ABSTRACT
In order to identify sources of resistance in cowpea against blackeye cowpea mosaic virus (BLCMV), two hundred cowpea germplasm accessions were evaluated under greenhouse conditions. Ten to 15 plants of each accession were artificially inoculated by sap prepared from virus-infected plants. Disease severity on each line was recorded at 15 days interval. The symptomless plants were tested by indirect enzyme-linked immunosorbent assay (ELISA) to separate the resistant plants from susceptible ones. Only two accessions (27168 and 27192) out of 134 of local origin were found resistant to BlCMV, whereas 23 (34.8%) were resistant out of 66 exotic lines. Twenty genotypes expressed segregation pattern towards disease reaction and the others were susceptible. In this study some new sources of resistance have identified which can be used to breed to breed virus-resistant cowpea cultivars.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2002.585.587
URL: https://scialert.net/abstract/?doi=ajps.2002.585.587
INTRODUCTION
Cowpea (Vigna unguiculata L. Walp.) also known as southern pea or blackeye pea, is an important food and fodder legume and is indigenous to Africa from where it was introduced in other tropical and sub-tropical countries (Cobley and Stelle, 1975). It is now grown in many regions of Africa, India, Brazil, USA, the West Indies, Australia, Pakistan, parts of Europe and South America (Rachie and Roberts, 1974). It provides more than half the amount of plant protein in human diets in the sub-humid tropics of Africa (Rachie, 1985).
Virus diseases are considered to be a major limiting factor for the production of cowpea in the tropical and sub-tropical countries (Mali and Thottappilly, 1986). More than 20 viruses are reported from various cowpea-growing areas worldwide (Thottappilly and Rossell, 1985), however, six viruses naturally infect cowpea in Pakistan (Ahmad, 1978; Bashir and Hampton, 1993) and five are seed-transmitted (Bashir et al., 2000). The most important and worldwide spread seed-borne viruses of cowpea are blackeye cowpea mosaic (BlCMV) and cowpea aphid-borne mosaic (CABMV) potyviruses (Bashir and Hampton, 1996). Losses caused by BlCMV depends upon virus strain and genotype interaction (Kuhn, 1989). Although, the information on yield losses in cowpea to BlCMV in Pakistan are not available, but Harrison and Gudauskas (1968) reported no seed yield in BlCMV-infected plants. A yield loss of 32 to 85% was reported when cowpea was infected with mixed infection of cucumber mosaic virus (CMV) and BlCMV (Kuhn, 1989). The BlCMV when occurs in mixed infection with CMV, it causes very severe infection due to synergistic effect (Pio-Ribeiro et al., 1978).
The identification and use of resistant sources against pests and diseases is an important component of genetic improvement programme. Although appropriate control of BlCMV can be achieved through several approaches, but the use of host plant resistance is the most economical and practicable method.
In the study, 200 cowpea germpalsm accessions obtained from local and foreign sources were evaluated against a seed-borne isolate of BlCMV with the objective to identify resistant genotypes to be used in breeding programme to develop virus resistant cowpea varieties.
Materials and Methods
Source of virus isolate: Blackeye cowpea mosaic virus (BlCMV) was isolated from infected seeds of small seeded local cowpea variety collected from Narowal (Punjab) market during May 2000. During June 2000, the seeds were grown in plastic pots filled with sterilized soil under greenhouse conditions at National Agricultural Research Centre (NARC), Islamabad, Pakistan. The cowpea seedlings showing characteristic virus-like symptoms were used for mechanical transmission on healthy susceptible cowpea plants to maintain the virus isolate. The identity of the virus isolate was confirmed by direct enzyme-linked immunosorbent assay (DAS-ELISA) as described by Clark and Adamas (1977) and by leaf dip electron microscopy and host reaction. The stock culture of the isolate was maintained by frequent mechanical inoculation on healthy susceptible cowpea plants in an insect-free greenhouse. The virus- infected plants served as the inoculation source for screening of cowpea germplasm. Precautions were taken to avoid isolate mixing with any other virus or viruses. The isolate has also been preserved in cowpea seeds for future use.
Source of cowpea germplasm: One hundred and thirty four cowpea germplasm accessions were obtained from Plant Genetic Resources Institute (PGRI), National Agricultural Research Centre (NARC), Islamabad. These accessions were collected from various parts of Pakistan and maintained in the gene bank at PGRI. Sixty six lines were received from International Institute for Tropical Agriculture (IITA), Ibadan, Nigeria during 1999.
Screening procedure: During first week of July 2000, ten to fifteen seeds (depending upon seed availability) of each accession were planted in plastic pots (12 inches diameter) filled with sterilized soil, sand and farmyard manure in the ratio of 20:1:1. Two pots with 10 to 15 plants were used for each accession. One week after planting when the primary leaves of the growing seedlings were fully expanded, they were inoculated mechanically by standard procedure as described by Bashir et al. (1995). Three weeks after inoculation the symptomless plants were re-inoculated to avoid any escape. The inoculated plants were kept under observations in an insect-free greenhouse at 28 to 30 oC.
Disease reactions were recorded at two weeks interval on 1-5 scale (- : no symptoms at all, + : mild symptoms, ++ : moderate symptoms, +++ : severe symptoms). This experiment was conducted in two parts (100 accessions were tested in each part) at one week interval.
Serological test: Three months after first inoculation in each experiment, only the symptomless plants were numbered and tested by direct antigen coating enzyme-linked immunosorbent assay (DAC-ELISA) as described by Hobbs et al. (1987). Composite leaves sample from symptomless plants (three leaves from each plant: lower, middle and upper) were collected and tested by DAC-ELISA. Absorbance at A 405nm was measured by a BIORAD multiscane ELISA reader machine 15 minutes after adding substrate (p-nitrophenyl phosphate) solution. The antiserum to BlCMV was provided by Dr. Jacqueline d’A. Hughes (Plant Virologist), IITA, Ibadan Nigeria.
RESULTS AND DISCUSSION
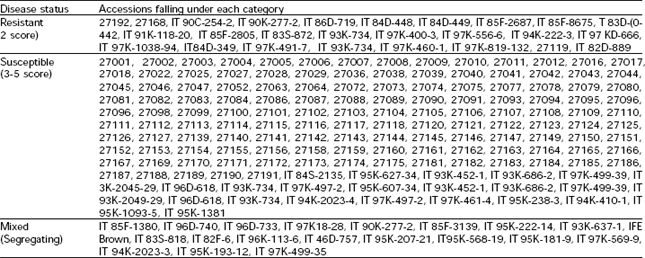
Based on disease reaction of each line, the 200 lines are grouped into three main categories such as resistant (25 accessions), susceptible (155 accessions) and segregating (20 accessions) (Table 1). The frequency distribution of each category has been showing in Fig. 1.
| Table 1: | Grouping of cowpea germplasm accessions tested against BlCMV under greenhouse conditions |
 | |
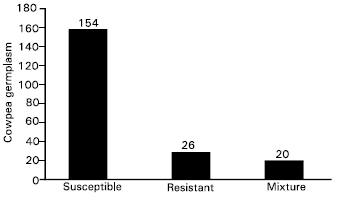 | |
| Fig. 1: | Frequency of two hundred cowpea germplasm accessions in each group based on 1-5 scale |
The reactions of inoculated leaves included the development of necrotic or chlorotic lesions. In majority of the cases, the development of local lesions was followed by systemic symptoms; systemic infection did not involve necrotic flecking. The chlorotic or local lesions were not observed in local material, whereas such type of symptoms were common in cowpea lines from IITA. The other reactions observed were mosaic, mottling, interveinal chlorosis, vein banding, leaf deformation and mild yellowing. Two types of resistance (immunity and tolerance) were detected, immunity being more common in the material from IITA. In a few lines delayed symptoms (ds) were expressed. These genotypes remained symptomless for three to four weeks, then developed mild to severe systemic symptoms. The lines with mild symptoms and low level of virus titer are considered as tolerant, whereas the lines with no visible symptoms and no detectable virus by ELISA are immune or highly resistant (Bashir, 2000).
Only two accessions (27168 and 27192) out of 134 were found resistant from local material (Table 1), whereas all others were highly susceptible indicating a very narrow genetic base of these accessions. Out of sixty six lines from IITA, Nigeria, 23 (34.8%) were resistant, 23 (34.8%) susceptible and 20 (30.3%) lines expressed a segregating pattern towards virus infection (Table 1). Resistant as well as susceptible plants were found in the lines expressing segregating pattern. In such lines resistant (no visible symptoms and no ELISA detectable virus) as well as plants with latent virus infection were found within the same genotype. This may be either due to mixing of seeds of the susceptible genotypes or specific genotypic behavior towards virus infection. Segregation pattern of cowpea germplasm from IITA towards virus infection within the same genotype has been reported previously (Ladipo and Allen, 1979). Segregating pattern in any line of local origin was not observed. Hypersensitive reaction of this isolate was not observed in any line which was reported in case of BlCMV isolates of diverse origin obtained from infected cowpea seeds (Bashir and Hampton, 1996).
A total of twenty five accessions (2 of local origin and 23 exotic) were found resistant (no degree of detectable virus propagation in inoculated plants by ELISA) from this material. Genetic resistance is widely recognized as the best means of controlling plant viral diseases, particularly cowpea viral diseases (Hampton et al., 1996). In a previous study, one local cowpea line (PAK-45443) was reported as resistant to BlCMV isolate derived from a line of Indian origin (Bashir et al., 1995). Although there are several reports on the identifications of resistance sources to BlCMV (Dijkstra et al., 1987; Mali et al., 1988; Bashir et al., 1995; Bashir and Hampton, 1996), but in this study report some additional sources of resistance from local and exotic material against a seed-borne isolate of BlCMV. Relatively higher number of resistant lines from exotic material indicates that IITA cowpea germplasm collection has a wider genetic base than local material. Resistance to BlCMV isolates of diverse origin from IITA material has been reported previously (Bashir and Hampton, 1996). These sources of resistance can be used in breeding programme to develop BlCMV-resistant cultivars with desirable agronomic attributes.
The BlCMV is known to be seed-transmitted from 0 to 22.6% (Mali et al., 1988; Bashir and Hampton, 1994) in cowpea. Planting of virus infected seeds may facilitate the introduction of pathogen in new localities and provide primary source of virus infection under field conditions. The identified cowpea lines resistant to BlCMV are potential source for establishment and production of certified seeds to avoid the introduction of BlCMV in new areas. There is also a potential for exploitation of resistance to seed transmission in cowpea germplasm and the selection for absence of seed transmission may prove to be useful approach for virus control in cowpea crop.
REFERENCES
- Bashir, M. and R.O Hampton, 1994. Seed and aphid transmission of some isolates of blackeye cowpea and cowpea aphid-borne mosaic potyviruses. Pak. J. Phytopathol., 2: 140-146.
Direct Link - Bashir, M. and R.O. Hampton, 1996. Detection and identification of seed-borne viruses from cowpea (Vigna unguiculata L.) Walp germplasm. Plant Pathol., 45: 54-58.
CrossRefDirect Link - Bashir, M. and R.O. Hampton, 1996. Identification of cowpea (Vigna unguiculata) cultivars and lines immune to variants of blackeye cowpea mosaic potyvirus. Plant Pathol., 45: 984-989.
CrossRef - Clark, M.F. and A.N. Adams, 1977. Characteristics of the microplate method of enzyme-linked immunosorbent assay for the detection of plant viruses. J. Gen. Virol., 34: 475-483.
CrossRefPubMedDirect Link - Dijkstra, J., L. Bos, H.J. Bouwmeester, T. Hadiastono and H. Lohuis, 1987. Identification of blackeye cowpea mosaic virus from germplasm of yard-long bean and from soybean and the relationship between blackeye cowpea and cowpea aphid-borne mosaic viruses. Neth. J. Plant Pathol., 93: 115-133.
CrossRef - Hobbs, H.A., D.V.R. Reddy, R. Rajeshwari and A.S. Reddy, 1987. Use of direct antigen coating and protein A coating ELISA procedures for detection of three peanut viruses. Plant Dis., 71: 747-749.
CrossRefDirect Link








