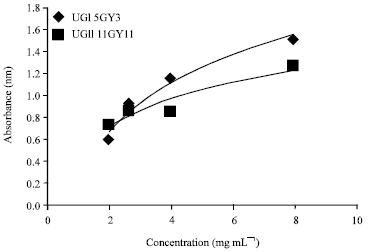
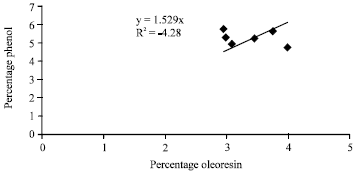
Research Article
Physico-chemical Properties and Antioxidant Potentials of 6 New Varieties of Ginger (Zingiber officinale)
Department of Biochemistry, National Root Crops Research Institute, Umudike, Umuahia, Abia State, Nigeria
LiveDNA: 234.33136
K.C. Eleazu
Department of Biochemistry, National Root Crops Research Institute, Umudike, Umuahia, Abia State, Nigeria
LiveDNA: 234.3349