Research Article
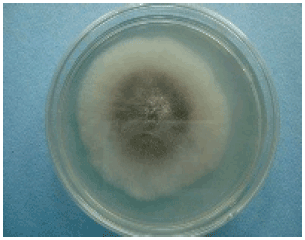
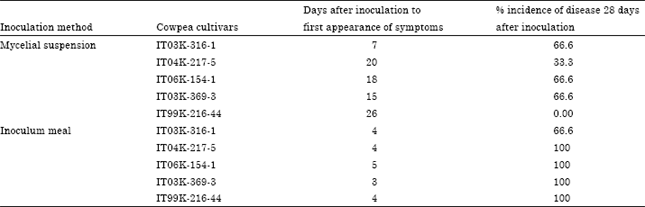
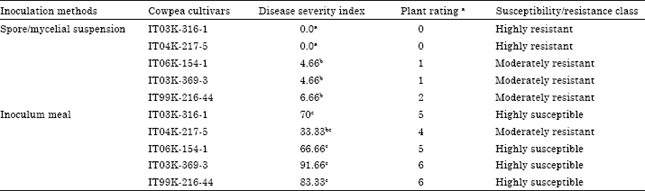
Screening of Cowpea Genotypes for Resistance to Macrophomina phaseolina Infection using Two Methods of Inoculation
Department of Crop Protection, Faculty of Agriculture, University of Ilorin, PMB 1515, Ilorin, Nigeria
O.S. Balogun
Department of Crop Protection, Faculty of Agriculture, University of Ilorin, PMB 1515, Ilorin, Nigeria
T. Shittu Busayo
Department of Crop Protection, Faculty of Agriculture, University of Ilorin, PMB 1515, Ilorin, Nigeria