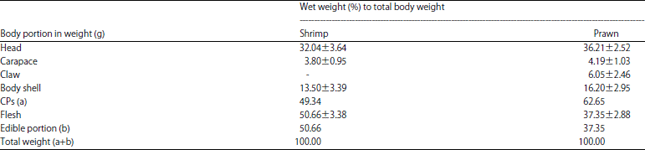
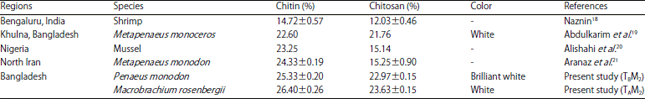
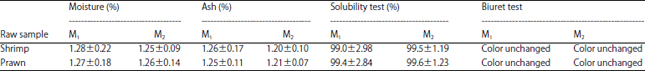
Research Article
Extraction and Worth Evaluation of Chitosan from Shrimp and Prawn Co-products
Department of Fisheries and Marine Science, Noakhali Science and Technology University, 3814 Noakhali, Bangladesh
M.S. Islam
Department of Fisheries and Marine Science, Noakhali Science and Technology University, 3814 Noakhali, Bangladesh
M.U.M.A. Zakaria
Department of Fisheries and Marine Science, Noakhali Science and Technology University, 3814 Noakhali, Bangladesh
S.C. Paul
Department of Applied Chemistry and Chemical Engineering, Noakhali Science and Technology University, 3814 Noakhali, Bangladesh
A.A. Mamun
Department of Fisheries and Marine Science, Noakhali Science and Technology University, 3814 Noakhali, Bangladesh
LiveDNA: 880.32397