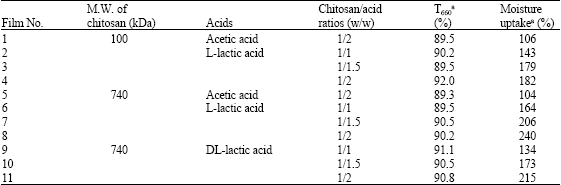
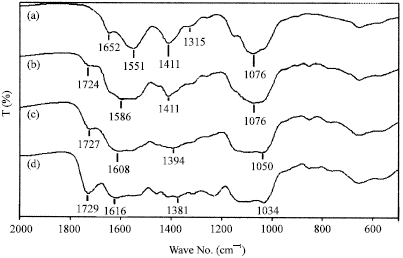
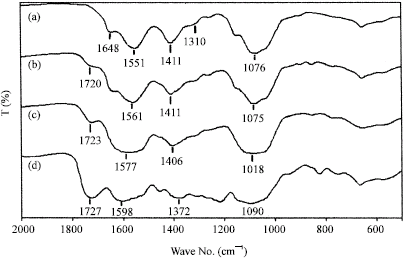
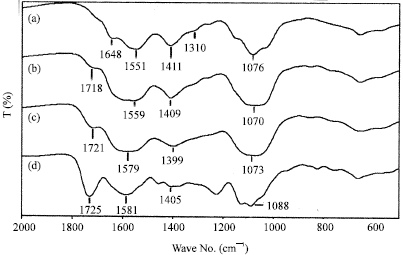
Research Article
Preparation and Characterization of Highly Flexible Chitosan Films for Use as Food Packaging
Center of Excellence for Innovation in Chemistry, Department of Chemistry, Faculty of Science, Mahasarakham University, Mahasarakham 44150, Thailand
Y. Baimark
Center of Excellence for Innovation in Chemistry, Department of Chemistry, Faculty of Science, Mahasarakham University, Mahasarakham 44150, Thailand