Research Article
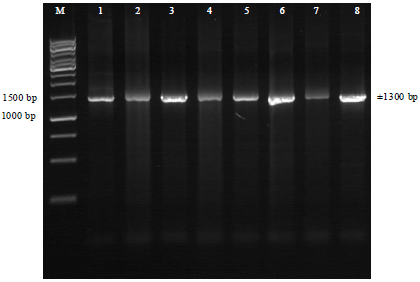
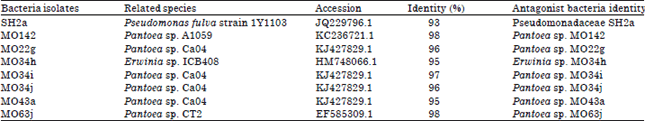
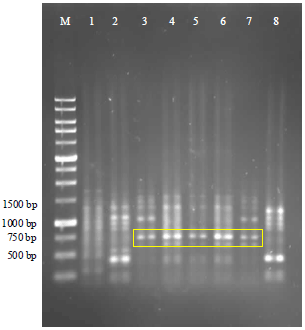
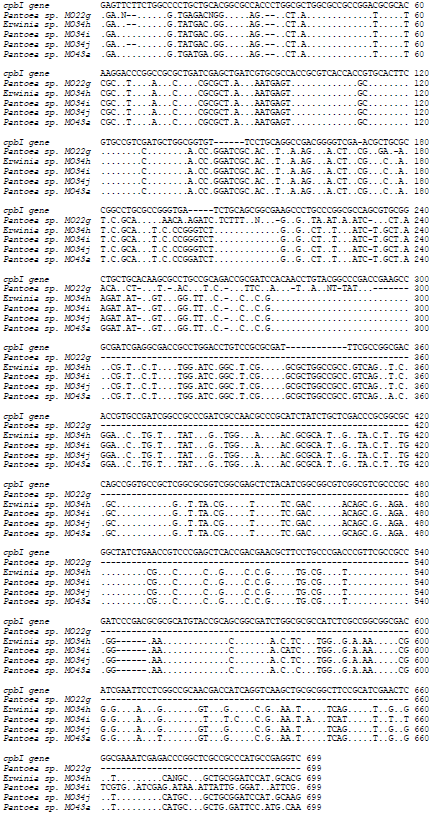
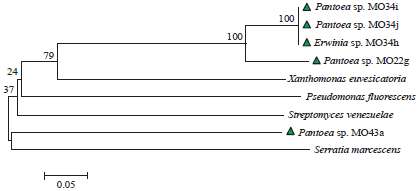
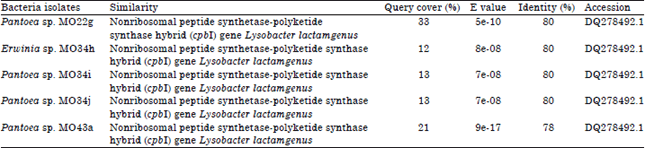
Molecular Identification of 16S rDNA and Polyketide Synthase Genes of Antagonist Bacteria against Xanthomonas oryzae Pathovar oryzae from Rice Phyllosphere
Department of Bioscience, Sebelas Maret University, Jl. Ir. Sutami 36A, Surakarta, 57126, Central Java, Indonesia
Suranto
Department of Bioscience, Sebelas Maret University, Jl. Ir. Sutami 36A, Surakarta, 57126, Central Java, Indonesia
Ari Susilowati
Department of Bioscience, Sebelas Maret University, Jl. Ir. Sutami 36A, Surakarta, 57126, Central Java, Indonesia
Aris Tri Wahyudi
Department of Biology, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, Kampus IPB Darmaga, Bogor, 16680, Indonesia