) one-year-old peach (Prunus persica Batsch. var. ‘Hikawahakuho’) trees grafted on wild peach rootstocks were used in this experiment in late July, 2003. The treatments were same as Expt. 1. There were total 9 trees used in the experiment.
13C labeling: The trees above the graft union were enclosed by transparent polyethylene bags. 13CO2 was generated by reacting 5.0 g BaCO3 with 50% lactic acid in the petri dish inside the bag. A small fan was used to circulate the air in the bag. Temperature in the bag during 13CO2 treatment was maintained at 25-30°C by intermittent misting over the bags.
Plant sample collection and 13CO2 analysis: 13C labeling was carried out for 3 h from 9:00 to 12:00 on July 31, 2003. Shoots, leaves, trunk and roots were collected separately after 24 h after 13CO2 feeding. The trees were uprooted and washed. The samples were oven dried (heated at 90°C for one h to terminate enzyme activity and dried at 60°C for three days), ground and used for analysis of 13CO2. One milligram of each sample was used to determine the isotopic ratio between 12C and 13C in the sample by combustion using an infrared 13CO2 analyzer (JASCO EX-130S; Japan Spectroscopic Co. Ltd., Tokyo). The 13C atom % excess was calculated by the subtraction of the concentration of 13C in nature from the measured 13C atom (%) in tissues fed with 13CO2.
RESULTS AND DISCUSSION
The effects of partial ringing and complete ringing on the sugar (fructose, glucose, sorbitol and sucrose) and starch content in new shoot and old shoot, leaves and roots of peach are presented in Fig. 1-6.
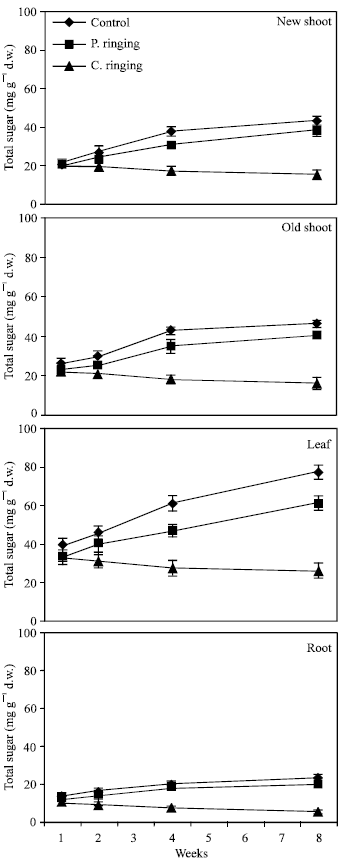 | |
| Fig. 1: | Effect of partial ringing on total sugars in new shoots (current year), two-year-old shoots, leaves and roots of peach trees. Vertical bars indicate SE (n = 4). Weeks are (1: June 20; 2: June 27; 4: July 11; 8: August 10). P. ringing = Partial ringing, C. ringing = Complete ringing |
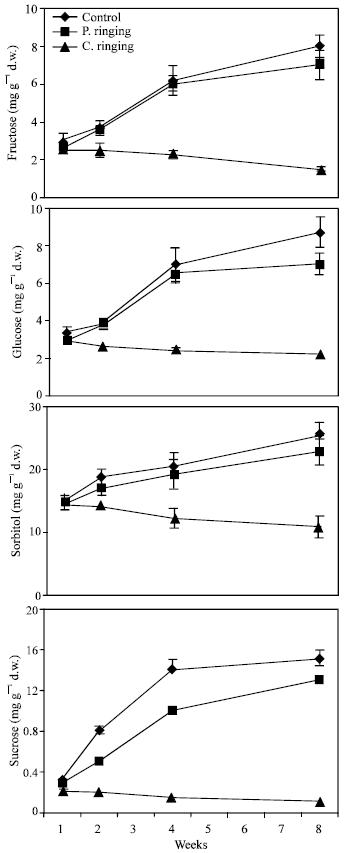 | |
| Fig. 2: | Effect of partial ringing on fructose, glucose, sorbitol and sucrose contents in new shoots of peach trees. Vertical bars indicate SE (n = 4). Weeks are (1: June 20, 2: June 27, 4: July 11 and 8: August 10) |
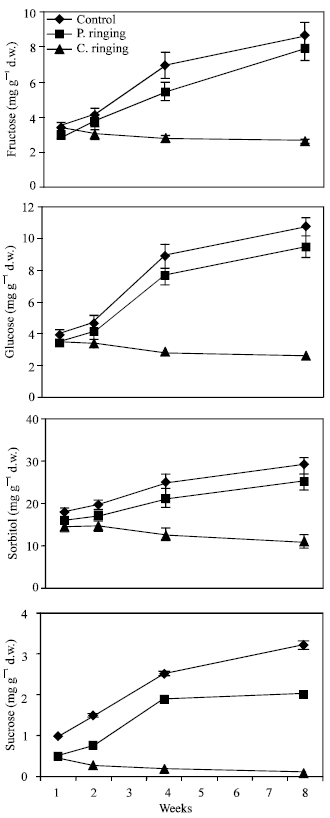 | |
| Fig. 3: | Effect of partial ringing on fructose, glucose sorbitol and sucrose contents in two-year-old shoots or peach trees. Vertical bars indicate SE (n = 4). Weeks are (1: June 20, 2: June 27, 4: July 11, 8: August 10) |
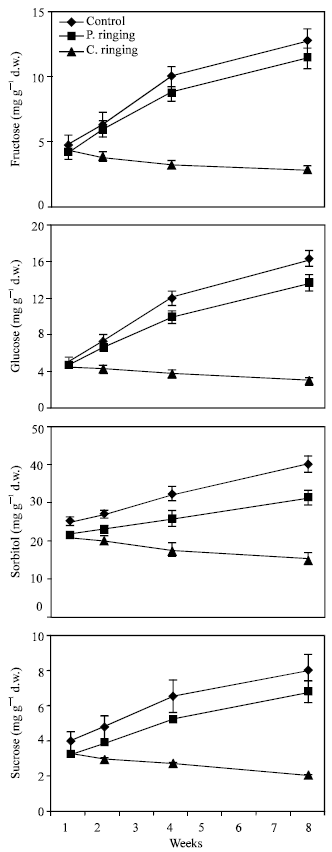 | |
| Fig. 4: | Effect of partial ringing on fructose, glucose, sorbitol and sucrose contents in leaves of peach trees. Vertical bars indicate SE (n=4). Weeks are (1: June 20, 2: June 27, 4: July 11, 8: August 10) |
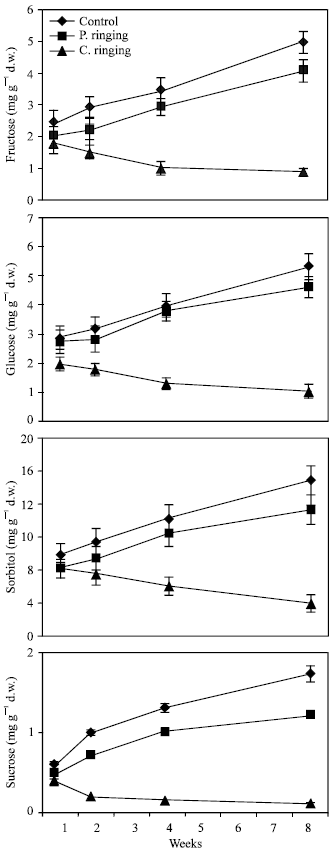 | |
| Fig. 5: | Effect of partial ringing on fructose, glucose, sorbitol and sucrose contents in roots of peach trees. Vertical bars indicate SE (n = 4). Weeks are (1: June 20, 2: June 27, 4: July 11, 8: August 10) |
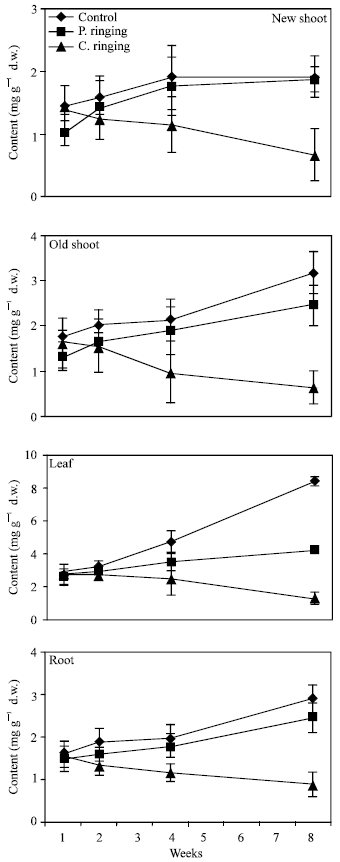 | |
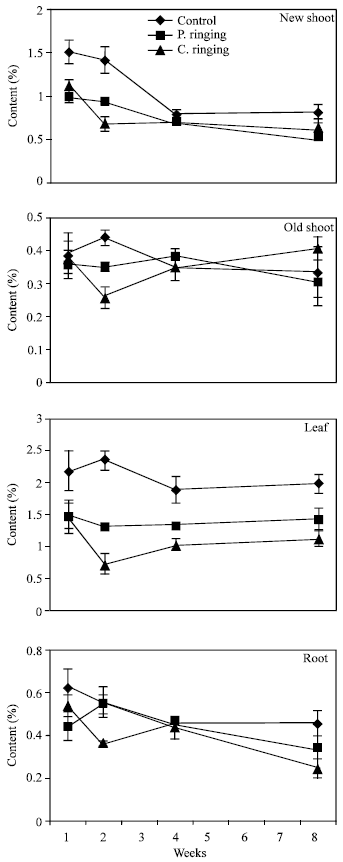
| Fig. 6: | Starch content of new shoots, old shoots, leaves and roots of peach trees as influenced by different ringing treatments. Weeks are (1: June 20, 2: June 27, 4: July 11, 8: August 10). Vertical bars indicate SE (n = 4) |
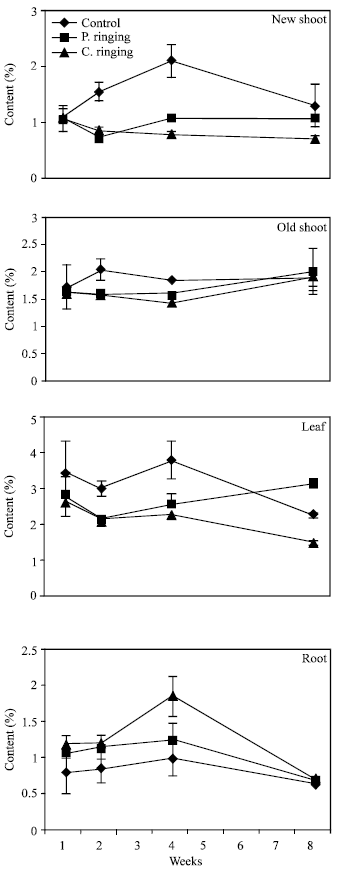 | |
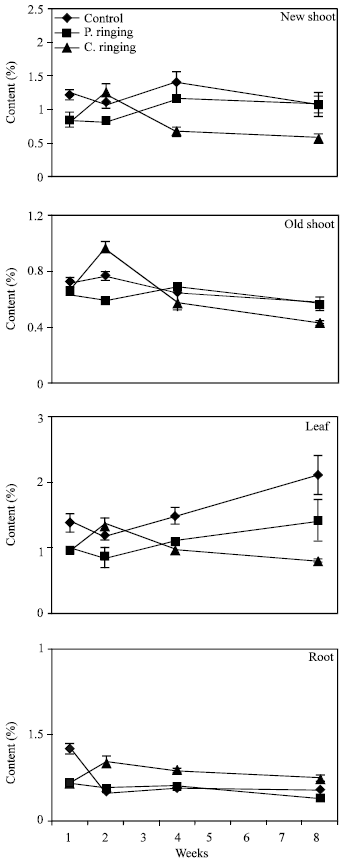
| Fig. 7: | Nitrogen content subjected to the ringing on new shoot, old shoot, leaves and roots in peach trees. Vertical bars represent SE (n = 4) |
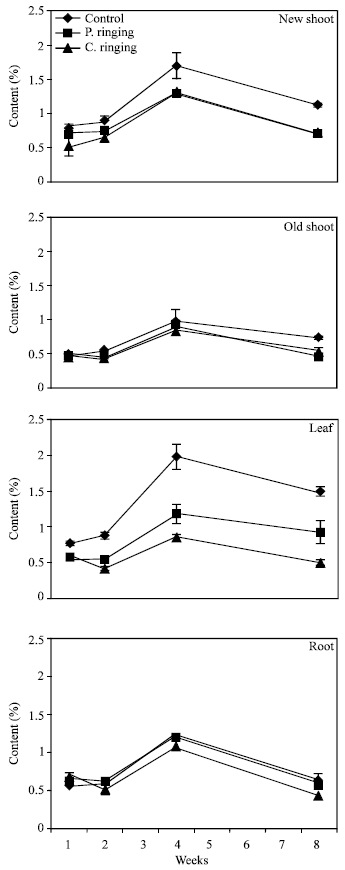 | |
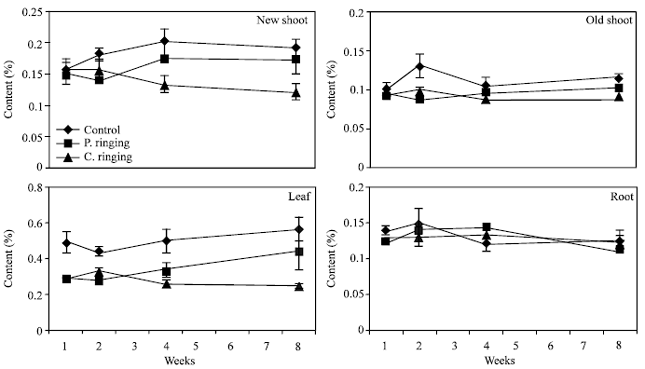
| Fig. 8: | Phosphorous content to the ringing on new shoot, old shoot, leaves and root. Vertical bars represent SE (n = 4) |
 | |
| Fig. 9: | Potassium content subjected to the ringing on new shoot, old shoot, leaves and roots. Vertical bars represent SE (n = 4) |
 | |
| Fig. 10: | Calcium content to the effect of ringing on new shoot, old shoot, leaves and roots. Vertical bars represent SE (n = 4) |
 | |
| Fig. 11: | Magnesium content subjected to the ringing on new shoot, old shoot, leaves and roots. Vertical bars represent SE (n = 4) |
 | |
| Fig. 12: | Photo shows 13CO2 feeding in potted (diameter 30 cm) one-year-old peach trees from 9-12 pm on July 31, 2003. A = Petri dish containing BaCO3 with 50% lactic acid, B = Small fan to circulate air in the bag. C = Potted peach trees covered with transparent polyethylene bags |
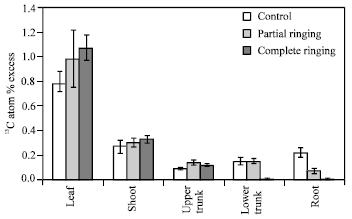 | |
| Fig. 13: | Effect of partial and complete ringing on the excess of 13C atom in different parts of peach trees after 13CO2 feeding on July 31, 2003. Vertical bars indicate SE (n = 3) |
Total sugar content was lower in the complete ringing than control trees (Fig. 1). In the control and partial ringing sugar content gradually increased, while there was a decline in sugar content in the complete ringing trees. Fructose, glucose, sorbitol and sucrose content are shown separately in Fig. 2-5. Each sugar was little bit lower in partial ringing than control trees. Sorbitol was the highest sugar component in all treatments examined. Starch was highest in control and lowest in complete girdling trees. Similar trend was found in starch content to sugar content (Fig. 6).
The percent N and Ca were lower in partial and complete ringing than control treatment in case of new shoot, old shoot and leaves, but it was higher in complete ringing in case of root (Fig. 7 and 10). It may be due to disconnect of bark between upper and lower parts of ringing. The percent P, K and Mg were lower in ringing treatments than control in case of new shoot, old shoot, leaves and roots (Fig. 8, 9 and 11).
In this experiment it was found that total sugar was lower in partially or completely ringed trees than control trees. It is clear that cutting the phloem prevented carbohydrate movement. Huguet et al.[14] reported that the phloemic flow brings sugars, its flow relative to other flows, changes the dry matter content and the quality of the fruits. Carbohydrate transport from leaves to roots smoothly through the phloem was suppressed because the phloem was reduced in width by ringing. It was reported that nutrient sap might diffuse laterally or vertically if normal phloem transport was checked by ringing cut[1]. In citrus, girdling caused a significant decrease in gibberellin level in the root system[15] and since gibberillins are presumed to be synthesized partly in the roots[16], the decrease may be attributed to the limited energy supply as a result of girdling. It might be that the reduced level of gibberellins lower α-amylase activity and thus prevents the hydrolysis of starch. The metabolic systems which may be involved in respiration, accumulation of 3-phosphoglyceric acid and pyrophosphorylase activity[17].
Complete ringing might affect physiolosical process of trees due to disconnection of cambial layer and phloem from upper to lower part of trees. Johnson[10] reported that photosynthates produced in the leaves are partially and completely stopped from reaching the roots. Gradually the root system declines to the point where it can no longer support the above ground parts. Arakawa et al.[4] and Elfving et al.[11] reported that Ca concentration in apple was reduced by girdling and mechanical ringing.
Davie et al.[13] reported that N, P and Ca levels were lower in leaves in the girdled branches compared with ungirdled branches. Johnson[10] reported that complete girdling is almost always fatal, even though death may take months to 2-3 years but the roots are still passing up water and nutrients through the undamaged wood in shrub trees. Arakawa et al.[4] reported that 90% girdling in apple trees can be reduced the growth. Our result shows that peach tree can be dwarfed by 96% girdling.
Fig. 12 shows 13CO2 feeding in one-year-peach trees. 13C atom % excess was higher in complete and partial ringing than control trees in shoots, upper parts of trunk and leaves, but it was lower in complete and partial ringing than control trees in case of root (Fig. 13). In the lower part of trunk there was no difference between control and partial treatments, whereas it was lower in complete ringing compared with control and partial ringing. So it is proved that there is no excess of 13C atom % from leaves to root due to disconnection between upper and lower part of ringing. From the present results we conclude that ringing affect overall nutrient status by preventing the supply of energy source (carbohydrates) to the roots.
ACKNOWLEDGMENTS
The authors are grateful to Ministry of Education, Culture, Sports, Science and Technology, Japan for providing A. B. M. Sharif Hossain and J. M. Onguso with Scholarship and to Mr. Atsushi Yokoi for his support in this research.
REFERENCES
- Jose, A., 1997. Effect of girdling treatments on flowering and production of mango trees cv. Tommy Atkins. Acta Horticulturae, 455: 132-134.
CrossRefDirect Link - Rose, M.A. and E. Smith, 2001. Overwintering Plants in the Landscapes. Horticulture and Crop Science, Ohio State University Extension, Columbus.
Direct Link - Arakawa, O., K. Kanno, A. Kanetsuka and Y. Shiozaki, 1997. Effects of girdling and bark inversion on tree growth and fruit quality of apple. Acta Hortic., 451: 579-586.
CrossRefDirect Link








