ABSTRACT
In the present study, Rhizobium was used as soil fertilizer to prevent the stress of lead on growing Pisum sativum. Three concentrations of lead were used (50, 100 and 200 ppm.) for the irrigation of growing plants (two times). After sowing, M2 seeds were collected, then cytological and biochemical studies were carried out. The cytological analysis revealed that soil enriched with Rhizobium increased the rate of mitotic division. A significant increase in mitotic index was detected. The kind of chromosomal abnormalities observed were; disturbances, stickiness, diagonals, chromosome breakage and c-metaphases. Also, a significant reduction in the percentage of abnormalities was observed. Biochemical studies included the analysis of M2 seed proteins using SDS-PAGE, isozyme analysis for both esterase and peroxidase and element analysis of Pb and N.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.416.424
URL: https://scialert.net/abstract/?doi=ajps.2004.416.424
INTRODUCTION
During the last two decades, increasing accumulation of heavy metals was observed in the Egyptian cultivated soils, lakes, River Nile and unfortunately even in air. These accumulations result from sewage, industrial wastes and several kinds of human activities. The increased accumulation of such pollutants in soil, water and air may cause a great annoying eutrophication problems for all living organisms including man not only in the polluted locations, but also in the non polluted areas[1].
Pollution with heavy metal elements received a great attention within the last few years. The interactions between such heavy metal elements and higher plants, particularly those of nutritive value for man and some edible animals is considered to be one of the most important problems in the field of biological research[1].
Lead was one of the first metals known to man as it was widely used for domestic industrial and medicinal purposes[2]. Evidence indicates that there is an increase in the Pb content of water and soil as a consequence of urbanization and industrialization[3,4]. The target area for Pb is the nervous system resulting in retardation of brain growth as lead inhibits certain enzymes in heme biosynthesis. In adults 10% of ingested Pb is absorbed and this ratio is increased to 40% in children[5].
Leguminous plants establish a symbiotic relationship with the soil bacterium Rhizobium. The symbiosis is manifested in the formation of root nodules. Also, in the interaction of rhizobia and plants one observes a high degree of host specificity (http://www.cnrs-gif.fr/isv/AK/introe.html).
Several studies dealt with the activities and role of Rhizobium in leguminous plants, nodule formation, the response of Rhizobium to salts and heavy metals the metabolic properties and stress tolerance of legume plants in the presence of Rhizobium[6-21].
The present investigation has been designed to evaluate soil enriched by Rhizobium and heavy metal effects on Pisum sativum plants. Three concentrations of lead acetate (50, 100 and 200 ppm) were used for soil irrigation (pure soil and soil enriched with Rhizobium). Cytological and biochemical studies were carried out after sowing. The cytological investigations were concerned with the mitotic division of M2 seed root tips. The biochemical studies included the SDS-PAGE analysis of M2 seed storage protein content, electrophoretic isozyme analysis of both esterase and peroxidase and finally the analysis of both Pb and N elements.
MATERIALS AND METHODS
The experimental plant used in this investigation was pea (Pisum sativum L.). Pure strains of the seeds were kindly obtained from the Egyptian Agricultural Organization and Vegetables Research Center, Ministry of Agriculture, Dokky-Cairo, Egypt. The heavy metal used was lead in the form of lead acetate Pb (CH3OO)2.
Treatments: Homogenous ten seeds of Pisum sativum L. were sowed in each pot (30 cm diameter) filled with clay loamy soil. Pot's soil were divided into two groups; the first group were pure clay loamy soil and the second group were mixed with Rhizobium leguminosarum (inoculated with Rhizobium leguminosarum) (enriched with Rhizobium) produced from the Egyptian Agricultural Organization and Vegetables Research Center (EAO and VRC) 10 g F-1. The pots were irrigated with tap water for weeks and then thinned to five uniform plants in each pot. Three concentrations of lead acetate were used for water irrigation (50, 100 and 200 ppm). Irrigation was done two times using these concentrations; the 1st time was after 25 days and the 2nd time was after 50 days. Pots of the control were irrigated only with tap water. After maturity and fruiting (75 days after planting), samples were collected for analysis of some elements (Pb and N), but for isozymes fresh leaves were used. Seeds of the first generation (M2) were collected for cytological and protein analysis.
Cytological studies: Cytological studies were carried out on root tips of germinated M2 seeds. Five root tips were taken of each treatment in addition to the control, then fixed in Crayon's fixative (1:3 acetic: alcohol) for 24 h and stored in 70% alcohol under refrigeration. The fixed roots were hydrolyzed for 6-8 min. in 1 N HCl at 60oC, stained in Feulgen stain and squashed in a drop of 45% acetic acid[22,23]. Mitotic index was calculated as the average number of dividing cells from 5 different root tips, for each treatment about 3000 cells were counted. Total percentage of abnormalities was calculated and types of abnormalities were photographed using Carl-Zeiss photomicroscope III at a magnification of x=2000.
Biochemical studies
Seed protein analysis: Seed proteins of Pisum sativum plants (M2) were analyzed using continuous polyacrylamide gel electrophoresis in the presence of sodium dodecyl sulphate (i.e. CONT-SDS PAGE) based on the method of Weber and Osbon[24], Laemmli[25]. For electrophoretic analysis the seed proteins were extracted in Tris-HCl pH 8.0 to obtain a good separation of polypeptide bands and electrophoresis in 12 and 12.6% polyacrylamide gel, respectively. Gel Pro-Analyzer software version 2.0 was used to determine the molecular weights of each protein band.
Isozyme analysis: The activity of some isozymes (Esterase and Peroxidase) were estimated in fresh leaves by the method of Harris et al.[26] and Brewer et al.[27]. Isozyme extraction from the samples was by homogenizing 0.5 g fresh leaves samples in 1 mL extraction buffer using a mortar and pestle. The extract was then transferred into clean eppendorf tubes and centrifuged at 10 000 rpm for 5 min. The supernatant was transferred to new clean eppendorf tubes and kept at -20°C until use for electrophoretic analysis.
Element analysis: For the analysis of elements (Pb and N) in dry weight of shoots the method of Gramberg et al.[28] was used.
RESULTS AND DISCUSSION
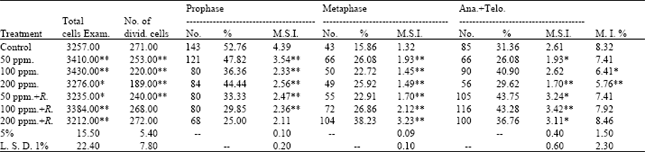
Cytological analysis: The frequencies of different mitotic phases of root tips of (M2) of Pisum sativum seedlings are shown in Table 1. With regard to the effect of lead on mitotic index, it appears that the mitotic index was decreased with all treatments of lead compared to that of the control. A high decrease in the M. I. was observed when the highest concentration (200 ppm) was used where it reached 5.769% as compared with the control value of 8.321%.
On the other hand, the mitotic index of root tips of (M2) of Pisum sativum seedlings observed after addition of Rhizobium for the soil and with the same treatments of lead was shown to be highly significantly increased at 200 ppm of lead (8.468%). However, the maximum value of mitotic index (8.468%) was recorded at 200 ppm+Rhizobium, whereas the minimum value of which (5.769%) was found at 200 ppm compared to the control value of 8.321%.
The relationship between the frequencies of mitotic stage indices (M.S.I.) and mitotic indices (M.I.) after treatment with the different concentrations of lead with Rhizobium in M2 seedlings are expressed in Table 1.
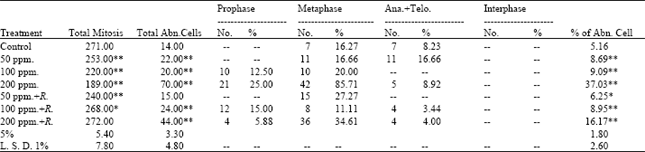
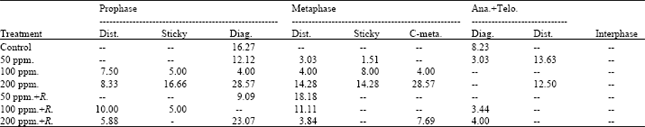
The obtained data presented in Table 4 indicated that the frequencies of mitotic abnormalities in root tips of (M2) seedlings of Pisum sativum varied according to the mitotic stage and the treatment applied. The abnormal prophase was observed with the high concentrations of lead and its frequency reached 25.000% (8.333+16.667) at 200 ppm of lead. Regarding the frequency of abnormal metaphase, the results showed that the low percentage of abnormalities (11.111%) was recorded at 100 ppm+Rhizobium. Abnormal ana+telophase were observed with high concentrations (100 and 200 ppm) used.
The maximum value of which (16.667%) was recorded at 50 ppm of lead and the minimum value (3.448%) was recorded at 100 ppm of lead+Rhizobium. The maximum value of abnormalities (85.714%) was recorded at metaphase stage at the treatment of lead of 200 ppm (Table 3).
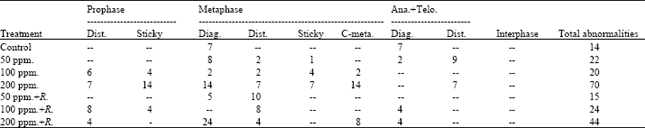
Different types of abnormalities in different mitotic stages were induced due to various lead treatments (Table 2 and Fig. 1). Diagonal was the most common type of abnormalities observed in metaphase and ana+telophase.
| Table 1: | Number and percentage of mitotic phases, mitotic stages index (M.S.I.) and the mitotic index (M.I.) of root tips of seedlings produced from M2 seeds of Pisum sativum |
 | |
| Table 2: | Number and types of abnormalities in interphase and in mitotic phases in M2 of Pisum sativum root tips |
 | |
| Table 3: | Mitotic index and percentage of the different mitotic phases in M2 of Pisum sativum seedlings |
 | |
| Table 4: | Percentage of abnormal mitotic phases and interphase of root tips of the seedlings produced from M2 seeds of Pisum sativum |
 | |
The highest value of abnormalities (37.037%) was recorded with the treatment of lead of 200 ppm, whereas, the lowest value (6.250%) was recorded with the treatment of lead (100 ppm)+Rhizobium.
Generally, adding of Rhizobium for soil treated with lead reduced the percentage of abnormalities compared with the recorded percentages of abnormalities in seedlings of M2 produced from treating the soil by lead only.
Disturbance and sticky were the only types of abnormalities observed in prophase stages. Within the metaphase stage, diagonal, disturbances, stickiness and c-metaphase were the types observed of abnormalities. Concerning the types of abnormalities of ana+telophase stages, diagonals and disturbance were observed (Table 2). The statistical analysis of the obtained data (Table 3) revealed that all treatments applied induced a highly significant increase in the percentage of abnormal mitosis except the recorded percent at the root tips produced from M2 of Pisum sativum seedlings treated with lead (50 ppm) + Rhizobium, the percentage of abnormal mitosis is significant only.
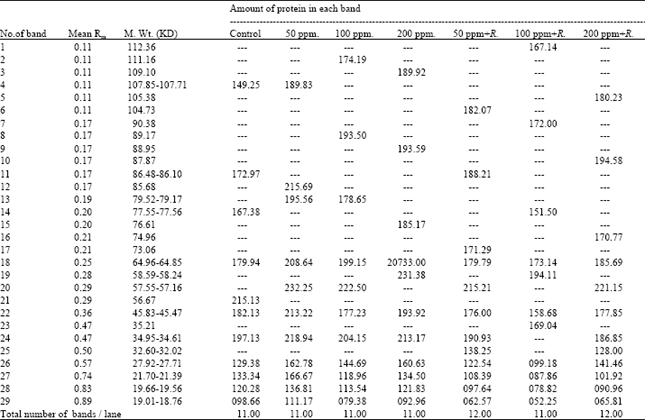
| Table 5: | Comparative analysis of relative treatment, mobility rate (Rm), molecular weight (M. Wt.) and amount of the protein bands in M2 seeds of Pisum sativum |
 | |
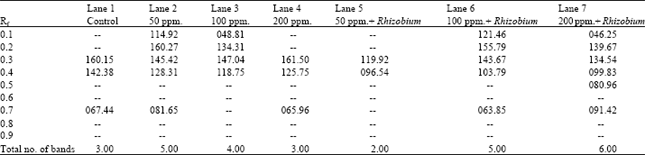
| Table 6: | Number and intensity of bands produced in esterase isozyme profile of the different treatments of Pisum sativum |
 | |
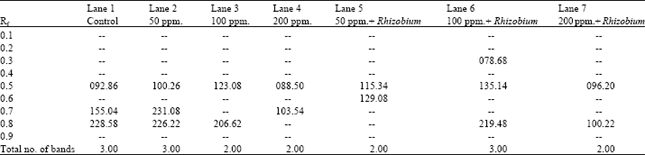
| Table 7: | Number and intensity of bands produced in peroxidase isozyme profile of the different treatments of Pisum sativum |
 | |
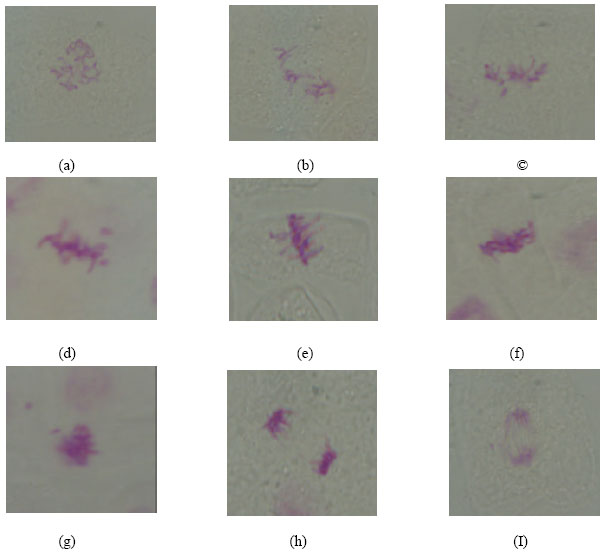 | |
| Fig. 1: | Types of abnormal phases observed in M2 root tips of Pisum sativum |
| (a) Disturbed prophase. (b) C-metaphase. (c) Disturbed metaphase with laggard chromosome. (d) Sticky metaphase with chromosome breakage. (e) Diagonal metaphase. (f) Sticky metaphase. (g) Sticky metaphase with chromosome breakage. (h) Diagonal anaphase. (I) Disturbed anaphase with chromosome breakage | |
Biochemical analysis
Seed protein electrophoresis: SDS-PAGE electrophoresis was carried out on M2 seeds of Pisum sativum after the different treatment using Tris-HCl (pH=8) as extraction buffer. The protein banding patterns are illustrated in Fig. 2 and the analysis of molecular weights, mobility rate and the amount of protein bands are recorded in Table 5.
The obtained data revealed that changes in protein patterns of Pisum sativum M2 seeds whose parent were previously grown in soil treated with different concentrations of lead or enriched by Rhizobium before the treatment by lead were limited. The total number of bands was 29 bands and the molecular weights of protein bands recorded ranged from 18.764 to 112.361 KDa. The highest molecular weight band (112.361 KDa) was recorded in M2 seeds produced from the treatment with 100 ppm lead in the presence of Rhizobium, whereas the lowest molecular weight bands (18.764-19.019 KDa) were observed in all protein profiles of M2 seeds.
The highest number of protein bands (12) was scored in both protein profiles of M2 seeds produced from the treatment of lead (50 and 200 ppm) in the presence of Rhizobium. On the other hand, the number of bands recorded in the remaining treatments was found to be similar (11 bands).
Comparing with the control, the recorded changes were expressed as variations in the number of separate bands, disappearance or appearance of certain bands, difference in the amount of one or more protein polypeptides and in band's relative mobility.
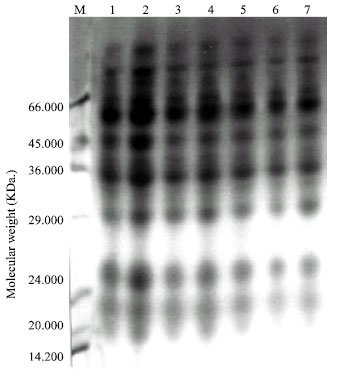 | |
| Fig. 2: | Protein profile of M2 seeds of Pisum sativum extracted in Tris-HCI buffer |
| M= Marker | |
| Lane (1): | Control |
| Lane (2): | M2 seeds collected from germinated Pisum sativum in soil treated with 50 ppm. lead |
| Lane (3): | M2 seeds collected from germinated Pisum sativum in soil treated with 100 ppm. lead |
| Lane (4): | M2 seeds collected from germinated Pisum sativum in soil treated with 200 ppm. lead |
| Lane (5): | M2 seeds collected from germinated Pisum sativum in Rihizobium soil treated with 50 ppm. lead |
| Lane (6): | M2 seeds collected from germinated Pisum sativum in Rihizobium soil treated with 100 ppm. lead |
| Lane (7): | M2 seeds collected from germinated Pisum sativum in Rihizobium soil treated with 200 ppm. lead. |
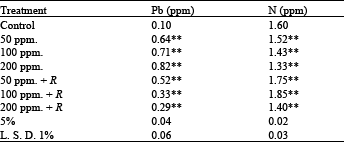
| Table 8: | Element content (μg g-1 fresh weight) at the different treatment of Pisum sativum plants |
 | |
Seed protein profile of the control M2 was found to have 11 bands. Seven bands of them (64, 45, 27, 21, 19 and 18 KDa) were found to be common in all treatments. On the other hand, the other five bands disappeared in certain treatments (Table 5).
M2 seeds produced from Pisum sativum grown in the soil treated with lead were found to have seven bands having the molecular weights; 111.169, 109.105, 89.177, 88.956, 85.685, 79 and 76.613 Kda, whereas M2 seeds produced from Pisum sativum grown in the soil enriched by Rhizobium and treated with lead were found to have nine new bands having the molecular weights; 112.361, 105.382, 104.739, 90.380, 87.878, 74.965, 73.062, 35.218 and 32 Kda.
Only two bands appeared in the yield produced from treated soil by lead enriched or not by Rhizobium. The first have the molecular weight of 58 KDa and presented in the concentration of 200 ppm lead and 100 ppm lead with Rhizobium. The second band having the molecular weight of 57 KDa and presented in all treatments by lead and Rhizobium except with the concentration by lead of 200 ppm and 100 ppm with Rhizobium.
Isozyme analysis: Esterase and peroxidase activities were studied electrophoretically in fresh leaves of germinated Pisum sativum in soil irrigated by lead concentrations enriched by Rhizobium or not.
Table 6 and Fig. 3 demonstrate the esterase banding patterns of Pisum sativum fresh leaves. A total of six bands were identified among the different treatments. The esterase banding patterns comprises two monomorphic bands as well as four polymorphic ones. Comparing with the control, there were remarkable variations in the number and intensity of bands. Only one band was observed in the profile of treated plants by 50 ppm lead in the presence of Rhizobium. On the other hand, six bands were recorded in the isozyme profile of treated plants by 200 ppm lead in the presence of Rhizobium in the soil. It is obvious that esterase isozyme electrophoretic patterns exhibited more new bands with the increase of lead concentration in the presence of Rhizobium.
Table 7 and Fig. 4 demonstrate the peroxidase banding patterns of the different treatments of Pisum sativum fresh leaves. Considering band 's number and intensity, it is obvious that there were remarkable variations between the control and soils enriched by Rhizobium or not. The number of bands is ranging from 2 to 3. The resulted banding patterns comprise only one monomorphic band as well as four polymorphic ones.
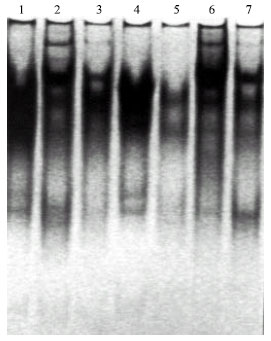 | |
| Fig. 3: | Asterase banding pattern of Pisum sativum sample |
| Lane (1): | Control |
| Lane (2): | Soil treated with 50 ppm. lead |
| Lane (3): | Soil treated with 100 ppm. lead |
| Lane (4): | Soil treated with 200 ppm. lead |
| Lane (5): | Rhizobium enriched soil treated with 50 ppm. lead |
| Lane (6): | Rhizobium enriched soil treated with 100 ppm. lead |
| Lane (7): | Rhizobium enriched soil treated with 200 ppm. lead |
Comparing with the control there was an increase in the intensity of the monomorphic band at all concentrations of lead in both enriched soil or not excepts in the treated plants by lead 200 ppm. germinated in soil not enriched by Rhizobium (Table 7).
Element analysis: The analysis of elements (Pb and N) in dry weight of shoots (Table 8) showed that there was a great reduction in nitrogen content, this decrease is proportional to lead concentration, but the inoculation with Rhizobium has adverse effect on nitrogen content, i.e. there was significantly increased in it. Dual inoculation resulted in the greatest effect.
Also, in case of lead uptake from the soil by plants, it is observed that, inoculation with Rhizobium deceased this uptake compared with uninoculated plants[6].Routray et al.[11], studied the effect of Rhizobium inoculation on growth and nodulation of cowpea in iron mine waste soil in a pot experiment and they found that shoot and root length, plant dry weight, number of nodules/plant and nodule dry weight increased with inoculation and were highest with a combination of the used two strains of Rhizobium.
 | |
| Fig. 4: | Peroxidase banding pattern of Pisum sativum samples |
| Lane (1): | Control |
| Lane (2): | Soil treated with 50 ppm. lead |
| Lane (3): | Soil treated with 100 ppm. lead |
| Lane (4): | Soil treated with 200 ppm. lead |
| Lane (5): | Rhizobium enriched soil treated with 50 ppm. lead |
| Lane (6): | Rhizobium enriched soil treated with 100 ppm. lead |
| Lane (7): | Rhizobium enriched soil treated with 200 ppm. lead |
They also found that the sand-amended soil gave better results than the mine soil alone. In the present work, soil enriched by Rhizobium recorded the highest growth as indicated by the values of mitotic division (M. I.) in germinated M2 seeds comparing with the control despite the lead stress.
Routray et al.[13], studied the response of cowpea (Vigna unguiculata) to inoculation with coselected vesicular arbuscular mycorrhizal fungi and Rhizobium strains in iron mine waste soil and they found that dual inoculations of Glomus fasciculatum with CC 50 Rhizobium strain increased growth, nodulation and rhizospheric microbial population of cowpea significantly compared with single strain inoculations.
Raizada et al.[16], studied the growth response of single (Rhizobium sp.) and dual inoculation of (Rhizobium sp. and VAM) on Albizia lebbeck, grown on eroded iron or mine soils under greenhouse conditions. They reported that, dual inoculation recorded significant improvements in seedling phytomass. They also concluded that concentration of N, P and K although higher in dual inculated plants, were not significantly different from no-inoculation, while seedling biomass and total N, P and K content (mg g-1) were significantly different. It is indicated that dual inoculation could be potential method for the improved establishment of tree seedlings under stressed environments of semiarid India.
Houngnandan et al.[20], studied the response of Mucuna pruriens to symbiotic nitrogen fixation by rhizobia following inoculation in farmer's fields in the derived savanna of Benin and they found that inoculation increased shoot dry matter by an average of 28% above the uninoculated treatments, but the increase depended on the field, location and year.
The obtained data from seed protein analysis, isozyme analysis and element content analysis in the present investigation support the previous concluion that the dual inocolation for soil which contain accumulation of heavy metals by Rhizobium could be a succsseive method to prevent this stress and improve the growth of legume plants.
REFERENCES
- Houngnandan, P., N. Sanginga, P. Woomer, B. Vanlauwe and O. van Cleemput, 2000. Response of Mucuna pruriens to symbiotic nitrogen fixation by rhizobia following inoculation in farmers fields in the derived savanna of Benin. Biol. Fert. Soils, 30: 558-565.
Direct Link - Weber, K. and M. Osborn, 1969. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J. Biol. Chem., 244: 4406-4412.
Direct Link - Laemmli, U.K., 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227: 680-685.
CrossRefDirect Link - El-Ghandour, I.A., M.A.O. El-Sharawy and E.M. Abdel-Moniem, 1996. Impact of vesicular arbuscula mycorrhizal fungi and Rhizobium on growth and PN and Fe uptake by faba-bean fertilizers and the environment. Proceedings of the International symposium held in Salamanca, Sept. 26-29, Fertilizer-Research Spain, pp: 43-48.
- Pal, S.C., M. Rahman, A.K. Podder, C. van Hove, Z.N.T. Begum, T. Heulin and A. Hartmann, 1996. The effect of heavy metals on legume- Rhizobium symbiosis-Biological nitrogen fixation associated with rice production. Proceedings of the Based on Selected Papers Presented in the International Symposium, Nov. 28-Dec. 2, Dhaka, Bengladesh, pp: 21-29.
- El-Mayas, H. and G.D. Hill, 1999. Selection of an effective strain of Rhizobium lupini on Lupinus nootkatensis Donn, used in reclamation of low temperture areas in Iceland. Proceedings of the Towards the 21st Century 8th International Lupin Conference, May 11-16, Asilomar, California, USA., pp: 16-19.
- Darlington, C.D. and L.F. La Cour, 1976. The Handling of Chromosomes. 6th Rev. Edn., Georg Allen and Unwin Ltd., London, UK., ISBN-13: 9780045740147, Pages: 201.
Direct Link








