Research Article
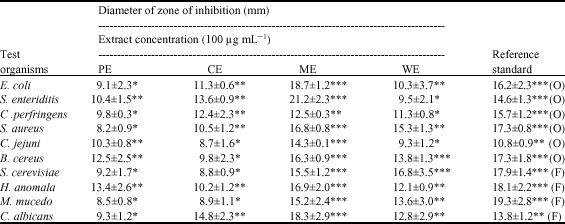
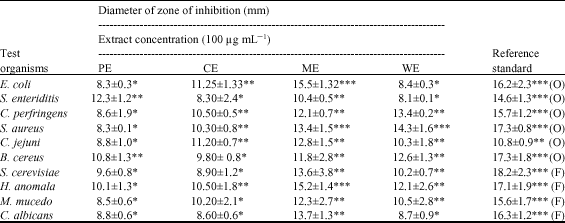
In vitro Antimicrobial Evaluation of Zingiber officinale, Curcuma longa and Alpinia galanga Extracts as Natural Food Preservatives
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia
LiveDNA: 91.1965
ORCID: 0000-0001-8194-3470
R. Suraj
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia
G. Rejitha
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia
K. Anandarajagopal
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia
A.V. Anita Gnana Kumari
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia
P. Promwichit
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia