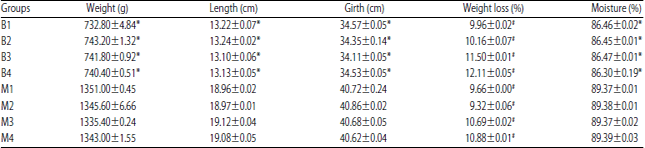
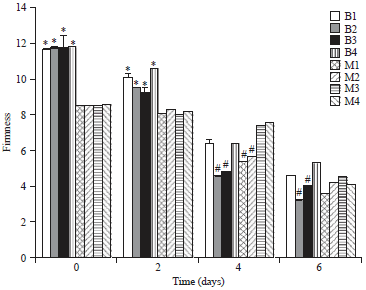
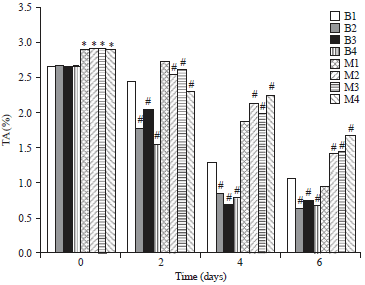
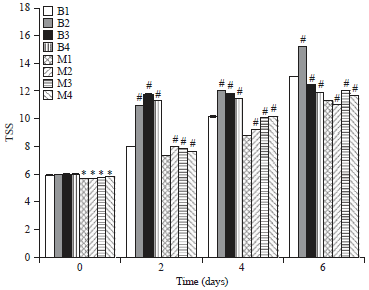
Research Article
Calcium Carbide Treatment on Some Physiochemical Characteristics of Broken and Mummy Mango Fruits
Department of Chemistry, Faculty of Science, Benue State University Makurdi, P.M.B 102119, Nigeria
LiveDNA: 234.24222
Ubwa Terver Simon
Department of Chemistry, Faculty of Science, Benue State University Makurdi, P.M.B 102119, Nigeria
Ahilem Ungwanen John
Department of Chemistry, Faculty of Science, Benue State University Makurdi, P.M.B 102119, Nigeria
Obochi Oche Godwin
Department of Biochemistry and Haematology, College of Health Sciences, Benue State University Makurdi, Nigeria
Nwannadi Ikenna Alexander
Department of Biochemistry and Haematology, College of Health Sciences, Benue State University Makurdi, Nigeria
Yusufu M. Ikagu
Department of Food Science, Federal University of Agriculture Makurdi, Nigeria