Research Article
A Harvest-induced ACC Oxidase Gene from Tips of Harvested Asparagus Spears and its Expression During Storage
Not Available
Toshiyuki Matsui
Not Available
Haruo Suzuki
Not Available
Yusuke Kosugi
Not Available
The plant hormone ethylene is produced in response to various kinds of environmental stress, such as wounding, physical load, disease, exposure to low temperature and chemicals and water stress (Hyodo, 1991). Harvest stresses are particularly severe on organs that are actively growing at harvest (Huber, 1987). Ethylene is synthesized from methionine via S-adenosylmethionine (SAM) and ACC in higher plants (Adams and Yang, 1979; Abeles et al., 1992). ACC synthase and ACC oxidase play essential roles in this pathway. ACC synthase catalyzes the conversion of SAM to ACC, whereas ACC oxidase catalyzes the oxidation of ACC to ethylene.
Ethylene production by asparagus is modest (2.1 - 4.9 μl-1 kg-1 hr-1) and increases with time after harvest (Haard et al., 1974). Harvesting and handling impose a series of stresses on the tissue, including wounding, dehydration, and separation from nutrient supply. The rate of ethylene production by harvested spears increased about 50% between 45 and 165 minute after harvest (Beever et al., 1985). As the asparagus spears are not a large ethylene producing commodity very few research have been conducted on the rates of ethylene production and its deteriorative effects during storage. However, ethylene within the storage environment, whether produced by the stored product or other sources is known to cause a significant stress to many harvested products. The hormone affects the rate of metabolism of many succulent plant products and is generally active at very low concentrations. In some recent studies on vegetables like broccoli, cabbage, carrots, and lettuce a very small amount of ethylene has been shown to increase the rate of respiration, alter the activity of a number of enzymes and increase membrane permeability. Cellular changes induced by ethylene result in an acceleration of senescence and the deteriorative processes that accompany it.
The early physiological, biochemical, and molecular changes that follow harvesting of asparagus are important aspects in order to define the factors contributing to postharvest deterioration. This study reports the cloning and characterization of the ACC oxidase gene from tip of harvested asparagus spears and the changes in enzyme activity and gene expression in relation to harvest-induced ethylene during storage.
Materials and Methods
Plant material: Asparagus spears were harvested from farmer’s field in Kagawa Prefecture, Japan during June-July, 2001. Spears were immediately brought to the laboratory (Faculty of Agriculture, Kagawa University, Japan) and the spear tips were stored at 20°C for five days. Assay of ethylene production was performed from non-stored (0 day) and stored spear tips (1 to 5 days storage period). After storage treatment samples were immediately stored at –80 0C until needed for assay of ACC oxidase and extraction of total RNA. The term spear tip refers to the apical 30 of 250 mm spears.
Assay of ACC oxidase activity and ethylene production: Enzyme extraction was performed according to the method described by Fernandez-Maculet and Yang (1992).
RNA isolation: Total RNA was extracted according to the Hot Borate method of Wan and Wilkins (1994).
Amplification of poly (A)+ RNA by RT-PCR: The first strand cDNA was synthesized from 2 g of the total RNA by reverse transcriptase with Oligo-(dT) primer according to the instruction of SUPER SCRIPTTM Preamplification System for First Strand cDNA Synthesis (GIBCOBRL, Tokyo, Japan). Polymerase Chain Reaction (PCR) was performed in a total volume of 25 μl containing the first strand cDNA reaction products, 10xPCR Buffer, MgCl2, dNTP, First Start Taq DNA Polymerase (Roche) and primers. The primers (5’-GCNTGYGARAAYT GGGGNTT-3’ as the upstream primer and 5’-GGYTCYTTNGCYTGRAAYTT-3’ as the downstream primer) were designed and synthesized on the basis of amino acid domains (ACENWGF and KFQAKEP respectively) conserved in various ACC oxidase genes. The Sal 1 and Not1 restriction site sequences were also included at 5’-end of the sense and antisense primer, to facilitate cloning of PCR product. The PCR procedure started with 10 min at 95 0C and was carried out 35 cycles of 30 s at 95, 50 and 72 0C and 10 min at 72°C with ASTEC Program Temperature Control System PC-700. The PCR products were confirmed by agarose gel electrophoresis.
Cloning and sequencing of cDNA: The amplified cDNA was ligated to the plasmid pSPORT1 and cloned into Escherichia coli (DH-5á ) Not 1- Sal 1 – cut (BRL, Tokyo, Japan). Sequencing was performed by the cycle sequencing method using GATCR-Bio Cycle sequencing Kit and a DNA sequencer GATC 1500 Long-Run system (GATC Gmbh, Konstanz, Germany).
Sequence data analysis: Sequence analysis was performed using computer software GENETYX-MAC Ver.7. Homology searches with the Genbank and the EMBL databases were performed using the homology program in the software. The phylogenetic tree was also constructed with the UPGMA method in the software.
Preparation of the digoxigenin (DIG)-UTP-labeled RNA probe: The cloned RT-PCR product including the encoded region of ACC oxidase gene was cleaved by Not 1 and Sal 1 from the pSPORT1 vector that had been amplified in Escherichia coli (DH-5á ) and it was purified and recovered by gel eletrophoresis. AntisenseDIG-labeled RNA probe was prepared according to the instructions of DIG RNA Labeling Kit (Boehringer Mannheim) using SP6 RNA polymerase.
Northern blot analysis: Ten μg of total RNA was subjected to electrophoresis on a 1.0 % agarose (Type II) gel containing 20 x MOPS and 37 % formaldehyde. After electrophoresis for 30 min, RNA was visualized with ethidium bromide under UV light to confirm equal loading of RNA in each lane. RNA was transferred to a positively charged nylon membrane HybondTM- N+ (Amersham Pharmacia Biotech) by capillary action with 20 x SSC and then after drying the membrane RNA was fixed under UV. The membrane was prehybridized at 50 0C with 5 x SSPE, 5 x Denhart’s solution, formamide and 10 % SDS for 3 h. Hybridization was performed at 50 0C using the gene specific antisense DIG-labeled RNA probe for 24 hours using the same prehybridization buffer. After hybridization, the membrane was washed twice with 2 x SSPE containing 0.1 % SDS for 10 min at room temperature, once with 1 x SSPE containing 0.05 % SDS for 15 min at 65 0C and once with 0.2 x SSPE for 10 min containing 0.05 % SDS at 65 0C. The membrane was also washed with a buffer containing maleic acid and Tween 20 at room temperature and was blocked with 2 % blocking reagent in maleic acid buffer for 30 minutes. Subsequently, the membrane was incubated with Anti-Digoxygenin –AP, Fab fragments (Boehringer Mannheim) in the blocking buffer for 30 min. Signals were detected by color reaction using 5-bromo-4-chloro-3-indolyl- phosphate and 4-Nitro blue tetrazolium chloride as the substrate.
Results
Isolation and identification of cDNA clone: The cDNA pAS-ACO is a partial clone encoding a harvest-induced transcript from the tips of asparagus spears. The encoded mRNA is 832 bp long (Fig. 2) and is highly homologous to the ACC oxidase gene of other plant (Fig. 3). The pAS-ACO sequence is 76.8 % identical to ACC oxidase from rice (X85747), 72.7 % identical to ACC oxidase from pea (M98357), and 73.8 % identical to the cucumber ACC oxidase (AB006806) all of which are wound-induced gene (Table 1). Allowing for conservative amino acid substitutions, the similarities are 78.9, 73.2 and 74.9 % for the rice, pea and cucumber sequences, respectively.
| Table 1: | Percentage of nucleotide and deduced amino acid homology between ACC oxidase from asparagus trip and other plants in the databases |
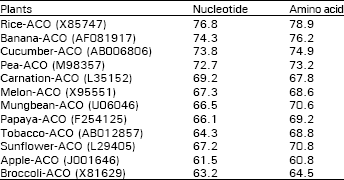 | |
| Asparagus spear tip-ACO (AB083187) is calculated as 100% | |
Harvest-induced ethylene synthesis: Ethylene production was induced in the spear tips after harvest (Fig. 1). The production rate started to increase after harvest, reaching a peak at 3 d after harvest, and then started to decline. The rate of production at the peak (3 d) was about three times higher than that of initial (0 d) production rate.
Expression of ACC oxidase gene: The expression of pAS-ACO was induced by harvesting.
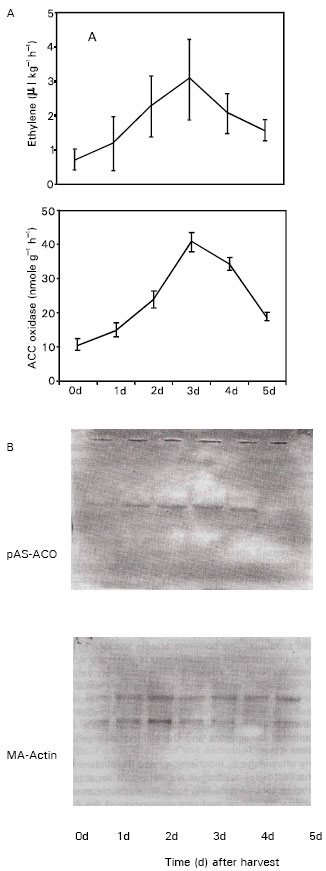 | |
| Fig. 1: | A, Changes in ethylene production and ACC oxidase activity in harvested spear tips. Each point represents the mean of three replications. Vertical bars indicates SE. B, Northern blot analysis of total RNA (10 μg lane-1) showing the expression of p AS- ACO. MA-actin was used as internal control |
 | |
| Fig. 2: | Nucleotide sequence and deduced amino acid sequence of the cDNA clone corresponding to pAS-ACO. The predicted amino acid sequence is given in single-letter code for each amino acid. The arrows indicate the positions of degenerated primers (→ sense, ← ntisense) used for RT-PCR. Numbering refers to total nucleotide residues on each line |
 | |
| Fig. 3: | Comparison of the deduced amino acid sequences from asparagus (AB083187), cucumber (AB006806), rice (X85747) and pea (M98357) by multi alignment. The amino acid residues are numbered at the beginning and end of the sequences on each line. Asterisks (*) denote the amino acid residues those are identical. Dashes in the amino acid sequences represent gaps introduced to maximize alignment of the polypeptides |
Maximum pAS-ACO expression appeared after 3 d of harvesting and coincided with the peak of ACC oxidase activity (Fig. 1) which correlated well with ethylene production.
A phylogenetic tree (Fig. 4) was generated from the alignment of the deduced amino acid sequences of pAS-ACO and other ACC oxidase gene in the data base. The pAS-ACO (accession No. AB083187), ACO from rice (X85747) and banana (AF081917) strongly clustered together in a subgroup, having closest relationship with rice. The same sub group has been identified by another phylogenetic analysis by Matsui et al. (2001).
To understand ethylene biosynthesis and its deteriorative effects on asparagus spears we have isolated a cDNA for ACC oxidase from harvested spear tips and shown that it is wound induced. The clone, pAS-ACO had high sequence similarity to rice, pea and cucumber cDNA that were also associated with wounding. Northern blot analysis revealed that the expression of pAS-ACO increased in harvested spear tips until 3 d of storage period at 20°C and afterwards it started to decline. Asparagus spear tips produced a considerable amount of ethylene in response to the wounding, associated with harvest, and ACC oxidase activity was also induced in the spear tips. The increase in ACC oxidase activity occurred concurrently with the increase in the abundance of pAS-ACO transcript (Fig. 1) suggesting well that the transcription of pAS-ACO led to the increase in wound-induced ACC oxidase activity and thus ethylene production in the spear tips. ACC oxidase can be induced by various factors such as fruit ripening (Ross et al., 1992; MacDiarmid and Gardner, 1993), auxin (Shiomi et al., 1998), wounding (Matsui et al., 2001; Kato et al., 2002), disease (Ohtsubo et al., 1999), and senescence of flower (Wang and Woodson, 1992) and vegetative tissues (Peck et al., 1993; Kim and Yong, 1994). These result was consistent with that observed in wound-induced bamboo (Matsui et al., 2001), cucumber (Shimoi et al.,1998), pea (Peck et al., 1993), and broccoli (Kato et al., 2002). However, in asparagus tips, the time lag between wounding and gene expression appeared to be longer compared with that in bamboo and cucumber.
Phylogenetic analysis of ACO sequences has revealed the existence of at least two major branches that contain characteristic conserved amino acid sequences, monocotyledon and dicotyledon, These are also subgrouped into wounding, ripening, senescence and auxin induced ACO.
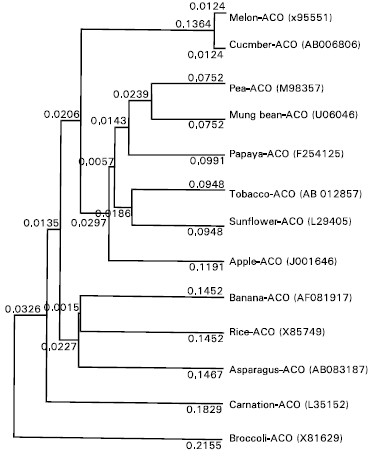 | |
| Fig. 4: | Phylogenetic tree of the alignment of pAS-ACO deduced amino acid sequence with other ACC oxidase in the database. Protein sequences were aligned using UPGMA, and a phylogenetic tree was constructed using GENETYX-MAC software. The GenBank accession numbers are shown in the parentheses |
The pAS-ACO belonged to the subgroup wound-induced monocotyledon as it was highly homologous to rice ACO and was closely related to banana ACO.
Therefore it can be concluded that induction of ethylene production in harvested asparagus tips is regulated by transcription of pAS-ACO in response to the wounding associated with harvest.
The financial support of the Ministry of Education, Science, Sports and Culture of Japan under scholarship program for foreign students is gratefully acknowledged.