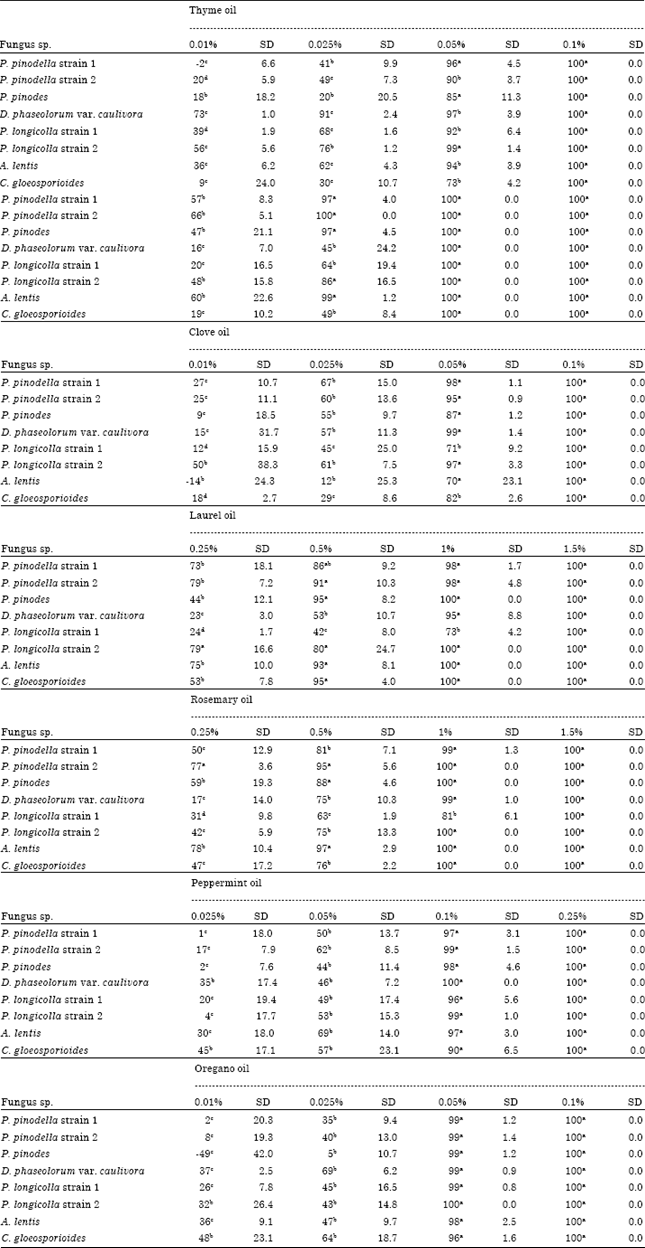
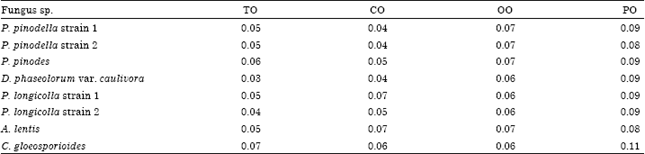
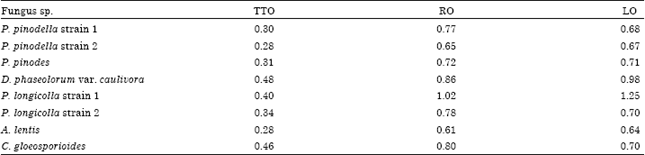
Research Article
Activity of Some Essential Oils against Pathogenic Seed Borne Fungi on Legumes
Consiglio per la ricerca e la sperimentazione in agricoltura, Centro di ricerca per la patologia vegetale (Plant Pathology Research Centre), Via C.G. Bertero, 22, I-00156 Rome, Italy
Laura Orzali
Consiglio per la ricerca e la sperimentazione in agricoltura, Centro di ricerca per la patologia vegetale (Plant Pathology Research Centre), Via C.G. Bertero, 22, I-00156 Rome, Italy
Elvira Lotti
Consiglio per la ricerca e la sperimentazione in agricoltura, Centro di ricerca per la patologia vegetale (Plant Pathology Research Centre), Via C.G. Bertero, 22, I-00156 Rome, Italy
Luca Riccioni
Consiglio per la ricerca e la sperimentazione in agricoltura, Centro di ricerca per la patologia vegetale (Plant Pathology Research Centre), Via C.G. Bertero, 22, I-00156 Rome, Italy