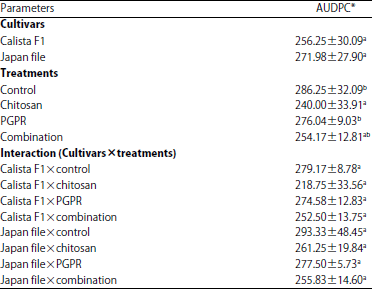
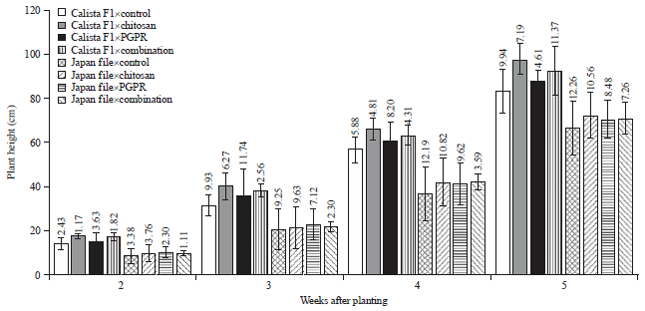
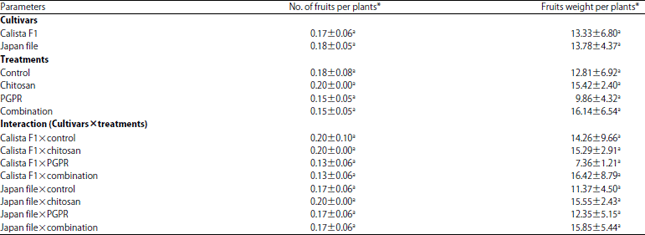
Research Article
Chitosan and Plant Growth Promoting Rhizobacteria Application to Control Squash mosaic virus on Cucumber Plants
Department of Plant Protection, Faculty of Agriculture, Bogor Agricultural University, Darmaga, 16680 Bogor, West Java, Indonesia
Widodo
Department of Plant Protection, Faculty of Agriculture, Bogor Agricultural University, Darmaga, 16680 Bogor, West Java, Indonesia
Sri Hendrastuti Hidayat
Department of Plant Protection, Faculty of Agriculture, Bogor Agricultural University, Darmaga, 16680 Bogor, West Java, Indonesia
LiveDNA: 62.16401