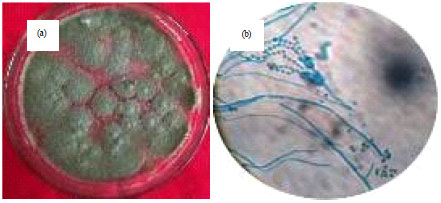
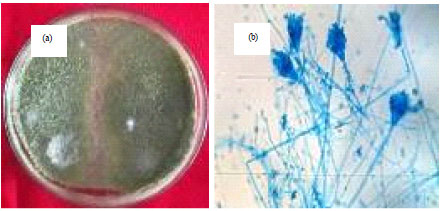
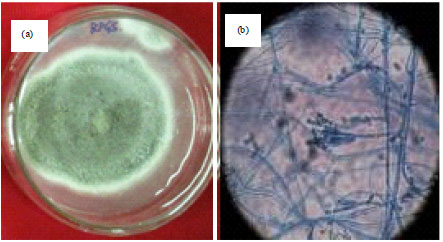
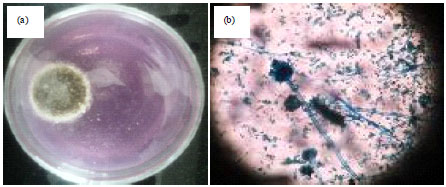
Research Article
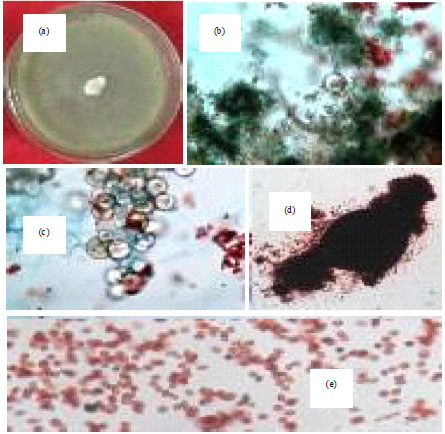
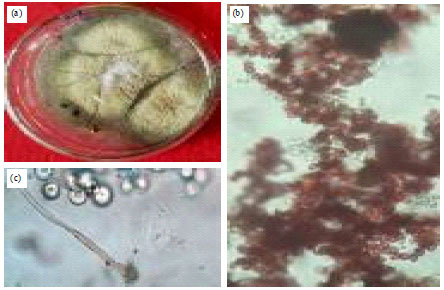
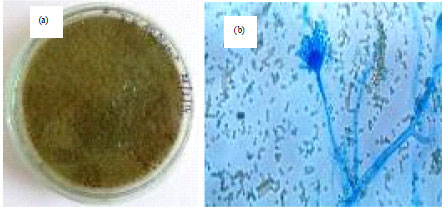
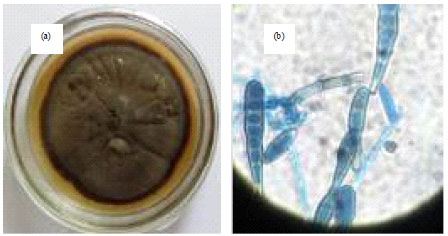
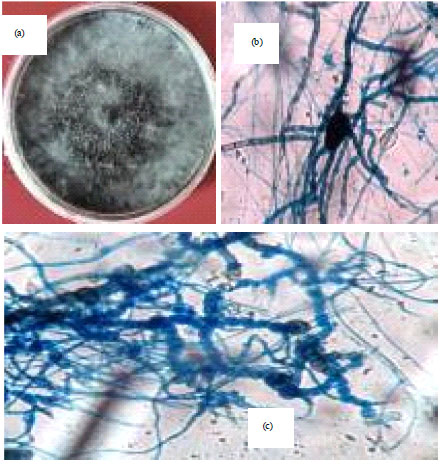
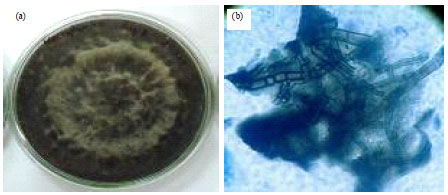
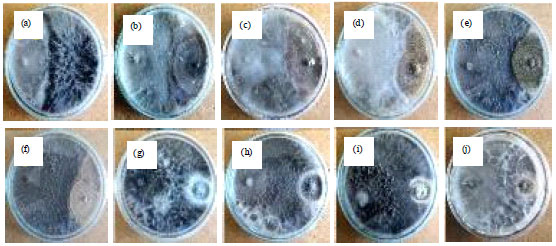
Isolation, Identification and in vitro Screening of Rhizospheric Fungi for Biological Control of Macrophomina phaseolina
Department of Microbiology, Yogi Vemana University, Kadapa, 516003, India
B. Jayapal Gowdu
Department of Microbiology, Yogi Vemana University, Kadapa, 516003, India