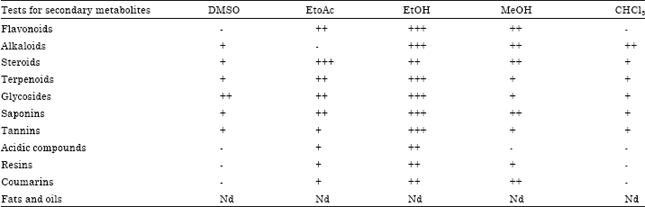
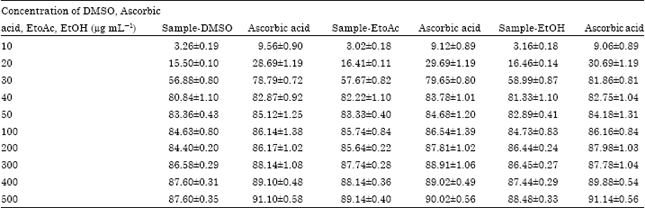
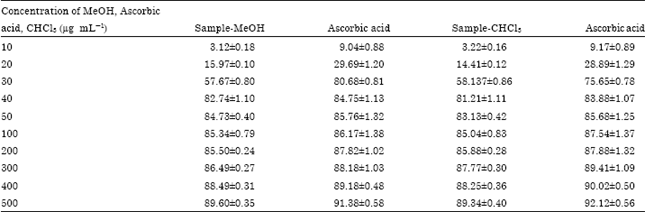
Research Article
In vitro Antioxidant and Chemical Constituents from the Leaves of Ormocarpum cochinchinense Elumbotti
Deparment of Botany, Annamalai University, Annamalai Nagar-608002, Tamil Nadu, India
D. Gajalakshmi
Deparment of Chemistry, University College of Engineering, Anna University, Ariyalur, 621 704, Tamil Nadu, India