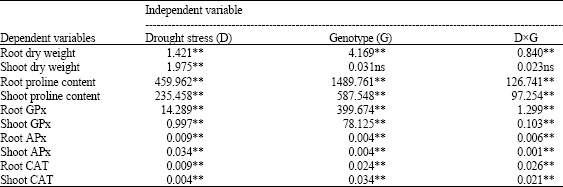
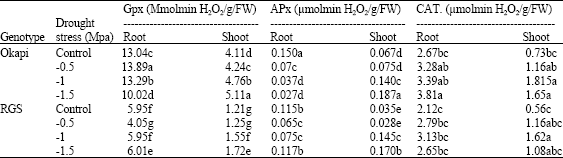
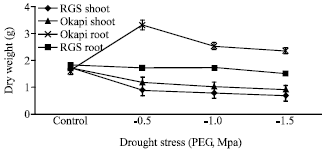
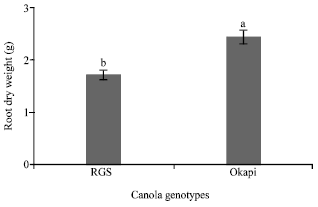
Research Article
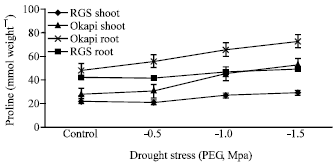
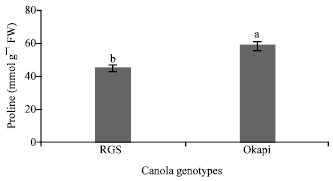
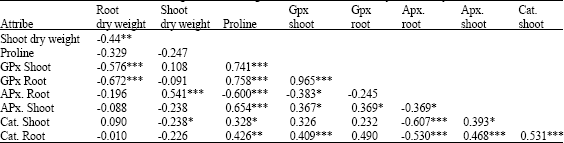
Changes of Proline Content and Activity of Antioxidative Enzymes in Two Canola Genotype under Drought Stress
College of Agricultural Sciences, Shahed University, P.O. Box 18151/159, Tehran, Iran
LiveDNA: 98.1127
ORCID: 0000-0002-2728-1469