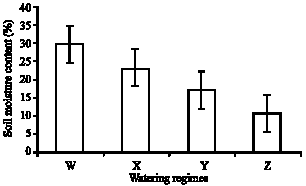
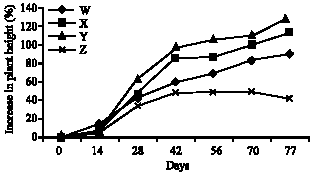
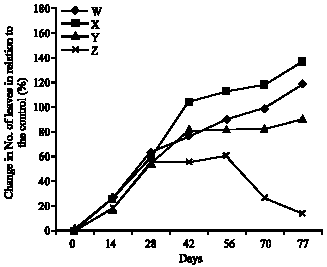
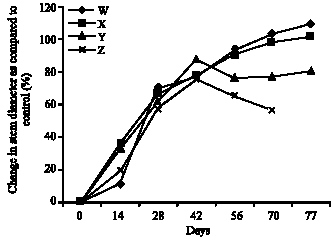
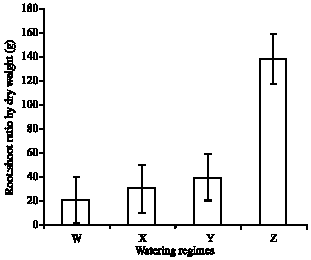
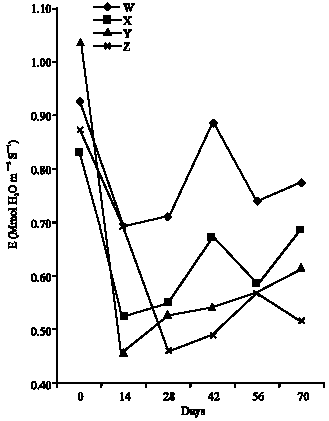
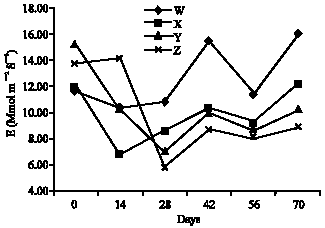
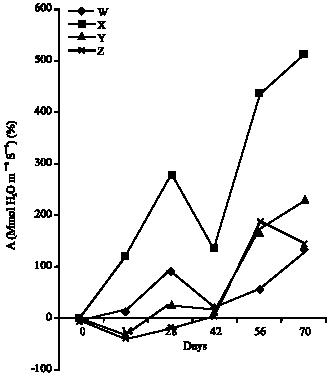
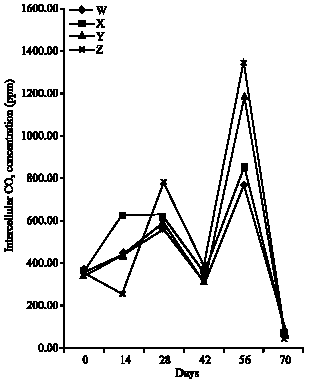
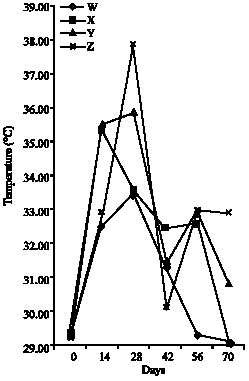
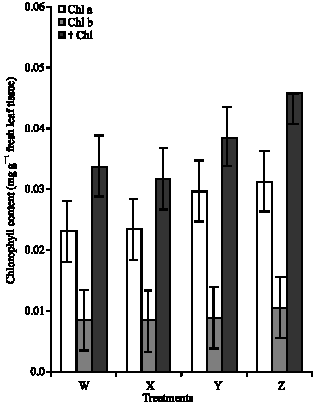
Research Article
Effect of Water Deficit on the Physiological and Morphological Characteristics of Mango (Mangifera indica) Rootstock Seedlings
Department of Botany and Horticulture, P.O. Box 333, Maseno, Kenya
G.W. Netondo
Department of Botany and Horticulture, P.O. Box 333, Maseno, Kenya
G. Ouma
Department of Botany and Horticulture, P.O. Box 333, Maseno, Kenya