Research Article
Antioxidative Effects of Pomposia Extract, on Lipid Oxidation and Quality of Ground Beef during Refrigerated Storage
Department of Biochemistry, Faculty of Agriculture, Cairo University, 12613, Giza, Egypt
LiveDNA: 20.16729
Fresh meat products are commonly marketed at refrigerated temperatures (2-5°C). However, many undesirable changes of the products can occur during refrigeration due to microbial growth and lipid oxidation, which give rise to quality reduction, meat spoilage and economic loss. Lipid oxidation is a major cause of quality deterioration in meat and its cooked products. Ground meat tends to become rancid and brown more rapidly, due to pigment and lipid oxidation. An oxidative reaction in muscle foods leads to degradation of lipid and proteins, resulting in deterioration of flavour, texture and nutritive value and is considered as one of the major problems in the development of new convenient meat products and processes (Gray and Pearson, 1987). Lipid oxidation is often responsible for quality loss via formation of rancid flavour (Asghar et al., 1988) and is affected by the duration and temperature of storage of meat (Sun et al., 2002; Smet et al., 2005; Tan and Chen, 2005).
Moreover, mechanisms for the control of lipid oxidation in meats have become increasingly important with the rise in popularity of pre-cooked and convenience foods. Many substances have been investigated as potential antioxidants to prevent such lipid oxidation. These include chemical substances such as Butylated Hydroxyanisole (BHA) and Butylated Hydroxytoluene (BHT). However, the use of synthetic antioxidants in food has been decreased due to their suspected action as promoters of carcinogenesis, as well for the general consumer rejection of synthetic food additives (Namiki, 1990; Djenane et al., 2004; Yanai et al., 2004). Several studies indicated that the use of synthetic antioxidants has begun to be restricted because of their health risks and toxicity (Williams et al., 1990; Duve and White, 1991; Buxiang and Fukuhara, 1997; Hirose et al., 1998; Farag et al., 2006). Therefore, the importance of replacing synthetic antioxidants with natural antioxidants from oilseeds, spices and other plant materials has greatly increased. In the past few years, various plant materials containing phenolic compounds have been demonstrated to be effective antioxidants in model systems. Flavonoids, the most potent antioxidative compounds of plant phenolics occur in vegetables, fruits, berries, herbs and tea leaves (Shahidi and Wanasundara, 1992; Kandaswami and Middleton, 1997; Skrede and Wrolstad, 2002; Das et al., 2006; Shaker, 2006; Farag et al., 2006, 2007; El-Anany, 2007). Non-meat ingredients that have antioxidative and/or reducing activity can stabilize meat color thus extending shelf life of meat and meat products (Greene et al., 1971; Mitsumoto et al., 1991a,b). Jamun (Syzygium cuminii L.) is a very common, large evergreen beautiful tree of Indo-Pak subcontinent. It belongs to Myrtaceae plant family. Its fruit is generally round in shape, deep purple or bluish in colour, having juicy, sweet pulp and a small stone. The keeping quality of fruit is very poor so the fruit is to be utilized within 24 h after picking (Shukla et al., 1991). Jamun fruit is one of those fruits that contain variety of important nutritional and medicinal elements. The fruit syrup is very useful for curing diarrhea. It is carminative and diuretic, apart from having cooling and digestive properties. It also contains Anthocyanins, flavenoids and other useful ingredients.
The aims of the current investigation were to evaluate antioxidant efficiency of crud juice and crude extracts of two varieties of pomposia by using β-carotene bleaching and free radical scavenging (DPPH) methods in comparison with BHT as synthetic antioxidant, evaluating the effect of adding various levels of the highest antioxidant efficiency of pomposia extracts on the quality of ground beef by determination the changes in TBA value during refrigerated storage compared to BHT and to evaluate the effect of adding different levels of pomposia extract on sensory properties of cooked ground beef at the end of storage period.
MATERIALS AND METHODS
Materials: Two varieties of ripened and freshly harvested pomposia, (Syzyygium cumini) season, July 2008, the first one Rajamun is big oblong, deep purple in colour having pink to grey juice, sweet flesh with small seeds. The second one is known as Kaatha which has small fruits with comparatively big seed and flesh acidic in taste were obtained from the farm of Faculty of Agriculture, Cairo University, Giza, Egypt.
The fresh beef (Flank) was purchased from a local market, Giza, Egypt. The fat content of the beef was 15.5%.
Chemicals: 2-Thiobarbituric Acid (TBA), 1,1-Diphenyl-2-picrylhydrazyl (DPPH), polyoxy-ethyene-sorbitanmonolaurate (Tween 20), β-carotene β-carotene and linoleic acid were purchased from Sigma Chemical Co. (St. Louis, MO, USA). All chemical used were of analytical reagent grade.
Preparation of crud juice and extracts of pomposia (Syzygium cumini): Pomposia fruit Rajamun and Kaatha varieties were cleaned, cut into pieces and then pressed by means of the hydraulic laboratory press model C S/N 37000-156 Freds from Carver (WI, USA). The resultant crude juices were centrifuged at 4000 rpm for 30 min, the supernatants were lyophilized using a freeze-dryer (Labconco Corporation Kansas city, Missouri 64132 USA). The concentrated juice was kept in a brown bottle at-18°C until further use.
Ethanolic and aqueous extracts of Pomposia fruit were prepared according to the method described by Sanbongi et al. (1998). The cleaned pieces of Pomposia fruits were diluted with 70% ethanol (1: 7 v/v). The mixture was stirred with magnetic stirrer for 2 h. at ambient temperature. The resultant mixture was left overnight and filtered through a Whatman No. 4 filter paper (Whatman International Ltd., Maidstone, UK), the remaining residue was re-extracted as described above. The filtrate was concentrated using a rotary evaporator (Buchi, Switzerland) at 40°C under pressure to remove ethanol residues. In addition, distilled water was used instead of ethanol to prepare aqueous extracts of Pomposia fruit. The ethanolic and aqueous extracts were lyophilized using a freeze-dryer (Labconco corporation Kansas city, Missouri 64132 USA). The concentrated juice was kept in a brown bottle at-18°Cuntil further use.
Preparation of ground beef: Beef meat (5 kg) was cut into small pieces and homogenized in stainless steel blender (Univest Co., 6th of October city, Industrial zone No. 3, Giza, Egypt). Ground beef were mixed by latex gloved hands with 0.5, 0.75 and 1% of either the highest antioxidant efficiency of pomposia extracts and BHT. Minced beef without additives was run as control. The abovementioned samples were packed in polyethylene bags, each bag contain 250 g and stored at 0± 0.5°C in refrigerator for 10 days.
Analytical methods
Determination of moisture content: A known weight of ground beef (5 g) was dried in an oven at 105°C until a constant weight was reached (AOAC, 2000).
Determination of ash content: The ash content of ground beef was determined by muffle furnace at 550°C until a constant weight was obtained (AOAC, 2000).
Determination of crude protein: The total nitrogen of ground beef was determined by the usual Kjeldahl method (AOAC, 2000). The crude protein was then calculated by multiplying the total nitrogen by a factor of 6.25.
Determination of total crude lipids: The crude lipids of ground beef was determined according method of Bligh and Dyer (1959) using a mixture of chloroform-methanol (2: 1, v/v).
Nitrogen Free Extract (NFE): The Nitrogen Free Extract (NFE) was calculated by difference.
Determination of total polyphenols: Total polyphenols were determined according to the method of Jayaprakasha et al. (2003). An aliquot from pomposia juice and extracts (0.1 mL) were dissolved in a 10 mL mixture of acetone and water (6:4 v/v). Sample (0.2 mL) was mixed with 1.0 mL of ten-fold diluted Folin-Ciocalteu reagent and 0.8 mL of 75 g L-1 sodium carbonate solution. After standing for 30 min at room temperature, the absorbance was measured at 725 nm. Phenolic contents were calculated on the basis of the standard curve for Gallic Acid (GAL).The results were expressed as mg of gallic acid equivalent per g of dry extract.
Determination of free radical scavenging activity (DPPH): The antioxidant activity of crude juice and extracts of pomposia were evaluated by using the 2, 2'diphenylpicrylhydrazyl (DPPH) assay (Cuendet et al., 1997; Burits and Bucar, 2000). Fifty microliters of the extracts were added to 5 ml of a 0.004% (w/v) of DPPH in methanol (100% v/v). After, a 30 min incubation period at room temperature the absorbance at 517 nm was compared to DPPH in methanol without an extract sample (blank). The percent inhibition of free radical formation (I%) was calculated as:
Determination of antioxidant activity: The antioxidant activity of crude juice and extracts of pomposia were by using β-carotene bleaching method, according to the method described by (Jayaprakasha et al., 2001). One milliliter of β-carotene solution (0.2 mg β-carotene mL-1 of chloroform) was transferred into a round-bottom flask containing 0.02 mL of linoleic acid and 0.2 mL of Tween 20. The Chloroform was evaporated at 40°C using rotary evaporator. The resultant mixture was immediately diluted with 100 mL of deionized water and mixed for 2 min to form emulsion. A mixture prepared similarly without β-carotene was used as a blank. A control, containing 0.2 mL of 70% ethanol instead of extract was also prepared. Five milliliter of the emulsion was transferred into test tube containing 0.2 mL of extract. The tubes were placed at 50°C in water bath for 2 h. The absorbance was recorded at 470 nm using UV-Visible spectrophotometer ((Shimadzu, Kyoto, Japan).
The percentage antioxidant activity was calculated based on the following equation:
where, A0 and A°0 are the absorbance values measured at zero time of incubation for extracts and control, respectively.
At and A°t are the absorbance values for extracts and control at t = 120 min.
Thiobarbituric Acid-reactive Substances (TBARS): The TBARS values were determined in triplicate samples by the extraction method of Mielnik et al. (2006). For extraction, 10 g meat was homogenized with 30 mL of a 7.5% aqueous solution of trichloroacetic acid (TCA). After filtration (vacuum filtered through a Whatman No. 41 filter paper (Whatman International Ltd., Maidstone, UK), 5.0 mL of extract was mixed with 5.0 mL of 0.02 mol L-1 aqueous Thiobarbituric acid (TBA) in a stoppered test tube. The samples were incubated at 100°C for 35 min in a water-bath and subsequently cooled for 10 min in cold water. Absorbance was measured at 532 nm by using UV-Visible spectrophotometer (Shimadzu, Kyoto, Japan). against a blank containing 5 mL distilled water and 5 mL TBA reagent. Results expressed as milligrams malondialdehyde kg-1 meat.
Cooking and sensory evaluation: Minced beef samples were mixed with 1% salt, pressed into a mould (8 cm diameter, 0.5 height) and cooked for 30 min in water bath at 75°C until an internal temperature of 75°C was reached. Cooked beef samples were left to be warm then served for sensory evaluation. Sensory evaluation method was conducted according to the method described by Mansour and Khalil (2000), cooked beef samples were served warm to 10-membered trained panel (Staff of Food Technology Research Institute, Agriculture Research Center, Giza, Egypt) and to 40 of nonspecialist panelists randomly recruited within the student and lecturers of Biochemistry Department, Faculty of Agriculture, Cairo University, without care of age or sex. The panelists were subjected to sensory evaluation using a 9-point hedonic scale for taste, color, odor and overall-acceptability. A numerical basis as a sort of evaluation from 1 to 9 was used where (1 = dislike extremely, 2 = dislike very much, 3 = dislike moderately, 4 = dislike slightly, 5 = neither like nor dislike, 6 = like slightly, 7 = like moderately, 8 = like very much, 9 = like extremely).
Statistical analysis: The data of the present study were subjected to analysis of variance and the least significant difference test, in order to compare the mean values of the investigated parameters.
Pomposia polyphenols: Jamun fruit is one of those fruits that contain variety of important nutritional and medicinal elements. It contains Anthocyanins, flavenoids and other useful ingredients. Crude juice and ethanolic extract of Kaatha variety had significantly (p<0.01) the highest levels of total polyphenols were 16.32 and 15.45 mg g-1, respectively, whereas aqueous extract had significantly (p<0.05) the lowest values of total polyphenols were 5.42 and 6.66 for the two varieties of pomposia Rajamun and Kaatha, respectively (Table 1).
Antioxidant activity and free radical scavenging activity (DPPH): Antioxidant tests could be based on the evaluation of lipid peroxidation or on the measurement of free radical scavenging potency (hydrogen-donating ability). The radical scavengers donate hydrogen to free radicals, leading to non toxic species and therefore to inhibition of the propagation phase of lipid oxidation. The use of DPPH radical provides an easy, rapid and convenient method to evaluate the antioxidants and radical scavengers (Soler-Rivas et al., 2000; Kansci et al., 2003; Argolo et al., 2004; Roginski and Lissi, 2005). Therefore, in the present investigation, pomposia juice and extracts were evaluated for their antioxidant activity and radical scavenging activity by β-carotene bleaching and DPPH techniques. Crude juice and ethanolic extract of Kaatha variety had significantly (p<0.05) the highest antioxidant activity were 90.00 and 89.00%, respectively, however, the lowest antioxidant activities was shown by aqueous extract of Rajamun was 62.00% (Table 1). One could report that there is high relationship between the content of total polyphenols of extract and its antioxidant activity, hence the content of total polyphenol of extract is increased as antioxidant activity of extract increased. The most effective radical scavenging activity was shown by crude juice of Kaatha was 85.22% while the least effective was the aqueous extract of Rajamun. Anyway, crude juice and ethanolic extracts of pomposia showed relatively high value of radical scavenging activity ranged from 67.67 to 85.22% (Table 1). This high antioxidant capacity may be due to the high concentration of phenolics in pomposia extracts. These compounds are an important group of natural antioxidants with possible beneficial effects on human health (Meyers et al., 2003). They can participate in protection against the harmful action of reactive oxygen species, mainly oxygen free radicals. These compounds are known also to possess antioxidant activity due to its ability to reduce free radical stability via electron or hydrogen-donating mechanism. (Lee et al., 1999; Ruberto and Baratta, 2000).
| Table 1: | Polyphenolic content, antioxidant and free radical scavenging activities of pomposia juice and extracts |
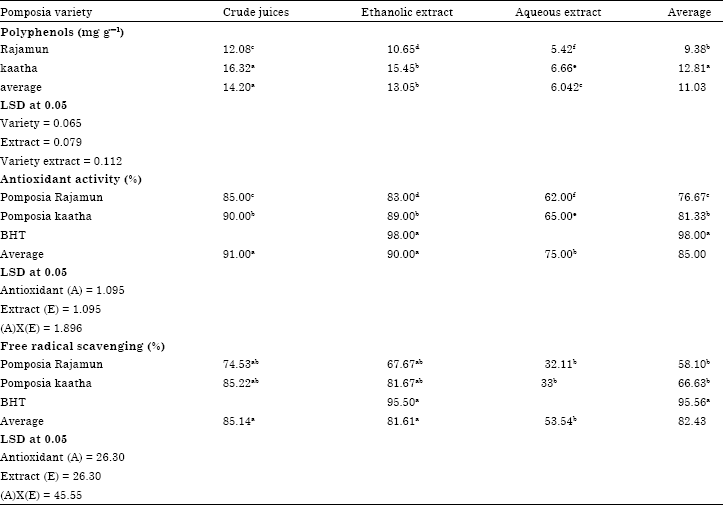 | |
Values are expressed as the mean of three determinations. Values followed by different letter are significantly different at (p<0.05). LSD refers to least significant difference test | |
The antioxidants activity of polyphenols is principally based on the redox properties of their hydroxyl groups and the structural relationships between different parts of their chemical structure (Rice-Evans et al., 1996).
Chemical composition of ground beef: The moisture content of ground beef was 65.39%. The protein content, crude fat and ash content of ground beef were 51.91, 37.71 and 3.06%, respectively. The Nitrogen Free Extract (NFE) was 7.33%. The obtained results indicate that beef contain relatively high levels of fat which consider a major cause of quality deterioration in meat and its cooked products (Table 2). These results are in good agreement with those obtained by Ismail and Yee (2006) and Abd El-Hamied et al. (2009).
Changes of TBA value of ground beef during refrigerated storage: Ground meat tends to become rancid and brown more rapidly, due to pigment and lipid oxidation. An oxidative reaction in muscle foods leads to degradation of lipid and proteins, resulting in deterioration of flavour, texture and nutritive value and is considered as one of the major problems in the development of new convenient meat products and processes (Gray and Pearson, 1987). In the present investigation based on the antioxidant and free radical scavenging activities results, the highest antioxidant and free radical scavenging activities was shown by the crude juice of Kaatha, therefore 0.5, 0.75 and 1% of either crude juice of Kaatha and BHT. were added to minced meat to evaluate it's effects on the lipid peroxidation of ground beef during storage process.
| Table 2: | Chemical composition of minced beef (based on dry weight basis) |
 | |
Values are expressed as the mean of three determinations. Values followed by different letter are significantly different at (p<0.05). LSD refers to least significant difference test | |
Generally, TBA values increased gradually and significantly (p<0.05) during storage period. The phospholipids in muscle membrane provide an ideal substrate for lipid peroxidation. Iron bound to negatively charged phospholipids promotes lipid peroxidation, resulting in generation of warmed-over flavor (Empson et al., 1991). Both nonheme and heme iron in beef muscle can catalyze lipid peroxidation (Kanner et al., 1988; Love, 1983; Monahan et al., 1993). However, mixing minced beef with various levels of crude juice of Kaatha caused a significant (p = 0.05) reduction of TBA values compared to control sample, These inhibitory effects of crude juice of Kaatha were dose-dependent. Control samples had significantly (p = 0.05) the highest TBA value was 1.98 mg malondialdehyde (MDA) kg-1 at the end of the storage period, the highest TBARS values of the control sample at the end of storage days might be due to an interaction between the natural substances (for example, polyunsaturated fatty acids) and catalysts (for example, iron ion) from the meat tissue during storage (Decker and Hutlin, 1990; Kim et al., 2000), while beef samples mixed with 1% of pomposia juice had significantly (p = 0.05) the lowest TBA values was 0.79 mg malondialdehyde (MDA) kg-1 at the end of the storage period. No significant (p>0.05) differences were observed in TBA values of ground beef mixed with 0.75% crude juice of Kaatha and that sample treated with 1.00% of synthetic antioxidant BHT. TBARS numbers in treated samples were much lower than the control sample. The inhibitory effect of pomposia juice on lipid oxidation might be due to scavenging of free radicals and chelating of transition metals (Chan et al., 1994; Lee and Hendricks, 1997; Niki, 1991). The amounts of TBARS as mg MDA kg-1 meat formed at the end of storage period for control and treated samples were far below the critical value of 3 mg kg-1 at which rancidity is detected (Wong et al., 1995), while Greene and Cumuze (1982) considered a TBARS range 0.6-2.0 mg kg-1 to be the minimum detectable level for oxidized flavour in ground beef. The obtained results indicate high antioxidative effect of polyphenolic compounds present in pomposia extract in reducing the formation of hydroperoxides during storage process (Table 3) These results are in good agreement with those obtained by Ismail and Yee (2006) who studied the effects of extracts of cocoa shell, Roselle seeds and a combination of them on the lipid oxidation of cooked beef and they reported that the tested extracts were more effective than BHT and tocopherols in inhibiting lipid oxidation of cooked-refrigerated beef and Azizah et al. (1999), who found that phenolic compounds of cocoa shells exhibited a strong antioxidant activity compared to synthetic antioxidant.
In this respect the results of Abd El-Hamied et al. (2009) observed that the addition of rosemary, sage and their combination showed high antioxidative effects during refrigerated and frozen storage of minced meat also Ali (2010) showed that mixing oil with various levels of pomposia extracts caused significant decrease of the formation of secondary products during frying process.
Sensory evaluation: Sensory characteristics of cooked minced beef as affected by adding different levels of pomposia juice and BHT are presented in Table 4. Treated sample had relatively high value of taste, odor, colour and overall acceptability compared to control sample. No significant (p>0.05) differences were observed in taste between samples mixed with 0.5 and 0.75% of crude juice of Kaatha and those mixed with 0.05% of BHT The addition of pomposia juice affected significantly (p<0.05) colour parameter of the final product, minced beef mixed with 1.00% of pomposia juice had significantly (p = 0.05) the highest value of colour was 9.00 whereas control samples had significantly (p = 0.05) the lowest value was 5.00.
Odor also is one of the most important quality factors of acceptance for cooked meat products. The results presented in Table 4 show that samples mixed with 0.5 of crude juice of Kaatha or BHT had significantly (p = 0.05) the highest value of odor was 9.00 and 8.94, respectively whereas control samples and those samples mixed with 1.00% of BHT had significantly (p = 0.05) the lowest odor values of were 7.50 and 7.44, respectively. All samples showed good overall acceptability; in all cases the values were higher than 6. However samples mixed with 0.75 and 1.00 of crude juice of Kaatha had significantly (p = 0.05) the highest scores of over all acceptability were 8.59 and 8.48, respectively. The results of sensory evaluation suggests that the crude juice of pomposia (Kaatha) can be successfully used as natural antioxidant and colourant.
| Table 3: | Effect of crude juice of Kaatha and BHT on TBA No. (mg MDA kg-1 meat) of minced beef during refrigerated storage at 0°C |
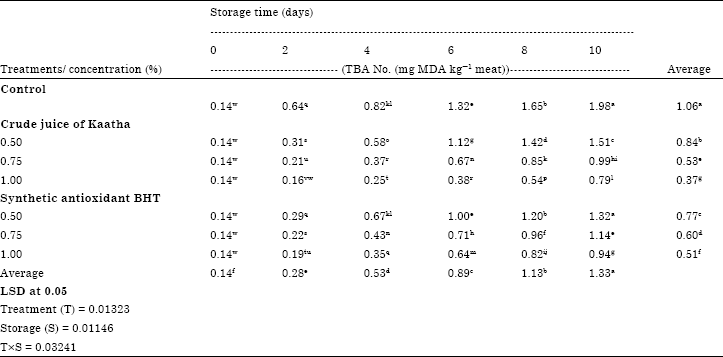 | |
Values are expressed as the mean of three determinations. Values followed by different letter are significantly different at (p<0.05). LSD refers to least significant difference test | |
| Table 4: | Sensory evaluation of cooked minced beef mixed with different levels of crude juice of pomposia and BHT |
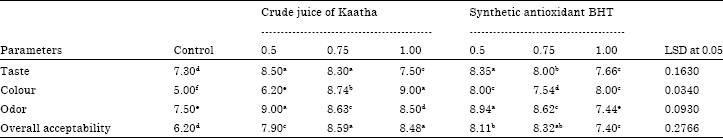 | |
Values are expressed as the mean of three determinations. Values followed by different letter are significantly different at (p<0.05). LSD refers to least significant difference test | |
The obtained results showed that the most effective radical scavenging activity was shown by crude juice of Kaatha was 85.22% while the least effective was the aqueous extract of Rajamun. Anyway, crude juice and ethanolic extracts of pomposia showed relatively high value of radical scavenging activity ranged from 67.67 to 85.22%. polyphenolic compounds present in pomposia juice and ethanolic extract of pomposia (Kaatha) had high antioxidative effect in reducing the formation of hydroperoxides during refrigerated storage, in the same time the results of sensory evaluation suggests that the crude juice of pomposia (Kaatha) can be successfully used as natural antioxidant and colourant for meat products.