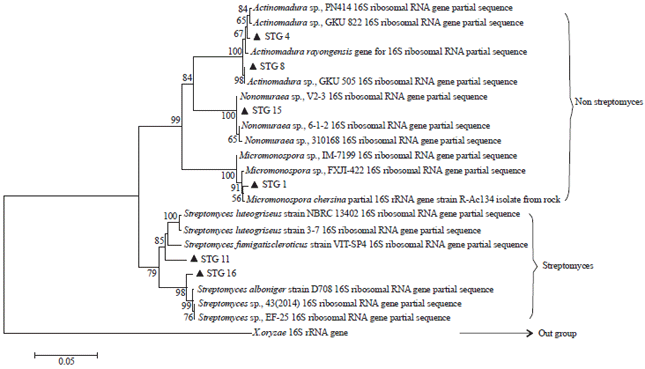
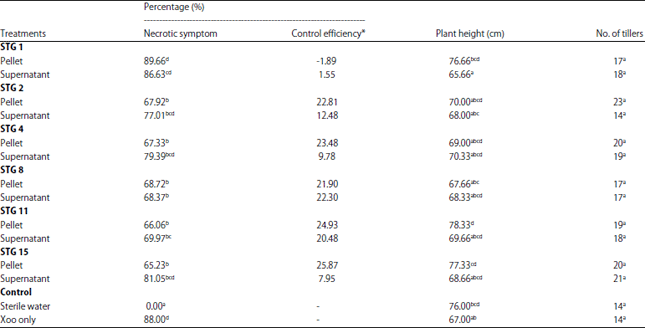
Research Article
Rice Phyllosphere Actinomycetes as Biocontrol Agent of Bacterial Leaf Blight Disease on Rice
Graduate School, Departement of Biology, Faculty of Mathematics and Natural Science, Bogor Agricultural University, Darmaga, Bogor 16680, West Java, Indonesia
Abdjad Asih Nawangsih
Department of Plant Protection, Faculty of Agriculture, Bogor Agricultural University, Darmaga, Bogor 16680, West Java, Indonesia
Aris Tri Wahyudi
Graduate School, Departement of Biology, Faculty of Mathematics and Natural Science, Bogor Agricultural University, Darmaga, Bogor 16680, West Java, Indonesia