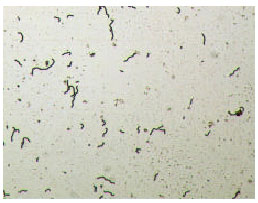
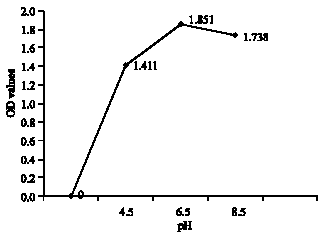
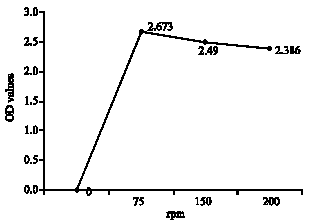
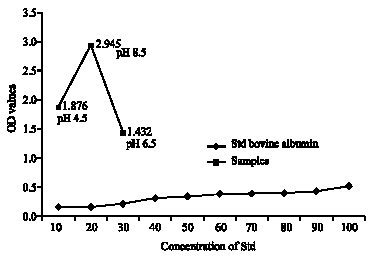
Research Article
Effect of Lactobacillus lactis cremoris Isolated from Kefir against Food Spoilage Bacteria
P.G. Department of Microbiology, Jamal Mohamed College, Trichy, Tamilnadu, India
P. Gajalakshmi
P.G. Department of Microbiology, Dhanalakshmi Srinivasan College of Arts and Science for Women, Perambalure, Tamilnadu, India
M. Mohamed Mahroop Raja
P.G. Department of Microbiology, Jamal Mohamed College, Trichy, Tamilnadu, India
M. Mohamed Imran
P.G. Department of Microbiology, Jamal Mohamed College, Trichy, Tamilnadu, India











karthikayen Reply
dear sir,
i reffered this paper that you published a good work and i wondered how the single strain that control the other bacteria. can you suggest is it possible to control contamination of microbes in food industries.