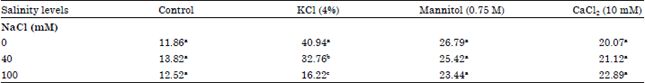
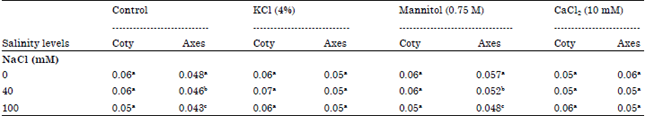
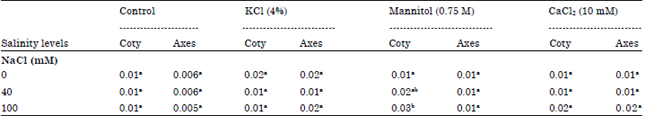
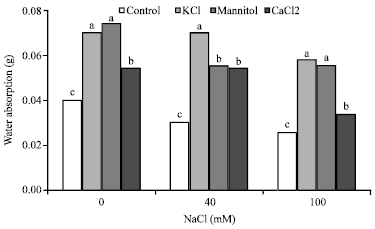
Research Article
Germination and Seedling Emergence of Primed Okra (Abelmoschus esculentus L.) Seeds under Salt Stress and Low Temperature
Laboratoire D`Agronomie, Institut Superieur Agronomique, Chott-Mariem, Sousse, 4042, Tunisia
Amani Issa
Laboratoire D`Agronomie, Institut Superieur Agronomique, Chott-Mariem, Sousse, 4042, Tunisia
Mounir Denden
Laboratoire D`Agronomie, Institut Superieur Agronomique, Chott-Mariem, Sousse, 4042, Tunisia