Research Article
Development and Nutritional Quality Evaluation of Kersting’s Groundnut-Ogi for African Weaning Diet
Department of Chemistry, Nasarawa State University, PMB 1022, Keffi, Nigeria
LiveDNA: 234.1611
ORCID: 0000-0003-1115-7874
B.G. Osinfade
Department of Science Laboratory Technology, Federal Polytechnic, Ilaro, Ogun State, Nigeria
S.K. Basu
Department of Biological Sciences, University of Lethbridge, Lethbridge, Alberta, TIK 3M4, Canada
B.E. Ablaku
Department of Chemistry, Nasarawa State University, PMB 1022, Keffi, Nigeria









Adazebra Gloria Reply
it is an interesting piece of information, but i will like to know the other nutrients that can be found in kersting groundnut.
Matthew Olaleke AREMU
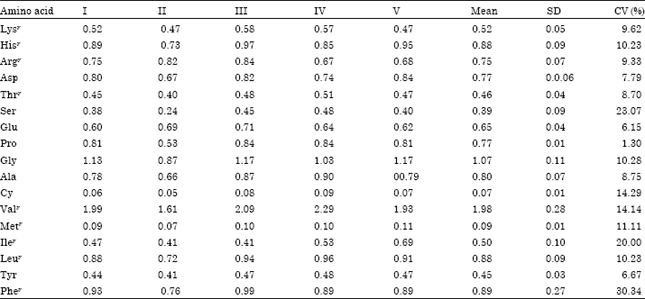
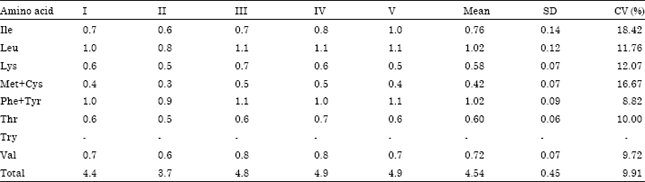
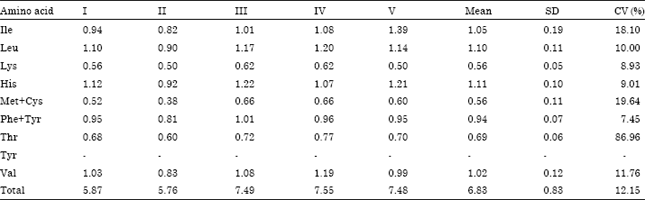
Fatty acid composition of kersting groundnut seed oils was determined. Essential fatty acids(oleic and linoleic) were found to be adequate.