Research Article
Effect of Seed Dressing Fungicides for the Control of Seedborne Mycoflora of Wheat
Not Available
M. Aslam Rajput
Not Available
G. Sarwar Shah
Not Available
A. Mubeen Lodhi
Not Available
Farrakh Mehboob
Not Available
Seeds play vital role in the transmission of plant pathogens causing plant diseases. The pathogen may be externally or internally seedborne or associated with seed as contaminant. The seedborne pathogens may cause seed abortion, seed rot, seed necrosis, or reduction in germination as well as seedling damage by systemic or local infection resulting in the development of disease at later stages of plant growth. Many important diseases of plants caused by fungi spread through seeds (Neergaard, 1977). Healthy seed plays an important role for increasing successful cultivation and yield of crops. Seedborne pathogens of wheat are responsible to cause variation in plant morphology and also reducing yield 15-90 % if infected seeds are planted in the field (Wiese, 1984). Several Seedborne pathogens are known to associated with wheat seed and responsible for deteriorating seed quality during storage. Seed treatment is a biological, chemical, mechanical, or physical process designated to mitigate externally or internally seed or soilborne microorganisms, resulting in the emergence of a healthy seedling and subsequently a healthy plant. Seeds may be treated to promote good seedling establishment, to minimize yield loss or to maintain and improve quality and to avoid further spread of pathogens.
The application of chemicals to seed is safest, cheapest and could be effective means of controlling most seedborne pathogens. Fungicidal seed treatment may kill or inhibit seedborne pathogens and may form a protective zone around seeds that can reduce seed decay and seedling blight caused by soilborne pathogens, resulting and vigorous seedlings. The use of fungicides as seed treatment is the most widely followed disease control practice used in all crops (Nene and Thapliyal, 1979 and Sharvelle, 1979). In recent past the efficacy of various fungicides to control seedborne fungal mycoflora has been reported (Misra and Singh, 1972; Agarwal, 1981; Raut et al., 1983; Singh et al., 1984). Randhawa et al. (1985) treated wheat-stored grains with Captan, Thiram and Vitavax and found that Thiram was highly effective in protecting seed viability. El-Tayed et al. (1987) found Dithane M-45 and Benlate to be more effective as seed dressing fungicides against Alternaria alternata and Fusarium roseum. Izhar et al. (1987) obtained beneficial effects with Vitavax on grain yield and plant characteristics of wheat. Gupta et al. (1990) used four seed dressing fungicides against mycoflora associated with wheat seed. Captan and Dithane M-45 reduced the incidence of Helminthosporium sativum and Alternaria alternata and increased germination rates 88 and 87% respectively than Benlate.
The application of a general seed Protestant to seed helps in producing better emergence and vigorous seedlings. Seed-Protestant chemicals differ from crop to crop and from region to region. An experiment was therefore carried out to test the available seed dressing fungicides against fungi associated with seeds of commercial wheat varieties.
Materials and Methods
Isolation: Samples of twelve commercial wheat varieties viz., Mehran-89, T.J-83, Soghat, Sarsabz, Anmol, Johar, C-591, Sindh-81, Pak-70, Mexi-Pak-65, H-68 and Faisalabad-85 were collected from godown of Wheat Section, Agriculture Research Institute, Tandojam, Sindh, Pakistan. Five samples of 200 seeds each were taken randomly from each variety. Isolations were made from 200 infected seeds of each variety under aseptic conditions by standard method. After 6-7 days the fungi associated with seeds were identified by preparing their mounts and the percentage of the various fungi was counted.
Effect of seed dressing fungicides on germination of wheat seed In pots: 100 seed taken from each variety were treated with seven fungicides i.e. Baytan, Benlate, Vitavax, Captan, Derosal, Dithane M-45 and Rizolex at 2 gm per kg seed. Five treated seeds were planted in the sterilized pots of 22-cm diameter containing sterilized soil. They were irrigated with sterilized water. After 6 weeks percentage of germinated seedlings was recorded. The root length and shoot length of seedlings was also recorded. The experiment was conducted in randomized complete block design with three replications. The untreated seeds were kept as control (Randhawa et al., 1985).
Field experiment: The experiment was conducted as Randomized Complete Block Design, subplots measuring 4 x 1.22 m2 having 7 rows with 20 cm apart at wheat section, ARI, Tandojam. Seven fungicides, Baytan, Benlate, Vitavax, Captan, Derosal, Dithane M-45 and Rizolex were treated as seed dressing on most susceptible commercial wheat variety (Pak-70) at 2 gm seed. Untreated seeds used as control. The crop was harvested at maturity on 15th April. The parameters studied were seedling emergence, plant height (cm) near soil line, number of grain per spike, 1000-grain weight, grain yield per plot and per hectare.
Five fungal species were isolated , the most common were Alternaria tenuis, Aspergillus niger, Stemphylium herbarum, Fusarium moniliforme and Curvularia lunata from the seeds of 12 wheat varieties (Table 1). Fungi were identified on the basis of their typical colony characteristics and conidial morphology.
| Table 1: | Frequency of grain storage fungi associated with 12 wheat varieties |
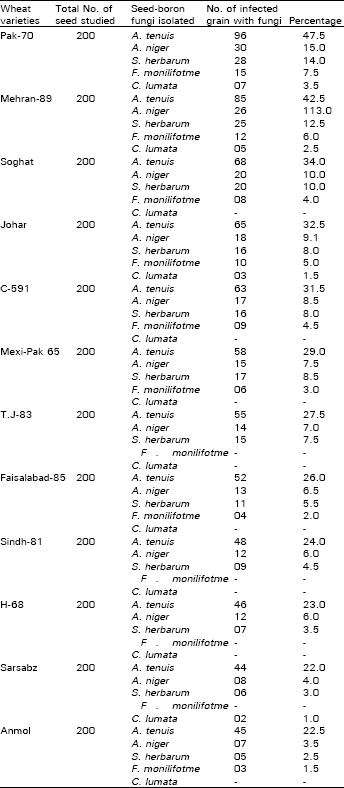 | |
Kamal and Mughal (1968), Khan et al. (1974) observed the presence of Alternaria, Helminthosporium, Fusarium, Curvularia, Stemphylium, Rhizopus, Cladosporium, Aspergillus and Penicillium species in wheat seeds. Grzelak and Sztrmer (1982) also found Alternaria tenuis, Botrytis cinerea and Fusarium spp. as predominant fungi from triticale seeds. The similar fungi were isolated and reported by Singh (1983), Martin et al. (1984) and Sejiny et al. (1984). Ghosh and Nandi (1986) observed that several species of Aspergillus and Penicilium jenseni are responsible for deteriorating wheat grains during storage.
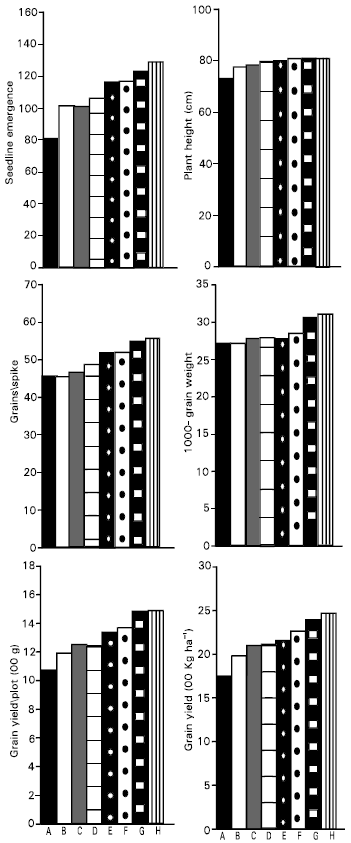 | |
| Fig. 1: | Effect of different fungicides on yield parameters of wheat variety (Pak-70) |
Kunwar (1989) also isolated Aspergillus spp., Penicillium spp., followed by Alternaria alternata from 50% samples of the stored wheat seeds.
| Table 2: | Effect of seed dressing fungicide on seed germination of wheat varieties in pots |
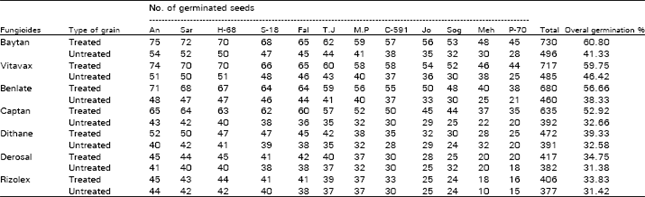 | |
| An=Anmol, Sar=Sarsabz, S-81=Sindh-18, Fsl=Faisalabad=85, M.P=Mexi-Pak, Jo=Johar, Sog=Soghat | |
| Meh=Mehran-89, T.J=T.J-83 and P-70=Pak-70 | |
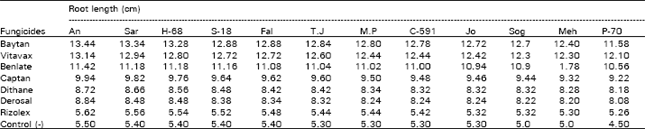
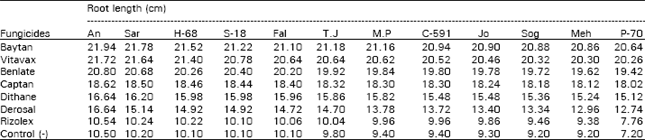
| Table 3: | Effect of seed dressing fungicide on root of seedlings of wheat varieties |
 | |
| Table 4: | Effect of seed dressing fungicides on shoot length of seedling of wheat varieties |
 | |
The maximum germination of wheat seedlings of all 12 varieties was recorded with Baytan followed by Vitavax, Benlate, Captan and Dithane M-45 (Table 2). The percent germination for seeds of all varieties treated with Baytan ranged from 45-75%, Vitavax 44-74%, Benlate 38-71%, Captan 35-65% and Dithane M-45 25-52%, respectively. There was no significant difference in the germination of seeds treated with Derosal and Rizolex. The overall germination percentage for seeds of all varieties treated with seven fungicides ranged from 3.83-60.80%, whereas the germination for untreated seeds ranged between 31.42 to 41.33% (Table 2). Kausar (1955) and Parashar (1970) used copper fungicides to reduce the incidence of black point of wheat. Misra and Singh (1971, 1972) tested Captan and Dithane M-45 against Alternaria tenuis and Helminthosporium oryzae. Khetarpal and Agarwal (1979) obtained the similar results with Carboxin (vitavax) and Benomyl (Benlate).
The greatest root length was seed of all varieties treated with Baytan and Vitavax followed by Benlate (Table 3). The root length was significantly decreased in seeds treated with Rizolex and Derosal, but it was higher than the untreated seeds of all the varieties (control). Maximum shoot length was obtained in the seeds of all varieties treated with Baytan (21.94), followed by Vitavax (21.72) and Benlate (20.80). Whereas shoot length was significantly decreased in seedlings grown from seeds treated with Rizolex (7.76). Shoot length of seedlings obtained from Captan, Dithane M-45, Derosal and Rizolex was also greatest than the seedlings germinated from untreated seeds (Table 4). Tragner et al. (1980) obtained 100% control with Baytan by improving seedling vigor, quality and quantity of grains. The similar results have been reported by Agarwal et al. (1981), Raut et al. (1983) and Singh et al. (1984).
All the fungicides used at 2gm per kg seed significantly increased the seedling emergence as compared to untreated seeds (control). The maximum number of seedlings emergence was recorded with Baytan followed by Vitavax, Benlate, Captan and Dithane M-45 (Fig. 1). There was no significant difference in the seedling emergence with Derosal and Rizolex. Significance increase in seedling emergence has been reported when seeds were tested with Captan and Vitavax (Randhawa et al., 1985) and by Bavistin, Benlate and Dithane M-45 (El-Tayed et al., 1987).
The maximum plant height was recorded with Baytan followed by Vitavax and Benlate (Fig. 1). Plant height was not significantly different obtained with Captan, Dithane M-45, Derosal and Rizolex respectively. All the fungicides increased plant height at 2 gm/1 kg seed as compared to control (untreated seeds).The maximum number of grains per spike was obtained by Baytan followed by Vitavax, Benlate, Captan and Dithane M-45 (Fig. 1). The remaining treatments Derosal and Rizolex did not show significant difference among their means when compared to control. Maximum 1000-grain weight was obtained with Baytan and Vitavax followed by Benlate (Fig. 1). The minimum grain was obtained with Rizolex. The 1000-grain weight obtained with Derosal and Rizolex was not significantly different with that of untreated seeds. Gupta et al. (1999) obtained similar results by using Captan, Dithane M-45 and Benlate against Alternaria alternata and Helminthosporium sativum associated with wheat. The maximum grain yield per plot and per hectare was recorded with Baytan and Vitavax followed by Benlate (Fig. 1). The lowest grain yield was obtained with Rizolex followed by Derosal. The grain yield was not significantly different among the treatments Dithane M-45, Derosal and Rizolex. All the fungicides increased the grain yield as compared to untreated seeds. Mahmuda et al. (1987) reported the similar results by using Vitavax and other fungicides against black point disease of wheat caused by Helminthosporium sativum.
From these finding it is infers that the seed health technique is primary need to avoid crop failure. Seed testing determine not only seed germination but it provides information of pathogen associated with seeds.