Research Article
Different Light Spectral Qualities Influence Sterol Pool in Porphyridium cruentum (Rhodophyta)
Department of Botany, Faculty of Science, Muharram Beck, Alexandria University, Alexandria, Egypt
Light availability is a function of the geographic location and the local climatic parameters (Duffie and Beckman, 1980). The spectral quality of the available light may vary as a result of selective absorption by various materials suspended or dissolved in the water column (Kirk, 1983). The variability of light components such as red, green, blue (Talarico and Maranzana, 2000) and ultraviolet radiations (Villafane et al., 2005) seems to play a role of regulating algal metabolism at the cellular and molecular levels (Bhandari and Sharma, 2006). Sterol is integral component of eukaryotic cell membrane lipid which serves in different vital physiological processes. Therefore the concentration of the sterol is a very important characteristic to determine the physiological state of microalgae (Fabregas et al., 1997) hence the study of the influence of light spectral qualities on the sterol pool is of interest.
Porphyridium cruentum (Ag.) Nägeli is a primitive rhodophycean microalga (Bangiophycidae) with spherical cells (5-8 μm) that lacking a cell wall (Ramus et al., 1989). The alga has an economic value since it is a source of sulphurized polysaccharides (Arad et al., 1985), tocopherol, vitamin K, isoprenoid quinines, carotenes (Antia et al., 1970) and phycoerythrin (Gantt, 1981) which is responsible for the characteristic red color of the alga. P. cruentum accumulates large amount of lipid (Yongmanitchai and Ward, 1992), it is an excellent source of different fatty acids especially arachidonic acid (C20:4ω6) and ecosapentaenoic acid (C20:5ω3) (Lee and Tan, 1988). Beastall et al. (1971) confirmed the presence of sterol in the alga followed by the detection of relatively high proportion of 4α-methylsterol fractions (Beastal et al., 1974). Therefore, the objective of this study was to evaluate the influence of different light spectral qualities on the concentration of the total, free and conjugated sterolic forms and their composition in P. cruentum.
Biological Material and Culture Conditions
Porphyridium cruentum was obtained from culture collection of algae laboratory, Faculty of Science, Alexandria University, Egypt. The alga was grown xenically in triplicate 1 L glass flasks with sterilized and enriched sea water (1g 100 mL KNO3; 0.1g 100 mL K2HPO4; 0.1g 100 mL MgSO4. 7H2O; 10 g/100 mL yeast extract) according to Jones et al. (1963). pH was 7.6 and the cultures were bubbled with air enriched with 2% (v/v) CO2 and incubated at 24±1°C. Light intensity was 100 μmol photons m-2 s-1 with a 12/12 h light/dark cycle provided from white (visible, 350-750 nm) and the monochromatic spectrum of yellow, green, red and blue fluorescent lamps (ROLA T12, China), corresponding to the wavelengths ~580, ~550, ~680 and ~470 nm, respectively. Cells grown under the monochromatic spectrum were collected at the concentration adjusted to those grown under the visible light of about 12x106 cells mL-1 at the beginning of the stationary growth phase. All Cultures were harvested by centrifugation and the pellets were immediately lyophilized.
Sterol Isolation and Analysis
One g dried samples of P. cruentum grown under different light spectral qualities was extracted in a Soxhlet apparatus for 4 h with diethyl ether. A part of total lipid was analyzed for total sterols according to Nadal (1971). The remaining part of total lipid was fractionated according to Véron et al. (1996b) by preparative Thin Layer Chromatography (TLC) developed in the first dimension in hexane: Ethyl acetate 92:2 (v/v) to separate the Steryl Esters (SE). And in a second dimension in dichloromethane: methanol: water, 90:10:0.5 (v/v) to separate Free Sterols (FS), Steryl Glycoside (SG) and Acyl Steryl Glycoside (ASG) fractions. FS, SE, SG and ASG bands were located according to Rf values of a commercial plant sterol mixture standards (Plant Sterol Mixture, Matreya Inc.). Spots of standards were visualized with Libermann-Burchard reagent. The different bands were scraped off and eluted with dichloromethane for FS and SE and with 2:1 (v/v) dichloromethane: Methanol for SG and ASG. SE were saponified by a 1 h reflux with methanolic KOH (6% w/v) and 0.5% (w/v) pyrogallol. SG and ASG were separately hydrolyzed by a 4 h reflux with ethanolic H2SO4 (1% v/v). Sterols were recovered by partition into hexane and acylated at room temperature in the dark for 48 h using acetic anhydride in anhydrous pyridine. Acetyl derivatives were purified by TLC on silica gel plates developed in dichloromethane with cholesterol acetate as the standard. Location was determined as described above. Sterols generated from preceding fractions were identified by comparison of their RRt with those of commercial standards using Gas Liquid Chromatography (GLC). Quantification was done by comparison of the sterol peaks area with the area of a known quantity of 5α-cholestane used as an internal standard.
Statistical Analysis
All analyses were made in three replicas, the means and the Standard Deviations (SD) were calculated.
Effect of Light Spectral Qualities on Total and Sterol Profile Contents
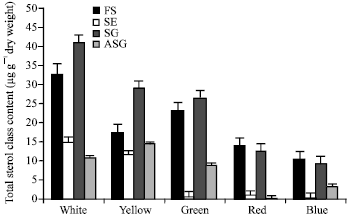
The total sterol contents (expressed as μg/g dry wt) of P. cruentum grown under different light spectral qualities are shown at Fig. 1. The results revealed a gradual decrease in the total sterol amount in the cells grown under different light spectral qualities according to the order: white>yellow>green> red>blue. The slow down in the sterol production in the cells grown under blue light was approximately 4-fold reduction of those grown under white light.
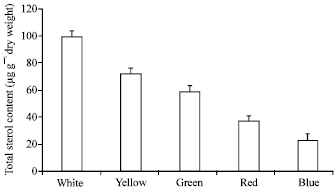 | |
| Fig. 1: | Total sterol content in Porphyridium cruentum grown under different light spectral qualities |
| Table 1: | Concentration of sterol profiles in Porphyridium cruentum grown under different light spectral qualities (data expressed as μg g-1 dry weight±SD) |
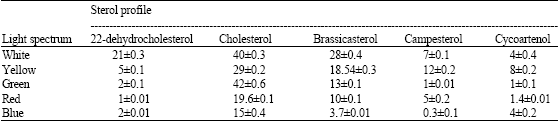 | |
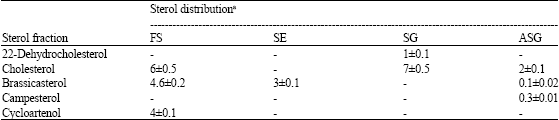
As described above sterol profiles have been isolated and identified according to their Rf values using TLC and the comparison of their RRt with those of commercial standards using GLC. In cells grown under visible light, the results revealed the presence of five sterol molecules; cholesta-5,22-diene-3β-ol (22-dehydrocholesterol); cholest-5-en-3β-ol (cholesterol); 24-Methylcholesta-5,22-diene-3β-ol (brassicasterol); 24-Methylcholest-5-en-3β-ol (campesterol) and cycloartenol.
In cells grown under different light spectral qualities, no change in the composition has been observed, but the amounts were strongly affected by the type of the spectrum used. In this study, whatever the spectral quality of light used, cholesterol was the dominant individual detected (Table 1).
Effect of Light Spectral Qualities on the Content of Different Sterol Classes
Our results demonstrated the presence of four sterol classes in P. cruentum; FS, SE, SG and ASG in cells grown under white illumination (Fig. 2). SG compounds were dominated (41% of the total sterols) followed by FS compounds (33%). SE and ASG compounds were 15 and 11%, respectively. In cells grown under yellow and green illuminations, SG compounds were the majors, while in cells grown under both red and blue light, FS compounds were dominated. The major amount of SE and ASG compounds was detected in cells grown under white and yellow light, respectively. The repartition of sterols between the different classes was found to be strongly dependent on the type of illumination.
Effect of Light Spectral Qualities on the Concentration of Sterol Composition of Each Class
Cholesterol was the dominant profile and mainly found as SG (Table 2-6). While brassicasterol was the major constituent of FS in cells grown under white, yellow and red light. Twenty two-dehydrocholesterol was found as SG under yellow, red and blue light, while under green light, it was found as ASG. Campesterol was found as ASG in the cells grown under blue light, but under green and red light, it detected as SE. Cycloartenol, the first molecule formed in the sterol biosynthesis, was distributed among different classes depending on the type of illumination.
| Table 2: | Concentration of sterol profiles of free and conjugated classes in Porphyridium cruentum grown under white light (data expressed as μg g-1 dry weight±SD) |
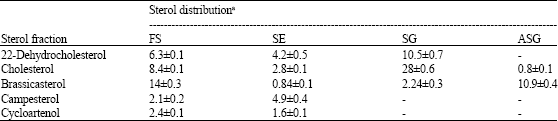 | |
| Table 3: | Concentration of sterol profiles of free and conjugated classes in Porphyridium cruentum grown under yellow light (data expressed as μg g-1 dry weight±SD) |
 | |
| Table 4: | Concentration of sterol profiles of free and conjugated classes in Porphyridium cruentum grown under green light (data expressed as μg g-1 dry weight±SD) |
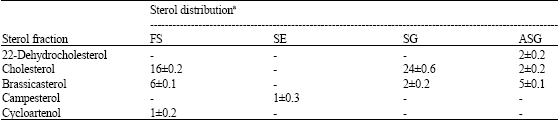 | |
| Table 5: | Concentration of sterol profiles of free and conjugated classes in Porphyridium cruentum grown under red light (data expressed as μg g-1 dry weight±SD) |
 | |
| Table 6: | Concentration of sterol profiles of free and conjugated classes in Porphyridium cruentum grown under blue light (data expressed as μg g-1 dry weight±SD) |
 | |
| aFS, free sterols; SE, steryl esters; SG, steryl glycosides; ASG, acyl steryl glycosides, -not detected | |
 | |
| Fig. 2: | Concentration of free and conjugated sterol classes in Porphyridium cruentum grown under different light spectral qualities. (FS, free sterols; SE, steryl esters; SG, steryl glycosides; ASG, acyl steryl glycosides) |
Our data here showed a dramatic decrease in the total sterol amount (25% of those grown under white light) in cells grown under blue light. The results agree with those obtained by Véron et al. (1996a) on the diatom Phaeodactylum trocornutum grown at 23°C. In this respect, Ma et al. (2001) concluded that the regulated genes in all light qualities were estimated to account for approximately one-third of the genome. Analysis of those light-regulated genes revealed more than 26 cellular pathways and they were distributed amongwith three-fifths up regulated and two-fifths down regulated by light. However, genes for sterol biosynthesis pathway were classified of down regulated type by light.
Sterol pattern in P. cruentum supports its systematic position among the primitive types of rhodophytes; the presence of cycloartenol with absence of stigmasterol and β-sitosterol. As demonstrated by Cvejic and Rohmer (2000), sterol biosynthesis in P. cruentum takes place in the cytoplasm via the mevalonic acid pathway, which is strongly influenced by the light (Bush and Grunwald, 1973). According to our results, cholesterol was the most abundant profile (40% of the total sterol content) produced in cells grown under white illumination, followed by brassicasterol. The profile concentration was found to be depending on the type of spectrum used; however, sterol composition was not affected. As explained by Fabregas et al. (1997) the differences in the sterol profile concentrations could be attributed to their dissimilar response towards the difference in the light availability.
The concentration and distribution of sterol profiles among the free and conjugates were shown here to be closely dependent on subjected light quality. The slowing down of the biosynthesis is accompanied by the decrease in the sterol amounts. In contrast to higher plants (Wojciechowski, 1991) and other species of Porphyridium (Duperon et al., 1980), FS compounds are not the dominant class in control culture, since 52% of the total sterols are glycosylated. However, FS slightly increased in cells grown under red and blue light. The data here showed a shift in the sterol amounts and distribution, this alteration, as postulated by Tuckey et al. (2002), mainly depend on the light type. Therefore,the sterol metabolism is greatly influenced by the chloroplast system (Anding et al., 1971).
Whatever the light conditions, cholesterol was the major component and mainly detected as SG form. This result go parallel with a decrease in the concentration of brassicasterol indicating, as explained by Véron et al. (1996a), an inhibition of the first methylation step at C-24. SG and ASG sterols are biosynthesized by a UDP-glucose: Sterol glucosyltransferase in eukaryotic organisms and the genes encoded the enzyme can use different sterols such as cholesterol as sugar acceptors. The biosynthesis of this enzyme is extremely affected by environmental stress (Warneckeet al., 1999). SE compounds detected in a minor amount in the cells grown under green, red and blue light; while this could reflect differences in responses to environmental variables (Jensen-Pergakes et al., 2001).
Ballantine et al. (1979) attributed the alteration in sterol pool by different spectral light qualities is a result of switching between 27C Δ7sterol and 28CΔ7 sterol in marine phytoplankton.
Jensen-Pergakes et al. (2001); Wentzinger et al. (2002); Laule et al. (2003) reported that, conjugation of sterols is a critical homeostatic response by all eukaryotic cells to an excess of either resource. The intracellular esterification reaction is mediated by enzymes known collectively as O-acyltrnsferases. The biosynthesis and activity of acyltransferases is closely correlated withlight induction and it is completely neglected in cells grown under complete darkness (Jelsema et al., 1982).