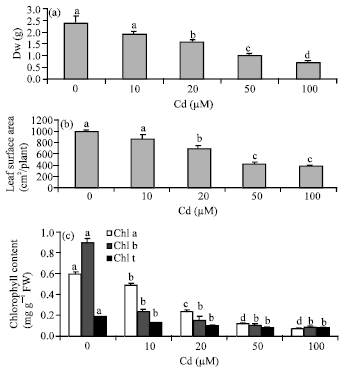
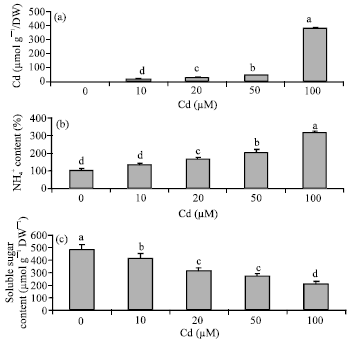
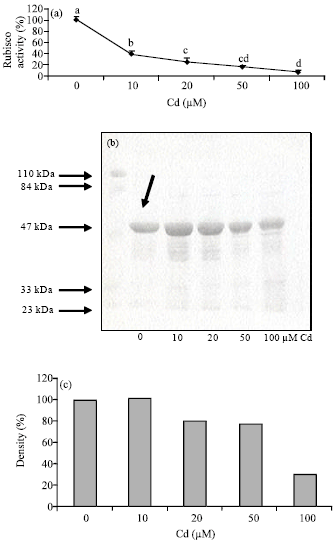
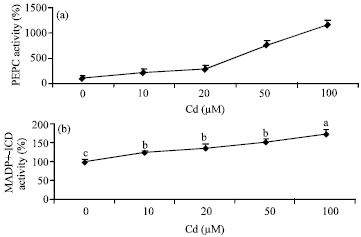
Research Article
Relationship Between Physiological and Biochemical Effects of Cadmium Toxicity in Nicotiana rustica
Unite de Recherche Nutrition et Metabolisme Azotes et Proteines de Stress, 99UR/09-20, Departement de Biologie, Faculte des Sciences de Tunis, Universite de Tunis Elmanar, 1060 Tunis, Tunisia
Saafi Leila
Unite de Recherche Nutrition et Metabolisme Azotes et Proteines de Stress, 99UR/09-20, Departement de Biologie, Faculte des Sciences de Tunis, Universite de Tunis Elmanar, 1060 Tunis, Tunisia
Bouthour Donia
Unite de Recherche Nutrition et Metabolisme Azotes et Proteines de Stress, 99UR/09-20, Departement de Biologie, Faculte des Sciences de Tunis, Universite de Tunis Elmanar, 1060 Tunis, Tunisia
Gouia Houda
Unite de Recherche Nutrition et Metabolisme Azotes et Proteines de Stress, 99UR/09-20, Departement de Biologie, Faculte des Sciences de Tunis, Universite de Tunis Elmanar, 1060 Tunis, Tunisia
Chaffei-Haouari Chiraz
Unite de Recherche Nutrition et Metabolisme Azotes et Proteines de Stress, 99UR/09-20, Departement de Biologie, Faculte des Sciences de Tunis, Universite de Tunis Elmanar, 1060 Tunis, Tunisia