Research Article
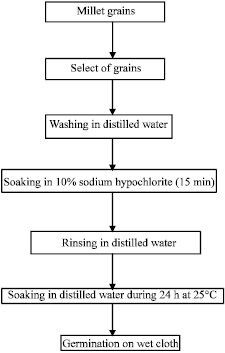
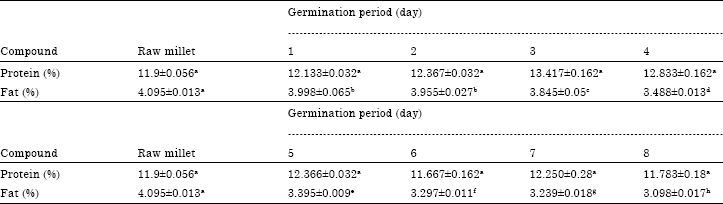
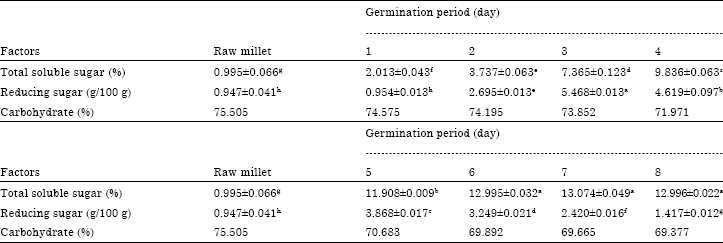
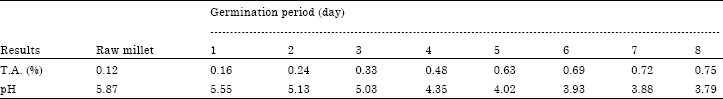
Evolution of Energetic Compounds, Antioxidant Capacity, Some Vitamins and Minerals, Phytase and Amylase Activity During the Germination of Foxtail Millet
State Key Laboratory of Food Science and Technology, Jiangnan University, Wuxi, Jiangsu 214122, People`s Republic of China
Jie Chen
State Key Laboratory of Food Science and Technology, Jiangnan University, Wuxi, Jiangsu 214122, People`s Republic of China