ABSTRACT
Stem transverse thin cell layers (tTCLs) from leading Japanese chrysanthemum (Dendranthema X grandiflora Ramat. Kitamura) ‘Shuhou-no-chikara’ cultivar were used to obtain defined morphogenic programs when select plant growth regulators (PGRs) were used, superior to the use of conventional explant sources. A single auxin or cytokinin, or numerous permutations of each, in conjunction with tTCLs, were utilized to manipulate the callogenic, caulogenic, rhizogenic and somatic embryogenic pathways/programs in vitro. Light or darkness played a significant role. Single PGRs were capable of activating morphogenic programs in chrysanthemum tTCLs: callus induction by TDZ, axillary shoots by BA, roots by 2,4-D or NAA and somatic embryos by IAA, while multiple PGR applications resulted in a multiple organogenic outcome.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2003.505.514
URL: https://scialert.net/abstract/?doi=ajps.2003.505.514
INTRODUCTION
A successful regeneration protocol of chrysanthemum (Dendranthema X grandiflora (Ramat.) Kitamura) is indispensable for the development of physiologically and morphologically normal control and transgenic plants. Dendranthema has been extensively tissue-cultured from various explant sources (leaves, stems, shoot tips, flower parts or pedicels and protoplasts), albeit often with a mixed organogenic response as a result of the cellular heterogeneity present in the initial explant source (Rout and Das, 1997; Tanaka et al., 2000). Various factors have been shown to influence the reaction of thin cell layers (TCLs) in Dendranthema in vitro culture, primarily polyamines (Teixeira da Silva, 2002a), carbon source and the use and choice of filter paper. TCLs allow for the directed control of morphogenic and developmental pathways of shoots, roots, flowers or somatic embryos derived from other specific or non-specific cells, tissues or organs when the milieu in which they are placed is also controlled (Tran Thanh Van, 1973). This capacity comes from the size and medium-dependent nature of the TCLs with minimal interference from internal factors and growth substances. TCLs, being small in size are excised either a) longitudinally (lTCL), being composed of a few tissue types or b) transversally (tTCL) consisting of several tissue types which are normally too small to separate and are explants that have been successfully utilized in the propagation of numerous ornamental species (Le and Nhut, 2000). Due to the sensitive nature of TCLs (due to their size and restricted cell number) to media manipulations, they provided a suitable means to detect the effect of PGRs on plant morphogenesis.
The regeneration of whole plants (primarily adventitious shoots without an intermittent callus phase) from tissue culture, a prerequisite for any transformation system, has been previously achieved in chrysanthemum from various explant sources (Rout and Das, 1997): leaves, stems, shoot tips, flower parts or pedicels and protoplasts, while few histologically confirmed reports on somatic embryogenesis in Dendranthema exist. It is believed that adventitious shoot regeneration derived from an initial callus phase may result in somaclonal variation and in chimerism while direct shoot regeneration from leaf or stem explants may eliminate such undesirables (Kaul et al., 1990), although regeneration and transformation capacities in various Dendranthema cultivars can be unrelated (de Jong et al., 1993; Teixeira da Silva and Fukai, 2002).
In most studies on chrysanthemum, the shoot regeneration capacity (SRC) is reported as the number of surviving explants and as the number of shoots formed per explant and in transformation experiments, this is normally on a selective (antibiotic-containing) medium. The principle objective of this study was to obtain defined organogenic programs (callus, adventitious shoots or roots, somatic embryos (SEs)) through the use of thin cell layer technology, in combination with plant growth regulators (PGRs) and light conditions. This study further demonstrates that the capacity to regulate chrysanthemum organogenesis in vitro has positive consequences for the success of genetic transformation protocols due to a refined regeneration protocol.
Materials and Methods
Plant material in vitro and greenhouse culture conditions: ‘Shuhou-no-chikara’ chrysanthemum (Dendranthema X grandiflora (Ramat.) Kitamura), the leading Japanese cultivar and a standard-type, shoots (at least 4 cm in height) derived from any in vitro medium (control and treatment plants) were harvested and placed in 1 L polycarbonate plant boxes (CB-3; Iuchi, Japan) on 100 ml of Hyponex® (soluble liquid fertilizer, HYPONeX, Co. Ltd., Japan; N:P2O5:K2O = 6.5:6:19; 3 g L-1) supplemented with 20 g L-1 sucrose and maintained under a 16 h photoperiod (40 μmol m-2 s-1) using plant growth fluorescent lamps (PGF; 45 μmol m-2 s-1 Homo-Lux, National Electric Co., Tokyo, Japan) at 25°C (Fig. 1). Control plants from which TCLs were derived were maintained on the same medium and under the same conditions. Chrysanthemum plantlets were acclimatized at 90% relative humidity and maintained in the greenhouse under long-day conditions before transfer to short-day (13-14 h darkness with a 4 h light break: 10 pm to 2 am) conditions for flower induction. Acclimatized plantlets were checked for morphological and flowering normality.
Explant preparation and treatment: Stem internode tissue of in vitro tTCLs (~200-500 μm thick and 1-2 mm in diameter; Fig. 1) containing all cell types were prepared from internode stems of 6-8 cm high in vitro plantlets (approximately 45 day-old plantlets). To test the effect of PGR on growth and morphogenesis, sterilized filter paper rings (Advantec #2), 9 cm in diameter, were placed a posteriori on 25 ml of different autoclaved MS-based (Murashige and Skoog, 1962) morphogenic program media containing a single PGR or a PGR combination utilizing the following auxins: 2,4-dichlorophenoxyacetic acid (2,4-D); indole-3-acetic acid (IAA); α-naphthyleneacetic acid (NAA)] and cytokinins [6-benzylaminopurine (BA), 6-furfurylaminopurine (Kinetin), thidiazuron (TDZ) in 9.5 cm wide petri-dishes (Sekisui, Japan). Control tTCLs were placed directly onto 25 ml of agar (i.e. without PGRs) of the same morphogenic media formulated to induce specific morphogenic programs.
All treatments were placed in the light (16 h photoperiod, 40 μmol m-2 s-1; PGF lamps) at 25°C.
Scoring of morphological response: All explants were scored for tTCL development and organogenesis i.e. the number of tTCLs forming shoots, roots, callus or somatic embryos (in any developmental state) after 60 days in culture, following treatment application. Greenhouse-acclimatized plants (n = 15) were checked and compared for vegetative and flowering characteristics (plantlet height, number of leaves, total fresh weight, and the number of disk and ray florets per flower head) using the experimental design as outlined below, as soon as flowers were fully open (approximately 60 days after acclimatization):
| ● | SRC of adventitious roots in the rooting program, |
| ● | the rooting capacity of adventitious shoots in the shoot program, |
| ● | the capacity of SEs derived from the SE program to establish plantlets on a PGR-free medium, |
| ● | the organogenic regeneration capacity of different callus types generated within the callus program, |
| ● | the somatic variation arising from organs from any of these four programs and |
| ● | greenhouse growth and flowering were tested. |
Histological analyses: tTCL explants from all treatments were observed under light microscopy and scanning electron microscopy (SEM) 30 days following treatment as described by Teixeira da Silva (2002a) to observe shoot formation as well as any histological changes arising from the treatments. SEM samples were fixed in FAA (formalin:acetic acid:70% ethanol=5:5:90), dehydrated in an ethanol series (50-100% EtOH for at least 6 h each (Fukuda et al., 1997), critical point-dried, sputter-coated with Pt and viewed under a SEM microscope Hitachi-2150 (Tokyo, Japan).
Flow cytometry: Nuclei were isolated from about 0.5 g of in vitro plant material (shoot and callus) derived from 60 days-old control and cefotaxime-treated plants (10 and 50 μg ml-1 kanamycin for chrysanthemum and tobacco, respectively, or 100 μg ml-1 cefotaxime for both) by chopping the sample in a few drops of Partec Buffer A [2 μg ml-1 4,6-diamidino-2-phenylindole (DAPI), 2 mM MgCl2, 10 mM Tris, 50 mM sodium citrate, 1% PVP K-30, 0.1% Triton-X, pH 7.5 (Mishiba and Mii, 2000). Nuclear fluorescence was measured using a Partec® Ploidy analyser (Partec GmbH, Germany) after filtering the nuclear suspension through 30 μm mesh size nylon filter (CellTrics®) and adding five times of solution A for 1 min. Three samples were measured for each treatment and relative fluorescence intensity of the nuclei analyzed when the coefficient of variation was <4%. A total of 2500 nuclei were counted for any sample.
Experimental design and statistical analyses: Experiments were organized according to a randomized complete block design (RCBD) with three blocks of 20 replicates per treatment. The effects of filter paper type and antibiotic on four programs (shoot, root, callus, somatic embryo) were separately analysed for each cultivar (tobacco, LIN, SNC), and data was subjected to analysis of variance (ANOVA) with mean separation (P=0.05) by Duncan’s new multiple range test (DMRT) using SAS® vers. 6.12 (SAS Institute, Cary, N.C.).
RESULTS AND DISCUSSION
It is possible to control different organogenic programe in Dendranthema in vitro culture in response to PGRs and to light or dark application when utilizing a TCL (Fig. 1). The utilization of a large stem (or leaf; data not shown) explant results in an organogenic moiety, as a result of the presence of excessive numbers of cells and heterogeneous tissue types and due to the medium-independent manner of nutrient/media additve utilization.
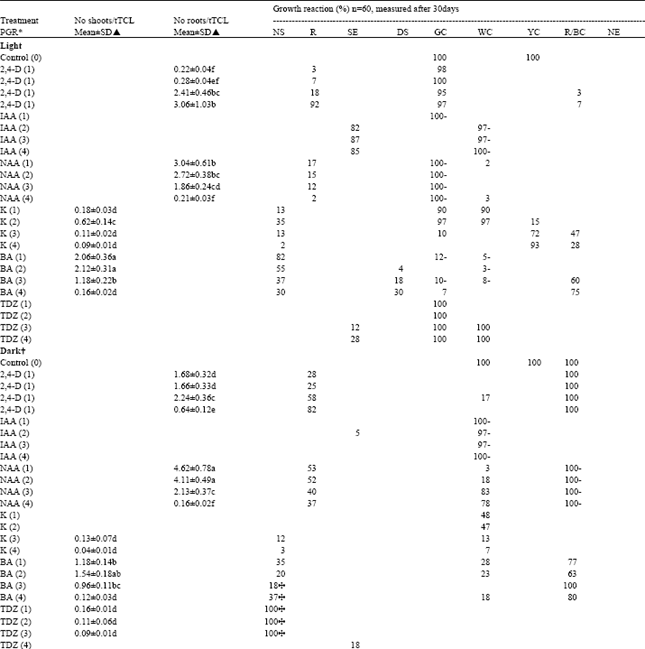
| Table 1: | Organogenic response (% of total explants) of D.grandiflora stem tTCLs to single PGRs and PGR combination in the light/dark |
 | |
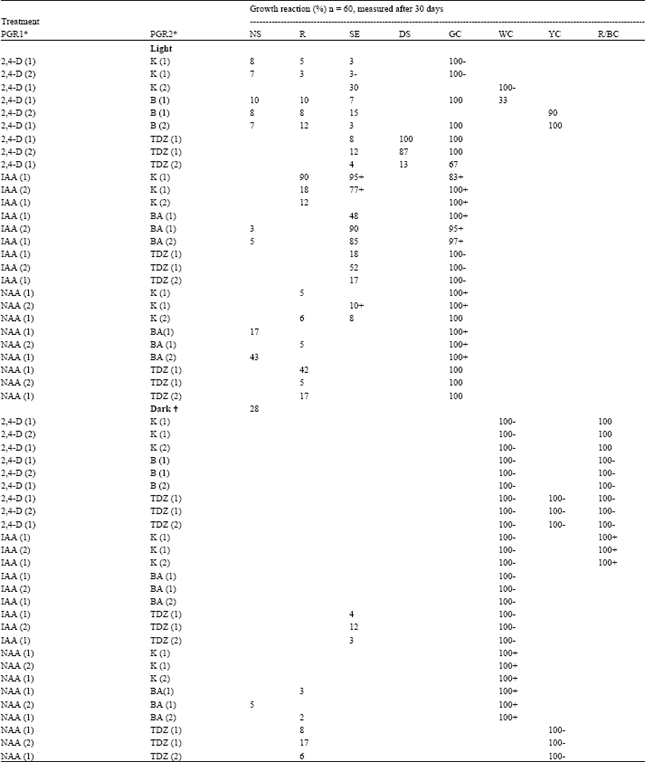 | |
| * Values () in mg l-1 of PGR; ▲ = Different letters within a column indicate significant differences at P=0.05 using Duncan’s New Multiple Range Test; NS = normal adventitious shoot; R = adventitious root; SE = somatic embryo; DS = deformed (fascinated, hyperhydric or other) shoot; GC = green callus; WC = white callus; YC = yellow callus; R/BC = red or brown callus; NE = necrosis; d = multiple shoot cluste; ^ = Shoot all etiolated and with long internodes; + or - indicate large or little quantities, respectively as compared to control | |
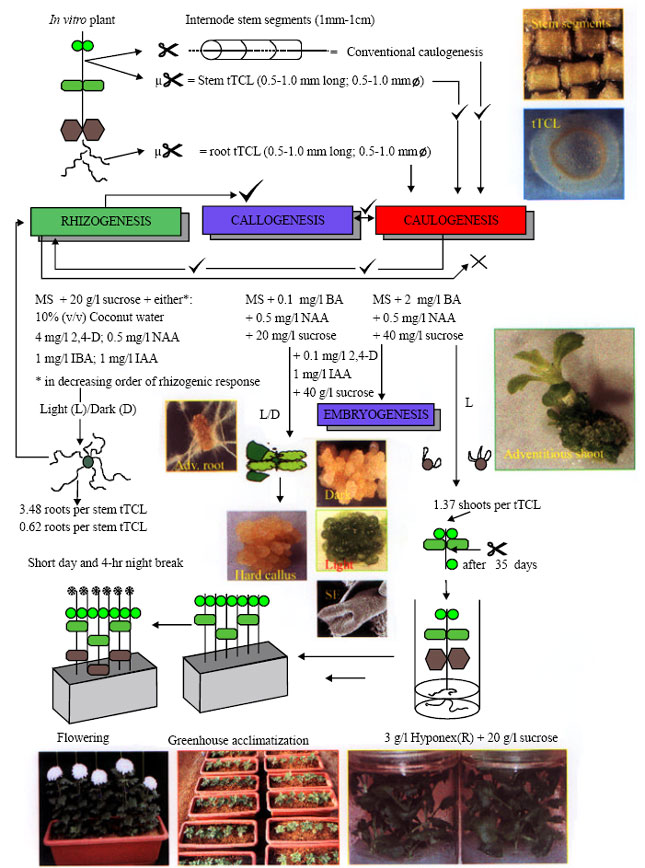 | |
| Fig. 1: | Chrysanthemum organogenesis is achieved by conventional explant (leaf/stem) preparation or by TCL technology |
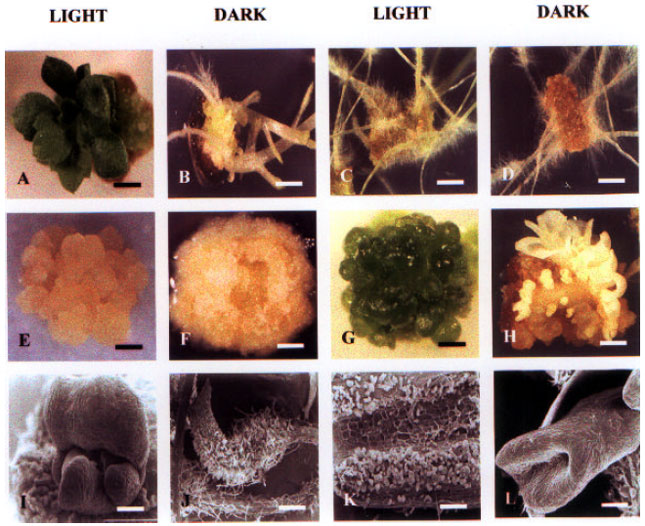 | |
| Fig. 2: | Chrysanthemum organogenic programs induced by PGR treatment of tTCLs in vitro (dark versus light). A) and B) adventitious shoots by 2 mg/l BA; C) and D) adventitious roots by 1 mg/l NAA; E) embryogenic callus and F) friable meristematic callus by 1 mg/l TDZ; G) and H) primarily globular somatic embryos with 2 mg/l IAA; I) - J) SEM of shoot primordium (I), root (J), friable callus (K) and somatic embryo (L). Scale bar: A,B,C,D,G,H = 1 CM; E,F = 0.5 CM, I,J = 200μM. |
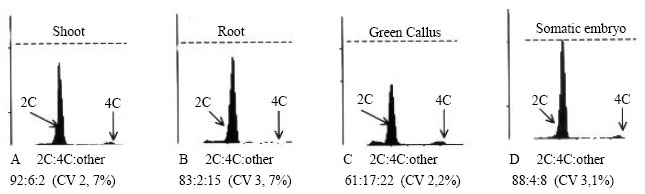 | |
| Fig. 3: | Flow cytometric analysis of different organogenic program products: shoots (A), roots (B), Yellow embryogenic callus (C) and somatic embryos (D); X-axis = staining intensity, Y-axis = number of events or cell count (dotted line = 500 nuclei) |
 | |
| Fig. 4: | Somaclonal variation in in vitro plantlets: (A) stem fasciation; B) hyperhydricity and C) multiple shoot clustering) and their acclimatization (D). Scale bar: A, C = 1cm; B = 200 μm; D, E = 10 cm |
This study demonstrates that it is possible to obtain primarily (>95%) callus (in the callus program) by TDZ (1 or 2 mg l-1) in the light, adventitious shoots (in a shoot program) by BA (2 mg l-1) in the light (Fig. 2A,I) or dark (Fig. 2B), adventitious roots (in a root program) by NAA (1-4 mg l-1 in the light (Fig. 2C,J), or by 1 mg l-1 in the dark, Fig. 2D) and Ses (globular, in an SE program; Fig. 2H) by the application of IAA (1 or 2 mg l-1) in the light or dark (Table 1). This may in part be due to an initial low number of cells within a tTCL explant, making it more medium-dependent. Green callus (hard or friable) generated in light treatments has the same consistency of yellow callus generated in dark treatments and is the same callus type since transferal of any dark treatment-derived yellow callus becomes green over 48 h under light conditions and with the same SRCs.
Adventitious shoots are the primary propagules in mass propagation and transformation studies of chrysanthemum. SRC was quantified in this study as being stimulated when the normal shoot (NS): deformed shoot (DS) ratio was high (indicating the favourable formation of normal shoots) and by high explant survival. A poor SRC was indicated by a low NS: DS ratio and by high necrotic and/or brown or red callus percentages, the latter two havinga zero SRC. While most chrysanthemum studies report on direct adventitious shoot formation (direct caulogenesis) without an intermediate callus phase, indirect regeneration via callus has also been reported from stem (Miyazaki et al., 1979), petal (Bush et al., 1976) and shoot tips (Huitema et al., 1989), but which may result in somaclonal variation. The only case so far reported of genotype independence of SRC in chrysanthemum was in ‘Polaris’, ‘Hekla’ and ‘Iridion’ (Sherman et al., 1998). In this study, an average of 2.70 adventitious shoots could be obtained from in vitro stem explants when on a non-selective medium, but was drastically reduced to 0.35 shoots per explant when on a 10 mg l-1 kanamycin selection medium in transformation experiments (Teixeira da Silva and Fukai, 2002), with a consequent decrease in the NS: DS ratio and the formation of yellow, embryogenic-like callus. Excessive wounding of plant material has a deleterious effect on explant survival although wounding has been shown to stimulate shoot formation (Hosokawa et al., 1998). A reduced number of shoots per explant in tTCLs as compared to conventional stem segments may be as a result of the excessive tissue damage caused in TCL preparation and due to their reduced cell numbers and area.
Similar organogenic responses may be obtained, but often with overlapping programmes as a result of the application of more than one PGR (Table 1). Under light the presence of 2,4-D together with either K, BA or TDZ results in a mix of all 4 types of organogenic programmes. The same treatments, however, under dark conditions, develop only white (Fig. 2F) and brown callus. The white callus reverts into as many organogenic programmes as those demonstrated under light conditions when transferred to the same PGR-free medium under light. Green and white friable callus can be placed on a rhizogenic, caulogenic or embryogenic programme medium to induce adventitious roots or shoots, or SEs, respectively.
Regeneration studies conducted on D. indicum and D. morifolium (Ledger et al., 1991) showed that whereas D. indicum produced at least 1-2 shoot loci when in vitro leaf pieces were placed on MS supplemented with 0.2 mg l-1 IAA plus either 3 or 5 mg l-1 BA, six D. morifolium genotypes regenerated poorly on the same medium. In another study (Kaul et al., 1990) however, eight out of eleven D. morifolium cultivars regenerated shoots on media containing 1 mg l-1 NAA and 1 mg l-1 BA. Extreme genotype dependent SRCs could be observed in D. grandiflora, ranging from 0 to 90%, when leaf or stem explants were placed on an MS with 2 mg l-1 BAP and 1 mg l-1 NAA medium. Similar reactions could be observed in D. grandiflora where a 27% SRC per explant in ‘Polaris’ versus 90-100% in ‘Iridon’ and ‘Hekla’ indicate genotype dependence of the regeneration protocol (Urban et al., 1994). Stem explants were shown to have a higher SRC than petioles or leaves, with a range of 2 to 10 shoots per explant, depending on the genotype (Yepes et al., 1995). SRC values (45 – 97% of explants capable of forming shoots) of depending on the time exposed to the regeneration medium (Sherman et al., 1998), when stem explants were cultured on a 2,4-D (0.5 mg l-1). In that study, the number of shoots per explant was high and cultivar-independent, with 8.2, 17.2 and 13.7 for ‘Polaris’, ‘Hekla’ and ‘Iridion’, respectively, when placed on a 3-step regeneration medium. Chrysanthemum morifolium was shown to form between 2.15 and 4.5 shoots per leaf or stem explant, when cultured on an IAA and BAP supplemented MS medium (Bhattacharya et al., 1990). Mechanical wounding – induced by brushing leaf explant surfaces – was shown to increase the SRC in D. grandiflora ‘Parliament’ (de Jong et al., 1993), while sonication in ‘Shuhou-no-chikara’ had a similar SRC-stimulating effect (data not shown). Studies on the effect of BA, Kinetin and NAA on ‘Orange Reagan’ chrysanthemums revealed a very low SRC (Oliveira et al., 1995), emphasizing the genotype-dependent nature of regeneration in this genus. Large genotypic differences could be observed between D. grandiflora cultivars ‘Shuhou-no-chikara’, ‘Shuhou-no-kokoro’ and ‘Lineker’ in response to the same so-called ideal regeneration medium and media additives, such as polyamines within it (Teixeira da Silva, 2002).
Embryoids were shown to form directly on the cut edges of garland chrysanthemum leaf explants on medium containing 0.1 mg l-1 BA and 1 mg l-1 NAA and originated from epidermal and sub-epidermal layers and not from callus (Oka et al., 1999). In ‘Shuhou-no-chikara’, the formation of globular somatic embryos occurred either randomly within callus derived from dark treatments (Fig. 2E) or uniformly on the surface of tTCLs when in the light (Fig. 2G). SEs are useful as in vitro propagules and due to their single-cell origin, when coupled with TCL technology and genetic engineering, may result in a transgenic somatic embryo programme. TCL organogenic programmes are important sources of cryopreservable (wild-type or transgenic) organs (research currently in progress). Callus derived from any treatment showed different SRCs, which could be conveniently classified according to colour: green, hard > green, friable > white, friable > yellow, friable > yellow, hard > red and/or brown. The use of callus for indirect shoot regeneration (although not desirable due to possible somaclonal variation) may be based on the colour coding.
Callus, adventitious shoots or roots and SEs do not show cytogenic or somaclonal variation (i.e. no endoreduplication or polyploidization) as observed by the consistent peaks in all material when subjected to flow cytometry. Only the relative 2C and 4C amounts differed between organogenic programme products as a result of the relative numbers of cells in active division (Fig. 3A-D), where the 4C value likely represents cells in the G2/M phase of the cell cycle. The higher the metabolic activity of cells, i.e. division, the lower the 2C:4C ratio, such that green callus > shoots > SEs > roots in terms of active cell division.
Transformation efficiencies (TEs) reported in the literature for chrysanthemum range from 0-35% (Rout and Das, 1997) and although without the scope of this report, the factors most affecting the improvement of TEs are: genotype-dependence of regeneration, gene introduction method (Teixeira da Silva and Fukai, 2002), timing of selection pressure and uncertainty concerning the origin of transgenic organ formation. Within our laboratory TCL regeneration studies were conducted in an attempt to improve the TE. A high level of transgenic shoots and TE can be achieved due to the small size of TCLs, despite a lower SRC, when Agrobacterium (ideal vector) infection of TCLs is followed by a high (30 mg l-1) selection (kanamycin) pressure.
Chrysanthemum is a source of various valuable metabolites (Schwinn et al., 1994) and the capacity to stimulate organ-specific regeneration in TCL programs, such as that already achieved in chrysanthemum roots (Teixeira da Silva, 2003) may allow for organ-targeted secondary metabolite production.
Shoots derived from any treatment had standardized rooting, physiological morphology and flowering (Fig. 4) when subcultured three times on Hyponex® in vitro to eliminate residual effects of PGRs. Occasionally stem fasciation (<1%), hyperhydricity (circa. 4%), multiple branching or shoot clustering (<3%) occurred (Fig. 4A-D). In conclusion successful organogenesis (callus, adventitious roots or shoots, globular SEs) could be induced with tTCLs in conjunction with select single PGRs, or mixed programmes could be induced with the use of more than one PGR. This capacity to control organogenesis and regeneration in Dendranthema will allow for the development and improvement of the biotechnology of the genus. The capacity to control organogenesis in chrysanthemum (and indeed any plant species) has far-reaching consequences. The programmed induction of roots may lead to improved secondary metabolite production, shoots may be utilized as vegetative propagules, callus as cryopreservable regeneration material or somatic embryos as potential artificial seeds or mass micropropagation units, thus creating enormous potential for the floricultural and pharmaceutical sectors. Since all exogenous factors (light, temperature, humidity and other environmental cues) may be strictly controlled in vitro and since the endogenous effect of TCLs is limited (medium-dependent), TCL technology, together with the organogenic-stimulating properties of PGRs may work hand-in-hand in improving protocols aimed at chrysanthemum flower amelioration through biotechnology and genetic transformation.
REFERENCES
- Bhattacharya, P., S. Dey, N. Das and B.C. Bhattacharya, 1990. Rapid mass propagation of Chrysanthemum morifolium by callus derived from stem and leaf explants. Plant Cell Rep., 9: 439-442.
Direct Link - Bush, S.R., E.D. Earle and R.W. Langhans, 1976. Plantlets from petal segments, petal epidermis and shoot tips of the periclinal chimer, Chrysanthemum morifolium Indianpolis. Am. J. Bot., 63: 729-737.
Direct Link - Jong, J., W. Rademaker and M.F. Wordragen, 1993. Restoring adventitious shoot formation on chrysanthemum leaf explants following cocultivation with Agrobacterium tumefaciens. Plant Cell Tissue Organ Cult., 32: 263-270.
Direct Link - Hosokawa, M., M.M. Hossain, T. Takemoto and S. Yazawa, 1998. Particle-gun wounding of explants with and without plant-growth regulators effectively induces shoot formation in African violet. Plant Tissue Cult. Biotechnol., 4: 35-41.
Direct Link - Huitema, J.B.M., W. Preil, G.C. Gussenhoven and M. Schneidereit, 1989. Methods for the selection of low-temperature tolerant mutants of Chrysanthemum morifolium by using irradiated cell suspensions. I. Selection of regenerants in vivo under suboptimal temperature conditions. Plant Breed., 102: 140-147.
- Kaul, V., R. Miller, J.F. Hutchinson and D. Richards, 1990. Shoot regeneration from stem and leaf explants of Dendranthema grandiflora Tzvelev. (syn. Chrysanthemum morifolium Ramat.). Plant Cell Tissue Organ Cult., 21: 21-30.
Direct Link - Ledger, S.E., S.C. Deroles and N.K. Given, 1991. Regeneration and Agrobacterium-mediated transformation of chrysanthemum. Plant Cell Rep., 10: 195-199.
CrossRefDirect Link - Mishiba, K. and K. Mii, 2000. Polysomaty analysis in diploid and tetraploid Portulaca grandiflora. Plant Sci., 156: 213-219.
Direct Link - Miyazaki, S., E. Kishida, Y. Tashiro and K. Kanazawa, 1979. Tissue culture of Chrysanthemum morifolium Ramat. V. Histological studies on the callus and shoot formation in stem segments cultured in vitro. Agric. Bull. Saga Univ., 46: 43-65.
Direct Link - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link - Oka, S., O. Muraoka, T. Abe and S. Nakajima, 1999. Adventitious bud and embryoid formation in garland chrysanthemum leaf culture. J. Jap. Soc. Hortic. Sci., 68: 70-72.
Direct Link - Rout, G.R. and P. Das, 1997. Recent trends in the biotechnology of Chrysanthemum: A critical review. Sci. Hortic., 69: 239-257.
CrossRefDirect Link - Sherman, J.M., J.W. Moyer and M.E. Daub, 1998. A regeneration and Agrobacterium-mediated transformation system for genetically diverse chrysanthemum cultivars. J. Am. Soc. Hortic. Sci., 123: 189-194.
Direct Link - Tanaka, K., Y. Kanno, S. Kudo and M. Suzuki, 2000. Somatic embryogenesis and plant regeneration in chrysanthemum (Dendranthema grandiflorum (Ramat.) Kitamura). Plant Cell Rep., 19: 946-953.
Direct Link - Teixeira-da-Silva, J.A., 2002. Polyamines in the regulation of chrysanthemum and tobacco in vitro morphogenic pathways. Prop. Ornamental Plants, 2: 9-15.
Direct Link - Teixeira-da-Silva, J.A., 2003. Thin cell layer technology for induced response and control of rhizogenesis in chrysanthemum. Plant Growth Regulat., 39: 67-76.
Direct Link - Van, M.T.T., 1973. In vitro control of de novo flower, bud, root and callus differentiation from excised epidermal tissues. Nature, 246: 44-45.
Direct Link - Yepes, L.C., V. Mittak, S.Z. Pang, C. Gonsalves, J.L. Slightom and D. Gonsalves, 1995. Biolistic transformation of chrysanthemum with the nucleocapsid gene of tomato spotted wilt virus. Plant Cell Rep., 14: 694-698.
Direct Link








