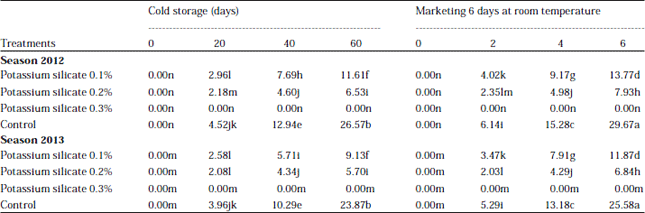
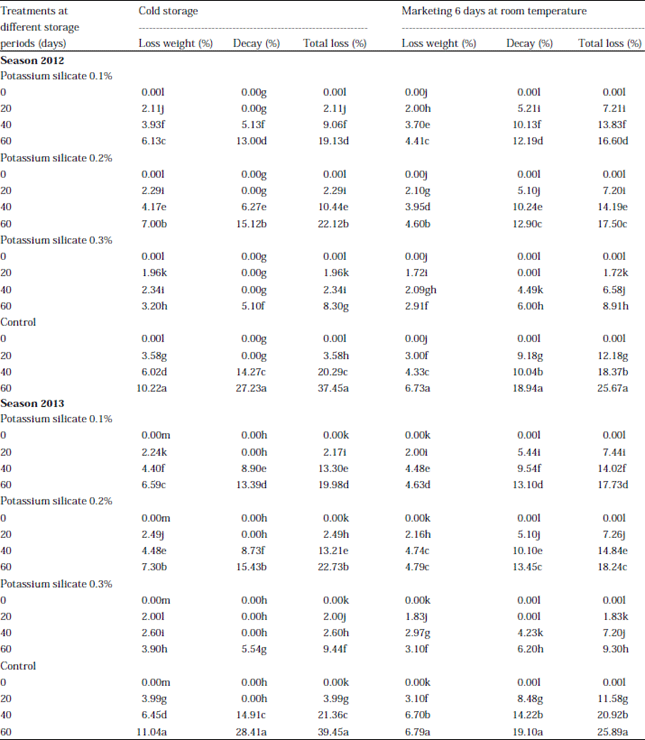
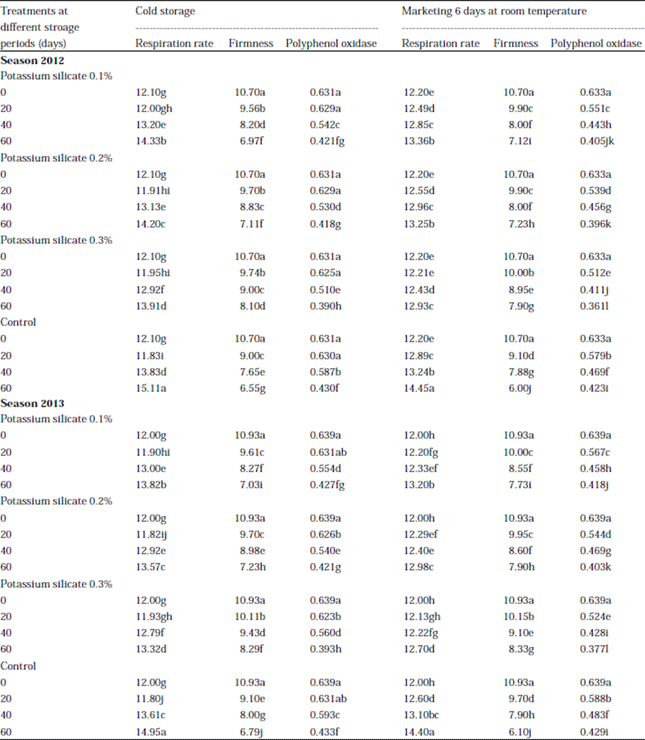
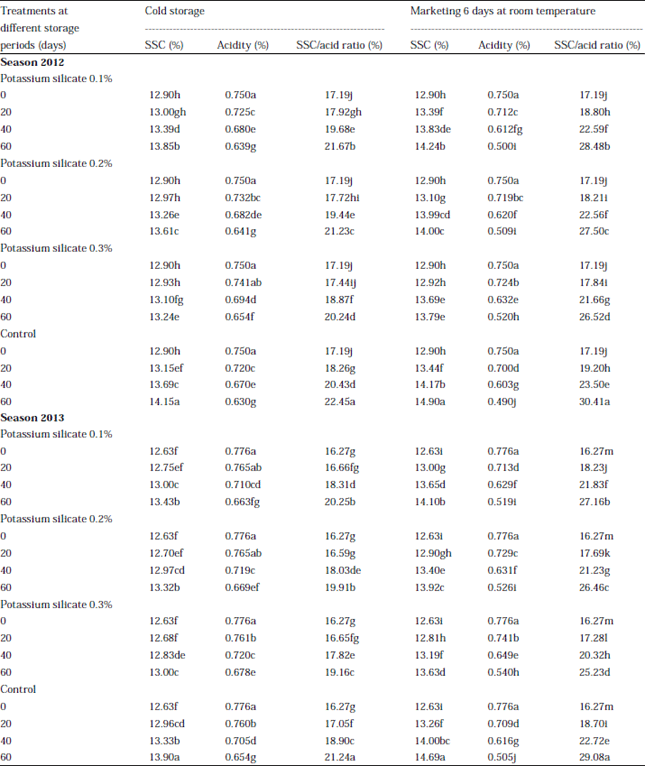
Research Article
Physiological and Pathological Impacts of Potassium Silicate on Storability of Anna Apple Fruits
Department of Fruit Handling, Horticulture Research Institute, Agricultural Research Center, Giza, Egypt
E.E. EL-Eryan
Department of Fruit Handling, Horticulture Research Institute, Agricultural Research Center, Giza, Egypt
M.A. EL-Metwally
Department of Mycological Research, Plant Pathology Research Institute, Agricultural Research Center, Giza, Egypt