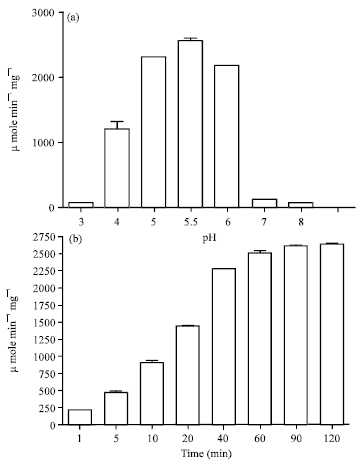
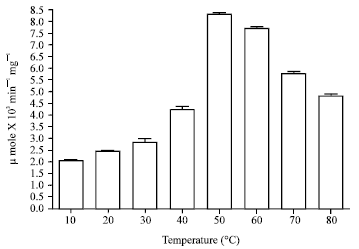
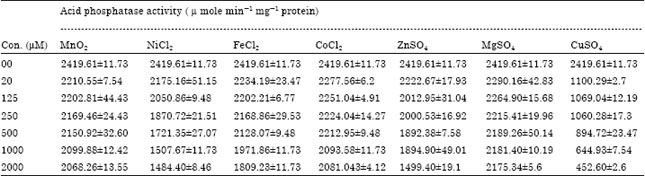
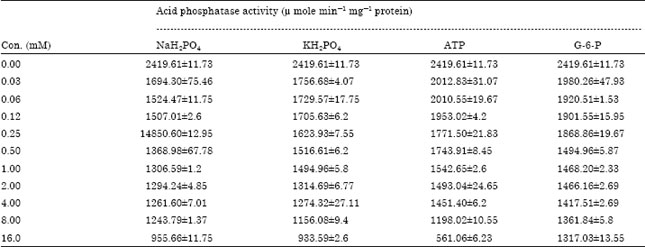
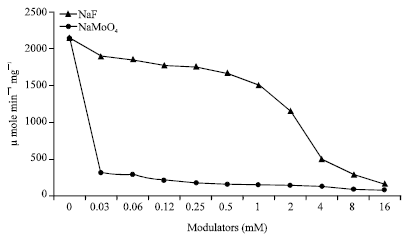
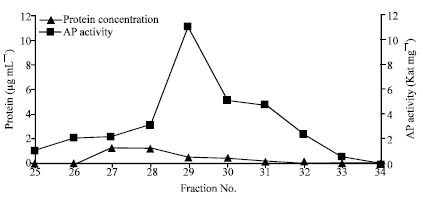
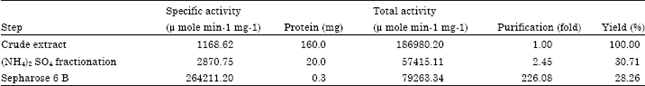
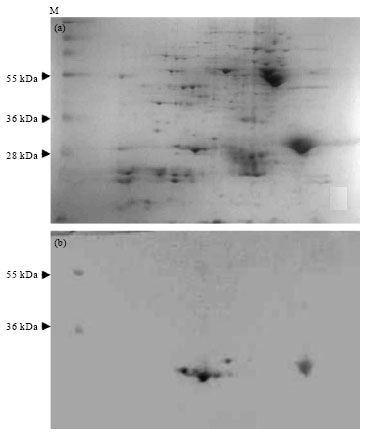
Popular Article
Identification, Characterization and Partial Purification of Acid Phosphatase from Cotyledons of Psoralea corylifolia L.
Medicinal Chemistry Division, Dr. B.R. Ambedkar Center for Biomedical Research, University of Delhi, Delhi 110007, India
Pratibha Mehta Luthra
Medicinal Chemistry Division, Dr. B.R. Ambedkar Center for Biomedical Research, University of Delhi, Delhi 110007, India