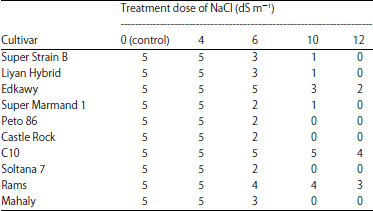
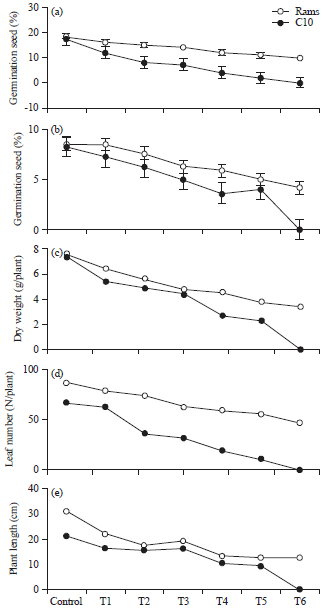
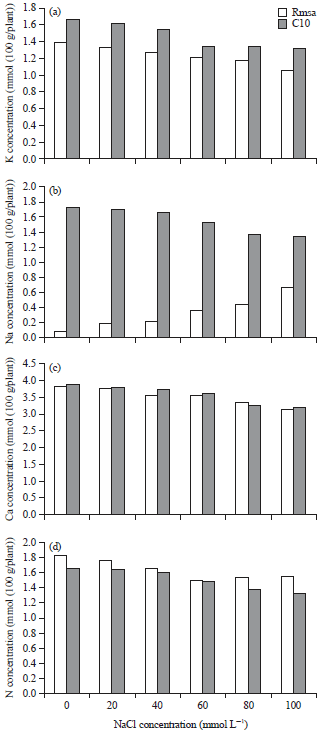
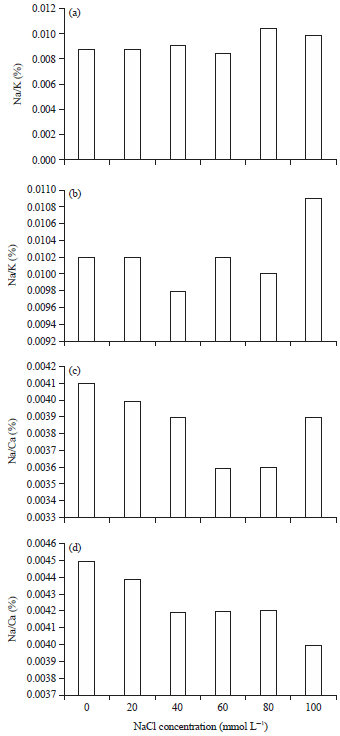
Research Article
Salt Tolerance of Some Tomato (Solanum lycoversicum L.) Cultivars for Salinity under Controlled Conditions
College of Agriculture and Food Sciences, King Faisal University, P.O. Box 55065, 31982 Al-Ahsa, Kingdom of Saudi Arabia
LiveDNA: 966.24176