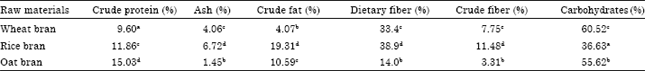
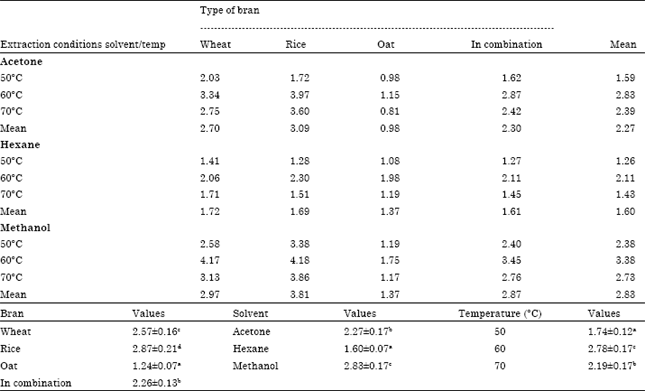
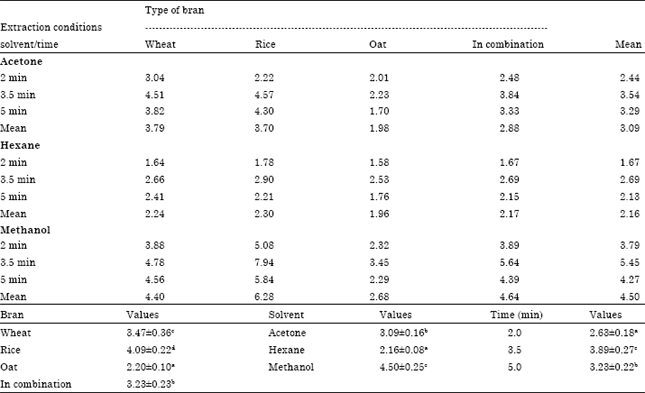
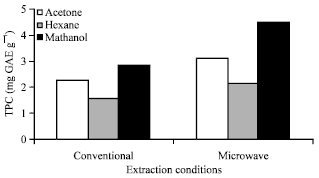
Research Article
Total Phenolic Content of Cereal Brans using Conventional and Microwave Assisted Extraction
Department of Food Science and Technology, Punjab Agricultural University, Ludhiana-141 004, India
Savita Sharma
Department of Food Science and Technology, Punjab Agricultural University, Ludhiana-141 004, India