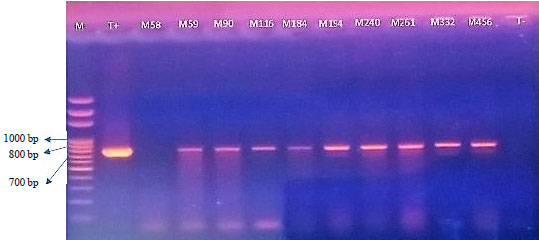
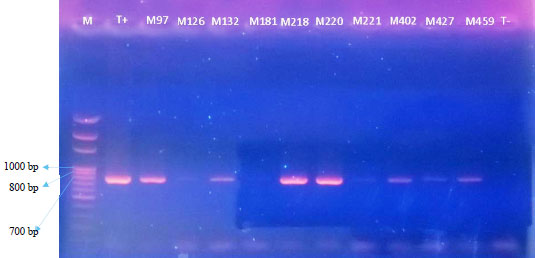
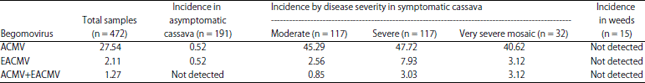
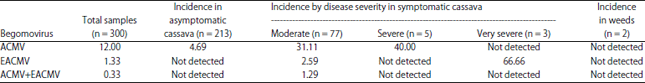
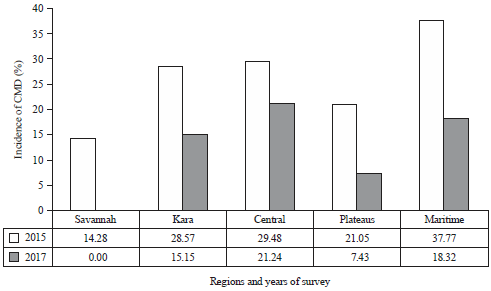
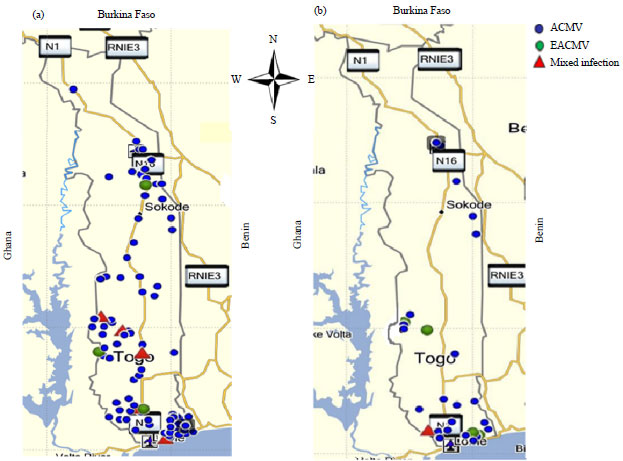
Research Article
Identification and Incidence of Cassava Mosaic Begomoviruses in Togo
Laboratory of Plant Virology and Biotechnology, Higher School of Agronomy, University of Lomé, Lomé, Togo
Kodjovi Atassé Dansou-Kodjo
Laboratory of Plant Virology and Biotechnology, Higher School of Agronomy, University of Lomé, Lomé, Togo
Djodji Kossikouma Adjata
Laboratory of Plant Virology and Biotechnology, Higher School of Agronomy, University of Lomé, Lomé, Togo
LiveDNA: 228.5838
Justin Simon Pita
Laboratory of Plant Virology, Université Félix Houphouët-Boigny, Pôle Scientifique et d'Innovation, Bingerville, Côte d'Ivoire, BP V34, Abidjan