ABSTRACT
In this study, development of anther wall of Helianthus annuus L. male fertile HA 89 “B” line and cytoplasmic male sterile HA 89 “A” line were compared by light and electron microscopy. It was observed that there was no difference between male fertile HA 89 “B” line and male sterile HA 89 “A” line development of anther wall until tetrads were formed. After tetrad stage in male fertile HA 89 “B” line, middle layer was lost and tapetum was parted from anther wall and surrounded the microspores. In cytoplasmic male sterile HA 89 “A” line middle layer became vacuolated and widened, tapetum also enlarged and filled anther sac. Besides it was determined that middle layer and tapetum were permanent with epidermis and endothecial cell during the further development of anther wall and also there was no secondary thickening in endothecial cells. In conclusion we suggest that the plasmodial tapetum which developed in fertile HA 89 “B” line transferred nutrients to microspores and helped them develop normally, but in cytoplasmic male sterile HA 89 “A” line the plasmodial structure did not develop. The inner tangential wall of tapetum, which was adjacent to the tetrads did not degenerate, so that sufficient nutrients were not transferred to microspores and thus they degenerated while in their callose walls.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.145.150
URL: https://scialert.net/abstract/?doi=ajps.2004.145.150
INTRODUCTION
Cytoplasmic male sterility is used in various crops for hybrid seed production. The first stable source of cytoplasmic male sterility (CMS) was discovered by Leclercq in 1969 from an interspecific cross involving Helianthus petiolaris Nutt. and Helianthus annuus L.[1]. Subsequent identification of genes for fertility restoration was discovered by Kinman in 1970 and allowed for efficient and economical production of hybrid seed[2]. The CMS system has been used for hybrid seed production in Helianthus annuus since 1972[3]. Male sterility in higher plants has been reviewed by Kaul[4]. He identified three types of male-sterility were recognized: 1. Genic male sterility (GMS) mutants often occur spontaneously; 2. cytoplasmic male sterility (CMS) generally arises through interspecific or intraspecific hybridization; 3. gene-cytoplasmic sterility involves both nuclear and cytoplasmic genes. There have been over 40 sources of CMS reported in Helianthus annuus since the original discovery by Leclercq in 1969[5]. There have been studies, conducted primarily at light and electron microscope levels, to ascertain aspects of anther wall (especially behavior of tapetum) and origin of the abortive process in CMS Helianthus annuus[6-8]. Horner[7] described 11 stages of microsporogenesis and microgametogenesis in the fertile Helianthus annuus, from premeiosis to engorged pollen stage. According to Horner[7], there was no more development after stage 5 in CMS line.
At the studies after the invention of male sterility, the period of microspore degeneration, early dissolution and persistent callose, the abnormal behavior of tapetum, endothecial and middle layers are still subjects of discussion in male sterile plants. The present study was compared histological aspects of anther wall and behavior of tapetum (which caused male sterility) in one N line sunflower with its CMS counterpart at both the light and electron microscopy levels.
MATERIAL AND METHODS
Helianthus annuus seeds were obtained from Trakya Agricultural Research Institute (Turkey). Fertile line, HA 89 B and CMS line, HA 89 A were selected. Plants were grown on experimental fields of Trakya Agricultural Research Institute. Flowers were dissected from closed and open inflorescences of both N and CMS lines to obtain anthers at all stages of development. Some anthers were fixed with a mixture of acetic acid-ethyl alcohol (1:3 v/v) for 24 hours and changed to 70% ethyl alcohol. After graded ethyl alcohol series, the anthers were infiltrated with xylene and embedded in paraffin (60oC). Sections from different developmental stages of anthers, cut 4 ìm thick with a rotary microtome, were stained with Delafield’s Hematoxylin[9]. For electron microscopy, anthers were prefixed in 3% glutaraldehyde in 0.1 M phosphate buffer for 2 hours. Prefixed anthers were washed in buffer and then postfixed 1% osmium tetroxide in the same buffer for 2 hours, washed with buffer, dehydrated in graded acetone series to propylene oxide and embedded in Epon 812.
RESULTS
In this study, 13 stages of microsporogenesis and development of anther wall were described in N and CMS lines of Helianthus annuus, from sporogenous mass stage to pistillate stage (Table 1).
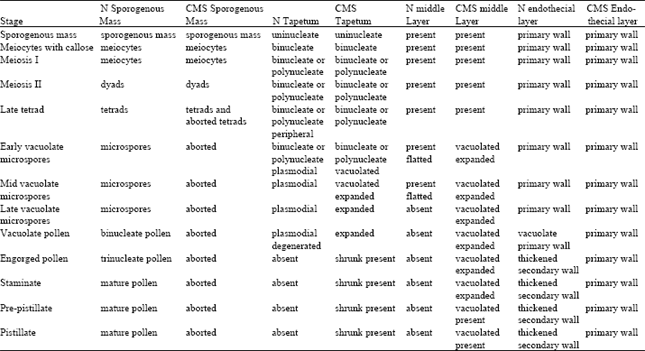
The anther wall of N HA 89 “B” line: At the sporogenous mass stage, the anther wall consists of four distinct layers; an epidermis, an endothecial layer, a middle layer and a tapetal layer (Fig. 1). The first nuclear divisions of tapetum cells occur before the meiosis. (Fig. 1). During the meiosis I, there was no change in epidermis and endothecium, However, the middle layer was flattened (Fig. 2). Different sizes and numbers of nuclei were observed as a result of secondary nuclear fusions and divisions in tapetum.
At the early vacuolate microspore stage, the tapetal layer was parted from the anther wall and the tapetal protoplasts were going through anther sac and surrounded the microspores (Fig. 3). This condition is called plasmodial or ameboidal type of tapetum behavior and is necessary for the development of microspores as pollen grains in sunflower. The tapetal protoplasts were mixed the microspores until the maturation of pollen grains (Fig. 4). In the late vacuolate microspore stage the mid layer disappeared.
At the vacuolate pollen stage, after the tapetum was disappeared, the endothecial cells widened radially, large vacuoles occurred in their cytoplasm and secondary thickenings occurred in their inner tangential walls (Fig. 5). At this stage, the connective tissues between adjacent locules of anther degenerated and at the staminate stage the anthers split open (Fig. 6).
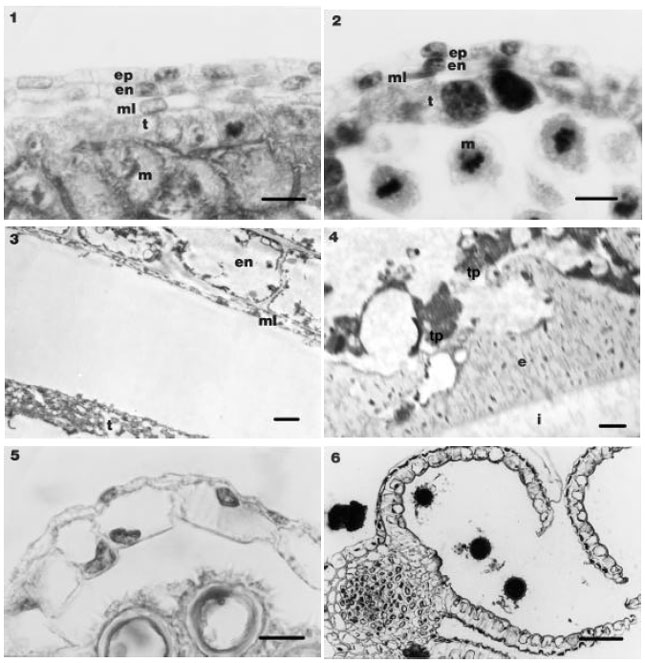
The anther wall of CMS HA 89 “A” line: Development of anther wall, up to tetrad formation was similar in N and CMS anthers (Fig. 7). After the late tetrad stage, the middle layer vacuolated and widened (Fig. 8). At this stage, the tapetum was also enlarged, but did not separate from anther wall. In early vacuolate microspore stage, the tapetum filled the anther sac and abortion of the microspores was completed. In the following developmental stage tapetal cells became small and the anther sac shriveled (Fig. 9).
| Table 1: | Stages of microsporogenesis and development of anther wall in fertile (N) and cytoplasmic male sterile (CMS) Helianthus annuus. |
 | |
 | |
| Fig. 1-6: | The anther wall in N anthers and meiocytes. 1. The anther wall in N anthers and meiocytes surrounded by callose (Bar=10 μm). 2. The anther wall in N anthers at meiose I stage (Bar=10 μm). 3. The anther wall in N anthers at early vacuolate microspore stage (Bar=3 μm). 4. The tapetal protoplast surround the microspores in N anthers at late vacuolate microspore stage (Bar=2 μm). 5. The tapetum is degenerated at N vacuolate pollen stage and endothecium cells contain secondary thickenings, (Bar=10 μm). 6. Opened locules at N staminate stage (Bar 50 μm) (en, endothecium layer; ep, epidermis layer; e, exine; ft, fibrous thickness; i, intine; m, meiocytes; ml, middle layer; t, tapetum; tp, tapetal protoplast). |
 | |
| Fig. 7-12: | The anther wall in CMS anthers. 7. The anther wall in CMS anthers and meiocytes surrounded by callose (Bar=10 μm). 8. The anther wall in CMS anthers at tetrad stage (Bar=10 μm). 9. The anther wall in CMS anthers at staminate stage (Bar=10 μm). 10. The anther wall in CMS anthers at pistillate stage (Bar=10 μm). 11. CMS anther shriveled and indehiscent (Bar=50 μm). 12. Inner tangential wall of tapetum is not degenerated at aborted microspores stage (arrow) (Bar=2 μ m) (am, aborted microspores; c, callose; en, endothecium layer; ep, epidermis layer; ml, middle layer; t, tapetum; tw, tapetum wall) |
Tapetal cells and middle layer cells were persistent until the wilted flower stages and the middle layer remained vacuolated and at this stage, vacuoles occurred also in epidermal and endothecial cells (Fig. 10). In endothecial cells no secondary thickenings occurred. The sterile anthers shriveled and were indehiscent (Fig. 11).
In CMS line, abortion occurred at the microspores stage while the inner tangential wall of tapetum was in contact with the tetrads was not disintegrated and the plasmodial structure that was seen in fertile line did not occur (Fig. 12). Thus sufficient nutrients were not transferred to microspores and they were degenerated in their callose walls.
DISCUSSION
Horner (7) described 11 stages of microsporogenesis in the fertile Helianthus annuus, from premeiosis to engorged pollen stage. He observed that no further development occurred within the CMS anthers beyond stage 5. He also reported that within a short time, the tapetum and tetrads were completely disorganized and abortion was completed. In our study, 13 stages of microsporogenesis and development of anther wall in N and CMS lines of Helianthus annuus, from premeiosis to tricellular mature pollen stage were described. After the stage 5, in the CMS anthers were revealed behaviors of layers tapetal, middle and endothecial and it was observed that tapetal layer was persistent until the wilted flower stage.
In male sterile plants, behavior of tapetum is remarkable. Many researchers[10-14] reported different behaviors in tapetal cells. Bino[10], in approximately 5% of tapetal cells has observed formation of a cell wall after karyokinesis. The others of tapetal cells were vacuolated and the cytoplasm disorganized. Formation of vacuoles in tapetal cells of sterile plants may manifest disturbance their functions[10]. A similar situation is observed in two genic male sterile of Zea mays, in which a cell wall is formed after karyokinesis[14]. Katti et al.[12] reported that the tapetum is persistent even within the fully mature sterile anther in Cajanus cajan (L.) Millsp. In CMS onions were found three types of abnormal tapetal behavior[11]. Also in genic male sterile soybean, tapetum displayed various behaviors[13]. Nakashima and Hosokawa[15] reported that the tapetum of the GMS sunflower remained peripheral enlarged and did not lose its cell walls at the time that the N line tapetum became plasmodial. This persistence was suggested as a cause of abortion[15]. The tapetum seems critical in the abortive process and its malfunctioning is often regarded as the direct or indirect cause of CMS[7,10,14,15]. In the present study, N line tapetum was plasmodial, in CMS line it did not separate from anther wall and inner tangential wall did not break down. For this reason sufficient nutrient were not transferred to microspores and they were degenerated in their callose walls. It was observed that tapetum was persistent until wilted flower stage.
In present study, N line at the stage the tapetum disintegrated, the inner tangential walls of endothecium cells had secondary thickenings and in CMS line endothecium cells had no fibrous thickenings. In many sterile plants, failure of endothecium development is attributed to the inhibition caused by the persistent tapetum[16]. According to Chauhan[16] in most of the anthers of GMS and CMS plants endothecium cells did not widen and no fibrous thickenings were observed. Because persistent tapetal cells continue to produce the inhibitor that checks the differentiation of endothecium[16]. However, Katti et al.[12] reported that in GMS Cajanus cajan (L.) Millsp. even though the persistent tapetum, endothecium was well developed. Also in Glycine max[17] and Zea mays[14] endothecial cells had elongated radially and developed secondary thickenings both in the N and CMS anthers.
In conclusion, it was determined that the persistent tapetum prevented the development of endothecium. It was postulated that the malfunctioning of tapetum is a cause for the induction of male sterility.
ACKNOWLEDGMENTS
We would like to thank in advance to Trakya Agricultural Research Institute for providing us the material. This study is a part of Ciler Meric's Ph.D. thesis and it was supported by The Scientific Research Fund of Trakya University (Project No: TUBAP-364).
REFERENCES
- Horner, Jr. H.T., 1977. A comparative light-and electron-microscopic study of microsporogenesis in male-fertile and cytoplasmic male-sterile sunflower (Helianthus annuus). Am. J. Bot., 64: 745-759.
Direct Link - Meric, C., F. Dane and G. Olgun, 2004. Histological aspects of anther wall in male fertile and cytoplasmic male sterile Helianthus annuus L. (Sunflower). Asian J. Plant Sci., 3: 145-150.
CrossRefDirect Link - Holford, P., J. Croft and H.H. Newbury, 1991. Structural studies of microsporogenesis in fertile and male-sterile onions (Allium cepa L.) containing the cms-S cytoplasm. Theor. Applied Genet., 82: 745-755.
CrossRefDirect Link - Katti, R.Y., H.S. Giddanavar, S. Naik, S.N. Agadi and R.R. Hedge, 1994. Persistence of callose and tapetum in the microsporogenesis of genic male sterile Cajanus cajan (L.) Millsp. with well formed endothecium. Cytologia, 59: 65-72.
Direct Link - Jin, W., H.T. Horner and R.G. Palmer, 1997. Genetics and cytology of a new genic male-sterile soybean (Glycine max (L.) Merr.). Sex. Plant Reprod, 10: 13-21.
Direct Link - Chaubal, R., C. Zanella, M.R. Trimnell, T.W. Fox, M.C. Albertsen and P. Bedinger, 2000. Two male-sterile mutants of Zea mays (Poaceae) with an extra cell division in the anther wall. Am. J. Bot., 87: 1193-1201.
Direct Link - Nakashima, H. and S. Hosokawa, 1974. Studies on histological features of male sterility in sunflower (Helianthus annuus L.). Proc. Crop Sci. Soc. Jap., 43: 475-481.
Direct Link - Ilarslan, H., H.T. Skorupska, H.T. Horner and P.G. Palmer, 1997. Cytology and genetics of a tissue culture-derived soybean genic male-sterile, female-sterile. J. Heredity, 88: 129-138.
Direct Link - Fick, G.N. and J.F. Miller, 1997. Sunflower Breeding. In: Sunflower Technology and Production, Schneiter, A.A. (Ed.). ASA, CSSA, SSSA Inc., Wisconsin, USA., pp: 395-439.
Direct Link








