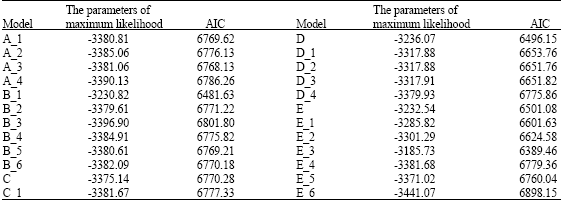
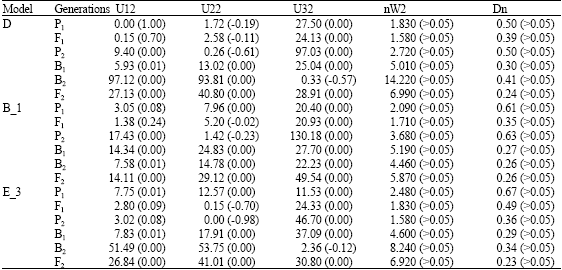
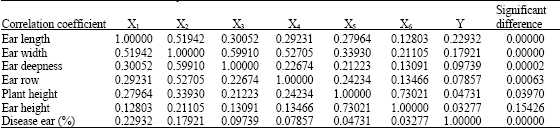
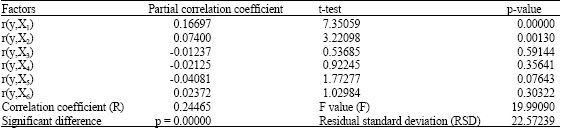
Research Article
Analysis of Gene Effects and Inheritance of Resistance to Fusarium moniliforme Ear Rot in Maize
Maize Research Institute, Sichuan Agricultural University, Ya`an, Sichuan 625014, China
Z. Fan
Maize Research Institute, Sichuan Agricultural University, Ya`an, Sichuan 625014, China
Z. Zhi-Ming
Maize Research Institute, Sichuan Agricultural University, Ya`an, Sichuan 625014, China
W. Yuan-Qi
Maize Research Institute, Sichuan Agricultural University, Ya`an, Sichuan 625014, China
Y. Ke-Cheng
Maize Research Institute, Sichuan Agricultural University, Ya`an, Sichuan 625014, China
R. Ting-Zhao
Maize Research Institute, Sichuan Agricultural University, Ya`an, Sichuan 625014, China
Y. Guang-Sheng
Maize Research Institute, Sichuan Agricultural University, Ya`an, Sichuan 625014, China
P. Guang-Tang
Maize Research Institute, Sichuan Agricultural University, Ya`an, Sichuan 625014, China