Research Article
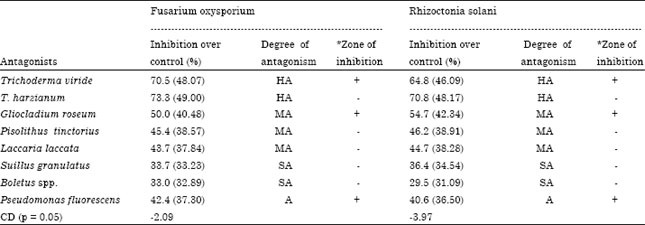
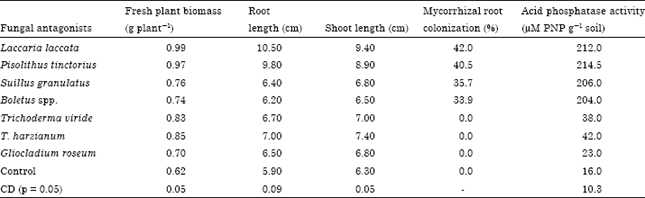
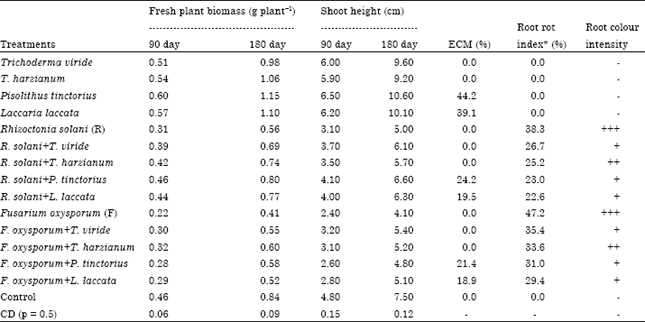
Management of Root Rot Caused by Rhizoctonia solani and Fusarium oxysporum in Blue Pine (Pinus wallichiana) Through use of Fungal Antagonists
Division of Plant Pathology, S.K. University of Agricultural Sciences and Technology of Kashmir, Shalimar, Srinagar, Jammu and Kashmir-191 121, India
M.A. Beig
Division of Plant Pathology, S.K. University of Agricultural Sciences and Technology of Kashmir, Shalimar, Srinagar, Jammu and Kashmir-191 121, India
F.A. Ahanger
Division of Plant Pathology, S.K. University of Agricultural Sciences and Technology of Kashmir, Shalimar, Srinagar, Jammu and Kashmir-191 121, India
Nadeem A. Ganai
Division of Plant Pathology, S.K. University of Agricultural Sciences and Technology of Kashmir, Shalimar, Srinagar, Jammu and Kashmir-191 121, India
M. Ashraf Ahangar
Division of Plant Pathology, S.K. University of Agricultural Sciences and Technology of Kashmir, Shalimar, Srinagar, Jammu and Kashmir-191 121, India