Research Article
Carbohydrate Compositions and Peroxidase Activity in Ungerminated, Cotyledon and Embryo Tissues of Vigna unguiculata L. Walp Seed Grown Under Stress Temperatures
Department of Agriculture, University of Arkansas at Pine Bluff, 1200 North University Drive, Mail Slot 4913, Pine Bluff, AR 71601, USA
R. Carvajal R. Carmen
Mississippi State University, Mississippi, USA
Jr. James O. Garner
Department of Agriculture, University of Arkansas at Pine Bluff, 1200 North University Drive, Mail Slot 4913, Pine Bluff, AR 71601, USA









anuj k. ahlavat Reply
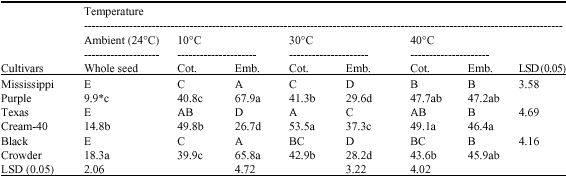
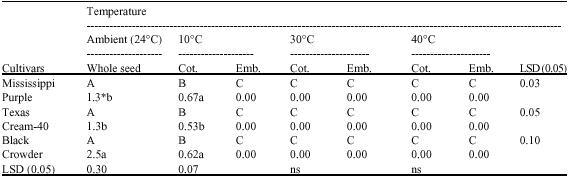
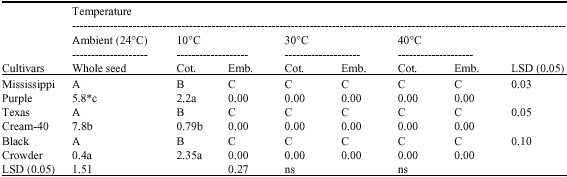
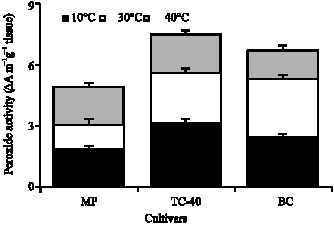
the paper is good enough to increase the knowledge about biochemical status of cotyledons and embryo during germination.