Research Article
In vitro Response and Shoot Multiplication of Banana with BAP and NAA
Not Available
K.M. Nasiruddin
Not Available
M.A. Amin
Not Available
M.N. Islam
Not Available
Banana is an important fruit all over the world. It is a nutritious fruit rich in carbohydrates and a good sources of vitamins. It is generally propagated vegetatively through suckers. But the rate of multiplication through conventional method is slow and a number of viral diseases (Bunchy Top virus, Banana streak virus) and other diseases are also transmitted to new generation. Thus the productivity of fruits decreases and finally the yield become very poor which affects national economy of the country. In Bangladesh during the last few years the production of banana is decreasing gradually from 703314 tonnes in 1986-87 to 628425 tonnes in 1997-98 [1]. It may be due to unavailability of healthy and virus free suckers. Normally four to five suckers are obtained per plant which are insufficient to replace the affected farms with healthy germplasm. The area under cultivation as well as yield could be increased if appropriate technologies supplement conventional technologies. Meristem culture offers an efficient method for rapid clonal propagation, production of pathogen-free materials and germplasm preservation[2-4]. Meristem culture for clonal propagation and virus eradication was done by Gupta[10]. The present study was planned to establish a system for virus-free plantlet production and multiplication of BARI-I through meristem culture.
Banana (Musa sapientum) cultures were initiated on MS medium with different concentrations of BAP (0, 1, 2, 3, 4, 5 and 6 mg L-1). Four weeks old suckers field grown banana BARI-I were collected and the meristems were used for establishment of culture. The meristems were prepared from the collected suckers through dissection and removal of leaf sheath and outer tissue to the size of 3x3 cm. These blocks were surface sterilized with 70% ethanol to which few drops of Tween-20 were added. After sterilization these explants were further trimmed to 2-3 mm in size under aseptic condition and used as explants. To initiate the cultures, MS medium supplemented with BAP (0, 1, 3, 4, 5 and 6 mg L-1) was used. After three weeks shoots were about 10-15 mm in size. These shoots were transferred to the prior to autoclaving at 121°C for 20 min. Data were collected on the effect of different treatments on response of meristem to shoot proliferation and rooting. The experiments were arranged in completely Randomized Design (CRD) with 4 replications. Each of replications consisted of 6 culture tubes.
The highest percentage of survivability (91.67%) was recorded from MS medium containing 5 mg L-1 BAP whereas the lowest (58.33%) without hormone. Regeneration of single shoot from meristem explant have been shown in Table 1. The rate of single shoot regeneration was higher (62.50%) with 5.0 mg L-1 BAP over other treatments. Days required for single shoot regeneration (8-12 days) was more or less same for different concentrations of BAP. Rabbani et al.[5] and Rehana[6] found time required for single shoot formation (10-15 days) which was close to the present investigation.
| Table 1: | In vitro cultured meristem cv. BARI-I at different concentrations of BAP. Data were recorded at 20 DAI |
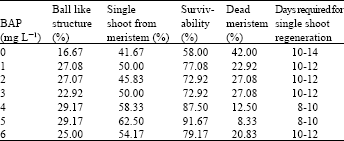 | |
Therefore, the single shoot formation from meristem had gradually increased with the increase of BAP concentration up to 5 mg L-1 and then declined at 6 mg L-1. The single shoot regenerated from meristem explant was thinner than shoot derived from shoot tip explant. The hard meristamatic ball like structures wee also developed from meristem explant, which was highest (29.17%) in MS medium supplemented with 5.0 mg L-1 BAP, whereas the lowest (16.67%) without hormone supplementation. Similar results were also obtained by Habib[7].
Meristem derived explant was then transferred to MS medium supplemented with different concentrations and combinations of BAP and NAA for subsequent shoot multiplication. Data were recorded at 10, 20 and 30 Days After inoculation (DAI). The results showed that 4.0 mg L-1 BAP gave the greatest number of shoots at 10, 20 and 30 DAI. The highest number of shoots proliferated (3.42/explant) at 30 DAI with 4.0 mg L-1 BAP and the lowest (1.28/explant) at 30 DAI was obtained from hormone free medium (Fig. 1). The main effect of NAA showed 1.5 mg L-1 NAA gave the highest number of shoots (2.93/explant) and the lowest (1.91/explant) from hormone free medium at 30 DAI.
The combined effect of BAP and NAA on shoot multiplication is shown in Table 2. Among the combinations and concentrations, BAP 4.0 + NAA 1.5 mg L-1 showed the highest shoot proliferation (4.52/explant) followed by BAP 5.0 + NAA 2.0 mg L-1 (3.83/explant) treatment at 30 DAI. The explants cultured without growth regulator did not produce any shoot. Khanam et al.[8], Robbani et al.[5] and Rehana[6] also reported similar results. Shoot length was significantly influenced by different BAP levels at 10, 20 and 30 DAI. The main effect of BAP and NAA showed the longest shoot (2.59 cm) from 4.0 mg L-1 BAP and (2.70 cm) from 1.5 mg L-1 NAA, respectively at 03 DAI.
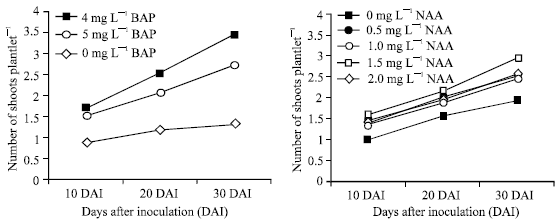 | |
| Fig. 1: | Main effect of different concentrations of BAP (left) and NAA (right) on shoot number of banana cv. BARI-I at different days after inoculation. Vertical bars represent LSD(0.05) values |
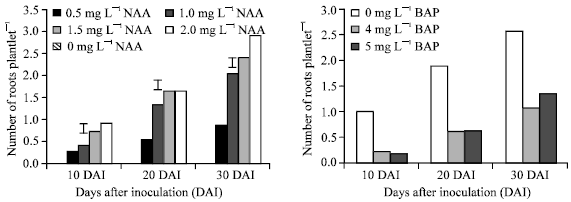 | |
| Fig. 2: | Main effect of BAP (left) and NAA (right) on root number of banana cv. BARI-I at different days after inoculation. Vertical bars represent LSD(0.05) values |
| Table 2: | Combined effect of different concentrations of BAP and NAA on growth of banana plantlet cv. BARI-I at different days after inoculation |
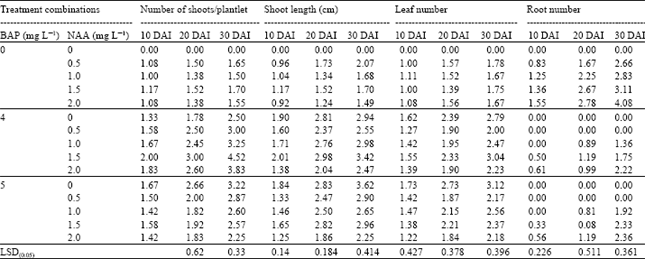 | |
Moreover, fluctuated results were observed when the media was supplemented with 0.5, 1.0 and 2.0 mg L-1 NAA. The smallest shoot (1.49 cm) was recorded with 2.0 mg L-1 NAA. Among the combinations, 5.0 mg L-1 BAP showed the longest shoot (3.62 cm) followed by BAP 4.0 + NAA 1.5 mg L-1 (3.40 cm) treatment. Rabbani et al. [5] found similar results from 5.0 mg L-1 BAP.
The main effect of BAP showed significant variation in leaf number of banana plantlets. The maximum number of leaves (2.50/plantlet) at 30 DAI was counted with 5.0 mg L-1 BAP which was identical with 4.0 mg L-1 BAP supplementation. The main effect of NAA in respect of leaf number was not significant at 10 and 20 DAI. But significant variation was found at 30 DAI, where maximum leaves (2.39/plantlet) obtained from 1.5 mg L-1 NAA and the minimum (1.97/plantlet) without NAA.
The combined effect of BAP and NAA showed that maximum number of leaves (3.12/plantlet) at 30 DAI produced with 5.0 mg L-1 BAP which was identical with the treatment of 4.0 mg L-1 BAP+1.5 mg L-1 NAA. The lowest number of leaves (1.67/plantlet) obtained from 1.0 mg L-1 NAA and 2.0 mg L-1 NAA supplemented MS media, respectively (Table 2).
The main effect of BAP showed the negative relationship between rooting and application, but NAA showed significant variation in root formation at different DAI. The highest number of roots (2.88/explant) at 30 DAI was obtained by 3.0 mg L-1 NAA and the lowest (0.89/explant) with 1.0 mg l-1 NAA, where no response was found in control treatment (Fig. 2). The combined effect of BAP and NAA showed that a small number of roots were produced by different combinations at different DAI. The highest number of roots (4.08/expoant) at 30 DAI was obtained with 2.0 mg L-1 NAA which is in support of the findings of Gubbuk and Pekmezci[9].
The present study concluded that the MS medium with 5.0 mg L-1 BAP is better for in vitro response including survivability of banana meristem, whereas 4.0 mg L-1 BAP+1.5 mg L-1 NAA should be recommended for higher shoot multiplicaiton.