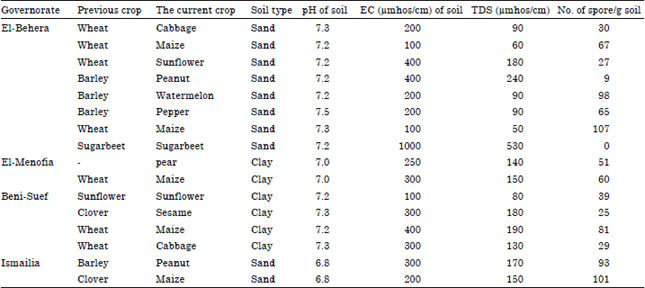
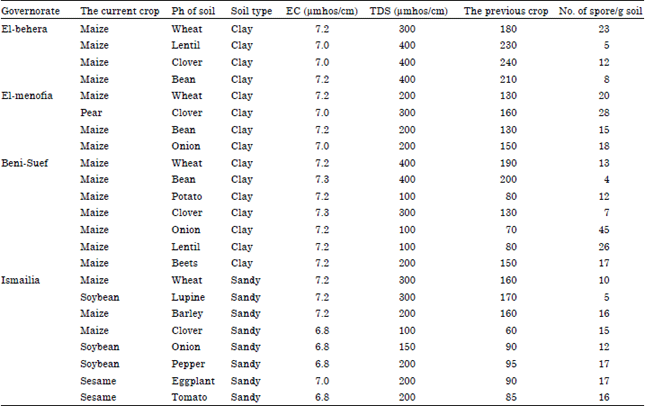
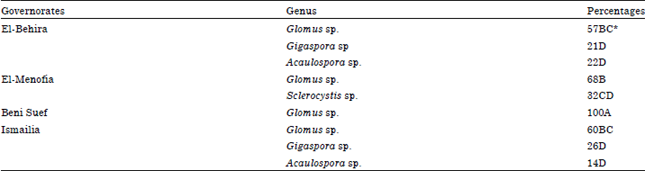
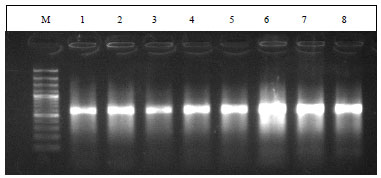
Research Article
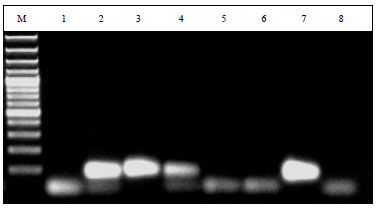
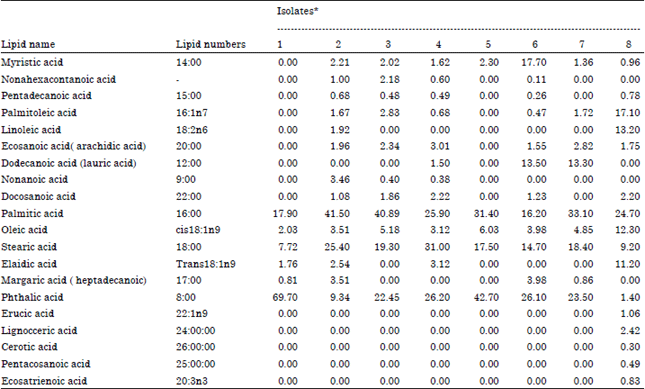
Differentiation Between Glomus Species in Egyptian Soil Using Fatty Acid Methyl Ester Profiles
Department of Plant Pathology, Faculty of Agricultural, Cairo University, Giza, Egypt
M.S. Mansour
Department of Plant Pathology, Faculty of Agricultural, Cairo University, Giza, Egypt
S.A. El-Hadad
Plant Pathology Research Institute, A.R.C., Giza, Egypt
Abeer M. Shaltout
Plant Pathology Research Institute, A.R.C., Giza, Egypt
Sozan E. El-Abeid
Plant Pathology Research Institute, A.R.C., Giza, Egypt