ABSTRACT
The effect of seed priming in culture filtrates of two blue-green algae namely Cylindrospermum muscicola and Anabaena oryzae (F1 and F2 respectively) on different growth criteria of Lupinus termis plants grown in siliceous soil collected from Gebel-El-Nargis was investigated. As compared to control plants, seed presoaking in the culture filetrate Cylindrospermum (F1) increased (P≤0.05) the photosynthetic activity, growth criteria, content of nitrogenous compounds and carbohydrates in the shoot of tested plant. On the other hand, these parameters seemed to be non-significantly affected when the seeds were presoaked in F2. In the majority of cases, seed pretreatment with F1 or F2 increased (P≤0.05) chl.a, chl.b, total chlorophylls and total pigments, whereas a reversed effect was manifested in carotenoid contents. The two culture filtrates; particularly F1 caused significant increase in auxin, gibberellic acid and cytokinin, whereas abscisic acid showed a reverse situation. Amylase, protease and aminotransferase activities were progressively increased in response to presoaking in the algal filtrates. Protein banding patterns generally showed disappearance of a 34 and 42 KDa protein and induction of 129 and from 3-8 KDa by F1 treatment, meanwhile certain protein (M wt: 18 KDa) were only induced following treatment with F2 but not occurring in either the control or F1 treatment. Application of both F1 or F2 increased the intensities of protein bands from 20-32 KDa.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2003.944.951
URL: https://scialert.net/abstract/?doi=ajps.2003.944.951
INTRODUCTION
Blue-green algal extract excretes a great number of substances that influence plant growth and development (Ordog, 1999). These microorganisms have been reported to benefit plants by producing growth promoting regulators (the nature of which is said to resemble gibberellin and auxin), vitamins, amino acids, polypeptides, antibacterial and antifungal substances that exert phytopathogen biocontrol and polymers, especially exopolysaccharides, that improve plant growth and productivity (Zaccaro et al., 1999). Moreover, Zaccaro et al (2001) reported that, foliar application of biochemical organic substances, which supply macro and micro nutrients, of increased demand.
Fresh water algae contain high percentage of macro and micronutrients bounded in their major biochemical constituents and metabolites such as carbohydrates and proteins (Wake et al., 1992). In this respect, Adam (1999) found that algal filetrate of the cyanobacterium Nostoc muscorum significantly increased germination of wheat seeds as well as their growth parameters and nitrogen compounds, compared to controls. Also, Lozano et al. (1999) stated that, the application of an extract from algae to soil or foliage increased ash, protein and carbohydrate contents of potatoes (Solanum tuberosum).
Recently, Ghallab and Salem (2001) studied the effect of some biofertilizer treatments; Cerealin (Azospirillum spp.) and Nemales (Serratia spp.) on wheat plant, in field experiment and found that the two biofertilizers increased growth characters and nutrients, sugar, amino acids and growth regulators (IAA, GA3 and cytokinin) and crude protein content in the plant. On the other hand, Abdel-Monem et al. (2001) reported that fertilization with Azospirillum brasilense or commercial biofertilizer Cerealin, improve the growth and yield of maize in rotation with wheat as affected by irrigation regime.
In the light of the abovementioned reviews, it was of particular interest to investigate the effect of the two algal filtrates; Cylindrospermum muscicola and Anabaena oryzae as F1 or F2 respectively on growth and some related enzymes, pigments, photosynthetic activity, nitrogen and carbohydrate content, protein patterns as well as growth regulators content of Lupinus termis plant grown in siliceous soil.
MATERIALS AND METHODS
Pure strain of Lupinus termis (var. Balady) seeds were surface sterilized with 0.01% HgCl2. The seeds were soaked in water (control) or the algal filetrate of Cylindrospermus muscicola (F1) or Anabeana oryzae (F2); each separately, for about 6 hours, the seeds washed thoroughly with tap water. The seeds were then sown in earthenware pots filled with equal amounts of the soil which collected from Gebel-El-Nargis. As described by Mashaly (1987), Gebel El-Nargis sand dunes is a very huge sand formation (with height ranges between 25-30 m) belonging to Baltim area in the Deltaic Mediterranean coast of Egypt. It is a siliceous soil characterized by low total plant cover; the physical and chemical analyses of siliceous soil is shown in Table 1 .The pots were kept in greenhouse under a normal day/night conditions and irrigated with equal amounts of tap water when required.
To prepare the two algal filtrates used in this study, the dinitrogen fixing blue-green algae which isolated from cultivated soil samples collected from Delta region, were grown in BG11-free nitrogen-media (Rippka et al., 1979).
Isolation and purification of cultures were carried out using the technique adopted by Venkataraman (1969). Identification of the maintained axenic cyanophytes was carried out according to Desikachary (1959) and Whiteford and Schnumacher (1973). The isolated organisms. Batch culture (100 ml) were grown autorophically in Erlenmeyer flask (250 ml) and were initiated with inocula of 7-days old algal cultures. The cultures were incubated at 28°C±1 at light intensity of about 3000 Lux. for 15 days then separation of the used algal filtrates takes place by filtration. These obtained filtrates used for seeds pretreatment described before (Table 2).
Samples from each set were taken after 18 days from sowing. The samples were ten replicates for measuring shoot length and total leaf area and triplicates for chemical analysis. The results were first subjected to an analysis of variance (ANOVA); a test for significant differences between means at P≤0.05 was performed using LSD (Snedecor and Cochran, 1980).
Extraction and measurements of leaf pigments were carried out as recommended by Kissimon (1999).
For Hill reaction assay, detached leaves were ground using a chilled blender, in 50 mM MgCl2 and the resulting homogenate was suspended in 1 mM Na-Tricine, 10 mM NaCl and 10 mM MgCl2 solution and kept at -4.0°C (Arnon, 1949). Photosystem II activity was determined as the rate of 2, 6-dichlorophenol indophenol (2, 6-DCPIP) photoreduction (Trebst, 1972) using a spectronic 21 D Spectrophotometer.
Extraction, clarification and determination of saccharides were carried out following the procedure adopted by Younis et al. (1969).
Nitrogenous constituents were extracted and determined by the method of Yemm and Willis (1956) as described by Haroun (1986).
Amylase and protease enzymes were extracted by Kar and Mishra (1976). The activities of amylase and protease were determined according the methods of Monroe and Preiss (1990) and Salmia et al. (1978) respectively.
| Table 1: | Mean of 5 samples representing the composition of the experimental soil |
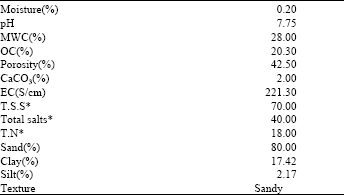 | |
Extraction and activity of aminotransferase was determined as μg ketoacids according to Dey and Harborne (1990).
The extraction and separation of plant growth substances followed the procedures described by Shindy and Smith (1975).The bioassay of auxin is determined as described by Foda and Radwan (1962). For measurement of gibberellin substances, the method adopted by Frankland and Wareing (1960) was followed. The technique used to assay the activity of cytokinins was that described by Esashi and Leopold (1969). Abscisic acid was bioassayed by the test recommended by Wright (1969).
For protein banding patterns the electrophoretic protein profiles of Lupinus shoot were analyzed by SDS-PAGE technique (Laemmli, 1970). Data were analyzed and identified by gel documentation system (GDS) with comparing polypeptide maps. Percentages of band intensity, molecular weight and mobility rate of each polypeptide in relation to standard markers using Gel-Pro Analyzer version 3 Media Cyberne Tice Imaging Experts Software.
RESULTS AND DISCUSSION
Cyanophyceae are a diverse group of prokaryotes, it distributed worldwide and improve the growth and development of the plants, with which they share the habitat, because they: 1- contribute to soil fertility in many ecosystems, 2- produce various biologically active substances and 3- have higher efficiency in biosorption of heavy metals i.e. bioremediation (Zaccaro et al., 2001). Moreover, Hedge et al. (1999) documented that, application of algal biofertilizers is useful for the reclamation of marginal soils such as saline-alkali and calcareous soils.
In this context, pretreatment of Lupinus termsi seeds in the two algal filtrates, led to significant increase (P≤0.05) (in response to F1) and non-significant effect (in response to F2) in shoot length, total leaf area and shoot fresh mass.
| Table 2: | Major chemical composition of cylindrospormum muscicola(F1) and Anabaena oryzae(F2) filtrates |
 | |
| Fig. 1: | Effect of algal filtrates (F1 or F2) on shoot criteria of Lupinus termis Vertical bar= value of LSD at p=0.05 |
Meanwhile, the dry mass of shoot increased (P≤0.05) by the two treatments, as compared to the control plants (Fig. 1a and b). This may be attributed to the secretion of plant growth regulators by the experimental organisms in the culture filtrates (Table 2). These results were in good harmony with those obtained by Adam (1999) and Mohiuddin et al. (2000). Ghallab and Salem (2001) stated that, in wheat plant, growth characters and nutrients, sugar, amino acids and growth regulators (IAA, GA3 and kinetin) as well as crude protein contents of the tested plant, increased by using biofertilizers; Cerealin (Azospirillum spp.) and Nemales (Serratia spp.). Furthermore, the height of cotton plant and their dry weight were increased by presoaking the seeds in Nostoc muscorum extract (Likhitkar and Tarar, 1995).
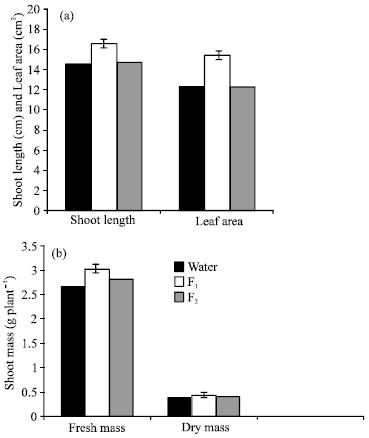
Presoaking of Lupinus seeds in F1 or F2 resulted in marked increases (P≤0.05) in chl.a, chl.b, total chlorophylls and total pigments content of the leaves, except for chlorophyll a and total pigments which non-significantly affected when the seeds were presoaked in F2 (Fig. 2a).
 | |
| Fig. 2: | Effect of algal filtrates (F1 or F2) on photosynthetic Pigments and Hill activity of Lupinus termis shoot |
On the other hand, carotenoid contents decreased significantly and non-significantly in response to treatment with F1 and F2 respectively. The stimulative effect of these filtrates could be attributed to elevated level of GA3 of the filtrates which is known to inhibit chlorophyllase activity (Drazkiewicz, 1994). Moreover, Wake et al. (1992) stated that cyanobacterial extract enhanced chlorophyll formation. Recently, Ordog (1999) documented that the suspension of extract of cyanobacteria and microalgae contain a special set of biologically active compounds including plant growth regulators, which can be used for treatment to decrease senescence, transpiration as well as to increase leaf chlorophyll, protein content and root and shoot development.
 | |
| Fig. 3: | Effect of algal filtrates (F1 or F2) on carbohydrate and nitrogen contents of Lupinus termis shoot |
The increase in pigment production in Lupinus leaves pre-treated with the algal filetrate of Cylindrospermum led to increase the photosynthetic activity and carbohydrate contents (Fig. 2a,b and 3a respectively) in plant tissue. In conclusion, these results may be attributed to increase photosynthetic electron transport (Bograh et al., 1997), stimulation of pigment biosynthesis (Younis et al., 1991) and interaction with the thylakoid membrane surface (Zheleva et al., 1994).
Presoaking of Lupinus termis seeds in the two filtrates, particularly F1 showed marked increase (P ≤ 0.05) in the photosynthetic characteristic representing by Hill activity (Fig. 2b). This is expected results due to the increase in the chlorophylls concentration in response to the algal filetrate treatments (Fig. 2a).
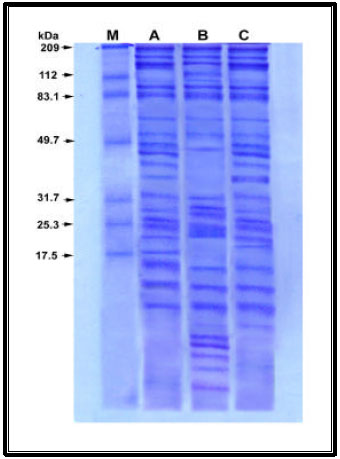 | |
A= control, B= F1, C= F2 | |
| Fig. 4: | Effect of the algal filtrates (F1 or F2) on protein Patterns of Lupinus termis shoot |
In this connection, Aldesuquy and Gaber (1993) found that, the increase in chlorophylls content was associated with an increase in Hill activity of Vicia faba plant under various conditions.
The increase in the photosynthetic activity by the algal filtrates is comparable with the finding of El-Shahaby (1992) who stated that growth regulators pretreatment were significantly increased the photosynthetic activity of stressed and unstressed Vigna sinensis plants and this may be due to the effect on the stomata and the photosynthetic apparatus (Roschke and Hadrich, 1985).
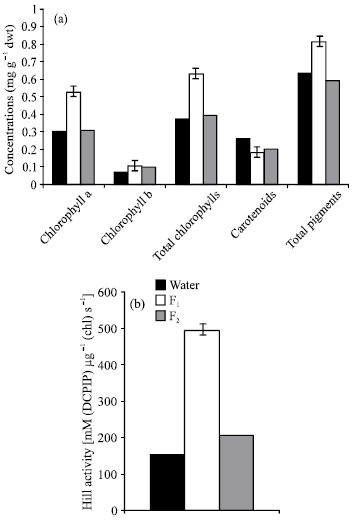
As compared to control plants, seed priming with F1 filetrate induced pronounced increase (P≤0.05) in total carbohydrates and polysaccharides of Lupinus plant, meanwhile these carbohydrate fractions appeared to be non-significantly affected in response to F2 treatment (Fig. 3a). The marked increase in carbohydrate fractions in Lupinus plants as a results of seed pretreatment with algal filtrates may presumably be due to the growth bioregulators present in these filtrates which may be involved directly or indirectly in saccharides and nitrogen metabolism.
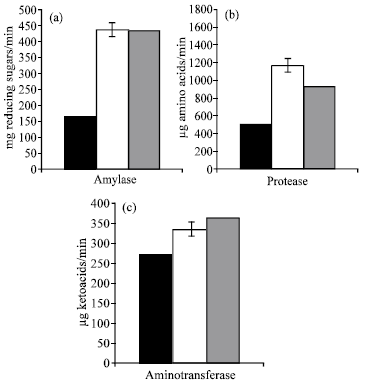 | |
| Fig. 5: | Effect of algal filtrates (F1 or F2) on enzymes activity of Lupinus termis shoot |
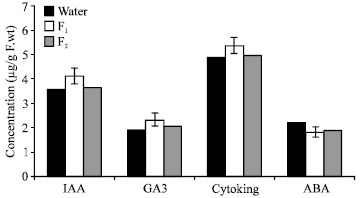 | |
| Fig. 6: | Effect of algal filtrates (F1 or F2) on growth regulators content of Lupinus termis shoot |
Furthermore, this increase may be a consequence of increased CO2 fixation which in turn led to a consequence increase in carbohydrate reserve for plant growth (Chaves et al., 1995). These results are in good agreement with the results obtained by Lozano et al. (1999). Furthermore, El-Shanshoury and Hamada (1988) stated that monosaccharides and disaccharides, endogenous IAA and mineral content of Zea mays were enhanced by treating with the extract of Streptomyces atroolivaceus under saline conditions.
Fig. 3b emphasized that ammonia-N, amide-N and total soluble-N increased markedly (P≤0.05) in response to F1 or F2, as compared with the control. On the other hand, total-N and protein content increased (P≤0.05) in response to F1 and non-significantly affected by F2 treatment.
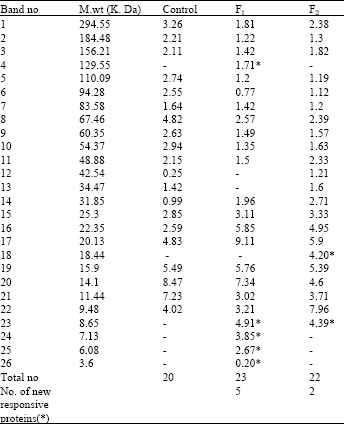
| Table 3: | Effect of the two algal filtrates (F1 or F2) on molecular weight and relative concentrations of different types of protein bands of Lupinus termis shoot |
 | |
The above-mentioned changes in insoluble-N and soluble-N may be due to either an increase or decrease in proteolytic enzymes which in fact degrade the storage protein to soluble nitrogenous compounds, which may in turn utilized by the various parts of the seedling (Fig. 5), in response to treatment with the used algal filtrates. Furthermore these increases could be attributed to the nitrogenase as well as nitrate reductase activities of
the algal extract associated with the surface of the seeds, or the amino acids and peptides produced in the algal filetrate and/or other compounds that stimulate growth of the plant (Adam, 1999). In relation to these results Martin et al. (2002) stated that nitrogen and carbohydrate status play the predominant role in regulating various aspects of seedling growth including storage reserve mobilization and photosynthetic gene expression.
As regards the changes in protein banding patterns in response to priming of Lupinus termis with the algal filtrates, Table 3 and Fig. 4 cleared that scanning of the gel indicated the occurrence of 20 protein bands; ranging from 3 to 294 KDa in the control shoot and the algal filtrates increased these bands to 23 and 22 in response to F1 and F2 respectively. Furthermore, these treatments increased the relative protein concentrations in Lupinus seedling and the effect was more elicited with 20-32 KDa and this may be due to gibberellin-induced processes during seed germination (Jones and Davies, 1991). Also this finding is in agreement with those obtained by Mohiuddin et al. (2000), who detected an increase in protein content of wheat treated with biofertilizers (Phosphert, Azofert, Biplin and Vitormone).
The appearance of new protein band with molecular wt 129 KDa in response to F1 and with 18 KDa in response to F2 could be considered as treatment-specific proteins (King, 1991) or indicate a changed pattern of gene expression on biofertilizer treatment (Popova et al., 1995). On the other hand, induction of the 3-8 KDa proteins, with F1 or F2 treatment could be also triggered by hormones which present in the filtrates and it is tentatively predicted that such proteins might represent phytohormone receptors (Davies, 1995 ). Moreover, the lower molecular weight proteins are known to have a role in the stress tolerance process (Waters et al., 1996).
Phytohormones have been implicated in the regulation of several enzymes and the metabolic changes occurring in the early stages of germination are the results of the activity of various enzymes (Kumar et al., 1987). In this context the results in figure 5 show that, seed priming with F1 or F2 caused a progressive (P≤0.05) increase in the determined enzymes (Proteolase, amylase and aminotransferase). In relation to control levels, the magnitude of the maintained increase was most significant in response to F1 (protease and amylase) and F2 (aminotransferase). These results could be related to the filetrate contents, which may play important role in enhancement of the biosynthesis of enzyme protein, enzyme activation and/or membrane permeability.
The observed significant increases in the determined enzyme in Lupinus shoot in response to pretreatment with the two algal filtrates are in good conformity with the increment in growth rate as well as nitrogen and protein content (Fig. 1, 3b and c). In accord with these results, Adam (1999) stated a significant increase in growth parameters and nitrogenous compounds in some crop plants treated with cyanobacteria as biofertilizer and he attributed this to the increase in nitrogenase as well as nitrate reductase activities of the algal filetrate. Moreover, Choudhury and Gupta (1998) found that treatment the seeds with GA3 increased germination and also α-amylase activity. Recently, Prasad et al. (2000) found that presoaking of maize and cowpea seed in biofertilizer (whey) for 18h increased the enzyme activity (amylase, proteinase and starch phosphorlase) of the used seeds as compared with control. In conclusion, there are a good correlation between the increase or decrease in protein, transamianse activity and soluble nitrogen content (Fig. 3c, 5 and 3b).
The two used algal filtrates (F1 or F2), as shown in figure 6, stimulate the endogenous level of IAA, GA3 and cytokinin and at the same time inhibit ABA level in Lupinus plant. The stimulation or inhibition of the endogenous plant hormones was more pronounced in response to F1 treatment. In this connection, there is a general agreement on the induction of the endogenous hormone levels by exogenous application of different growth regulators (El-Shahaby, 1992). These are in conformity with the findings of Haroun (1986) that the pretreatment of Pisium sativum seeds with either GA3 or IAA gave the saline-treated plants some tolerance through the inductive changes in the endogenous levels of growth regulators.
It is worthy to mention that the algal filtrates of Cylindrospermum muscicola (F1) or Anabaena oryza (F2) were most efficient, particularly F1, in improving growth of Lupinus termis grown in siliceous soil and this results was in good harmony with Hedge et al. (1999) who stated that the application of algal biofertilizer is useful for the reclamation of marginal soils and also with Zaccaro et al (2001) who documented that, the biofertilizers are likely to assume greater significance as complement and/or supplement to chemical fertilizers in improving the nutrient supplies to cereal crops because of high nutrient turn-over in the cereal production system, exorbitant cost of fertilizers and greater consciousness on environmental protection.
REFERENCES
- Adam, M.S., 1999. The promotive effect of the cyanobacterium Nostoc muscorum on the growth of some crop plants. Acta Microbiol. Polonica, 48: 163-171.
Direct Link - Aldesuquy, H.S. and A.M. Gaber, 1993. Effect of growth regulators on Vicia faba plants irrigated by sea water Leaf area, pigment content and photosynthetic activity. Biol. Plant., 35: 519-527.
CrossRefDirect Link - Arnon, D.I., 1949. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol., 24: 1-15.
CrossRefPubMedDirect Link - Bograh, A., Y. Gingras, H.A Tajmir-Riahi and R. Carpentier, 1997. The effects of spermine and spermidine on the structure of photosystem II proteins in relation to inhibition of electron transport. FEBS Lett., 402: 41-44.
CrossRefDirect Link - Chaves, M.M., J.S. Pereira, S. Cerasoli, J. Clifton-Brown, F. Miglietta, A. Raschi and P. Statt, 1995. Leaf metabolism during summer drought in Quercus ilex trees lifetime exposure to elevated CO2. J. Biogeography, 22: 255-259.
CrossRefDirect Link - Choudhury, S. and K. Gupta, 1998. Studies on the germination mechanism of Catharanthus roseus (L.) G. Do NCV. Alba seeds: Effect of promoters and pH. Seed Sci. Technol., 26: 719-732.
Direct Link - Drazkiewicz, M., 1994. Chlorophyllase: Occurrence, functions, mechanism of action, effects of external and internal factors. Photosynthetica, 30: 321-332.
Direct Link - Esashi, Y. and A.C. Leopold, 1969. Cotyledon expansion as a bioassay for cytokinins. Plant Physiol., 44: 618-620.
Direct Link - Frankland, B. and P.F. Wareing, 1960. Effect of gibberellic acid on hypocotyls growth of lettuce seedlings. Nature, 185: 255-256.
CrossRef - Ghallab, A.M. and S.A. Salem, 2001. Effect of biofertilizer treatments on growth, chemical composition and productivity of wheat plants grown under different levels of NPK fertilization. Ann. Agric. Sci., 46: 485-509.
Direct Link - Kumar, V., A.S. Basra and C.B. Malik, 1987. Enzymes of non-photosynthetic C4 dicarboxylic acid metabolism in germinating seeds of wheat. Biochemie und Physiologie der Pflanzen, 182: 261-265.
CrossRefDirect Link - Laemmli, U.K., 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227: 680-685.
CrossRefDirect Link - Lozano, S.M., J.V. Star, R.K. Maiti, A. Oranday, H. Gaona, E. Aranda and M. Rojas, 1999. [Effect of an algae extract and several plant growth regulators on the nutritional value of potato (Solanum tuberosum L. var. gigant)]. Arch. Latinoam. Nutr., 49: 166-170, (In Spanish).
PubMed - Ordog, V., 1999. Beneficial effects of microalgale and cyanobacteria in plant soil systems, with special regard to their auxin-and cytokinin-like activity. Proceedings of the International Workshop and Training Course on Microalgal Biology and Biotechnology, June 13-26, Mosonmagyarovar, Hungary, pp: 13-26.
- Popova, L.P., Z.G. Stoinova and L.T. Maslenkova, 1995. Involvement of abscisic acid in photosynthetic process in Hordeum vulgare L. during salinity stress. J. Plant Growth Regul., 14: 211-218.
Direct Link - Salmia, M.A., S.A. Nyman and J.J. Mikola, 1978. Characterization of proteinase present in germinating seeds of scots pine Pinus sylvestris. Physiol. Plant., 42: 252-256.
Direct Link - Shindy, W.W. and O.E. Smith, 1975. Identification of plant hormones from cotton ovules. Plant Physiol., 55: 550-554.
CrossRefDirect Link - Snedecor, G.W. and W.G. Cochran, 1980. Statistical Methods. 7th Edn., Iowa State University Press, Iowa, USA., ISBN-10: 0813815606, Pages: 507.
Direct Link - De Mule, M.C.Z., G.Z. de Caire, M.S. de Cano, R.M. Palma and K. Colombo, 1999. Effect of cyanobacterial inoculation and fertilizers on rice seedlings and postharvest soil structure. Commun. Soil Sci. Plant Anal., 30: 97-107.
CrossRefDirect Link - Zheleva, D., T. Tsonev, I. Sergiev and E. Karanov, 1994. Protective effect of exogenous polyamines against atrazine in pea plants. J. Plant Growth Regul., 13: 203-211.
CrossRefDirect Link - Rippka, R., J. Deruelles, J.B. Waterbury, M. Herdman and R.Y. Stanier, 1979. Generic assignments, strain histories and properties of pure cultures of cyanobacteria. Microbiology, 111: 1-61.
CrossRefDirect Link








