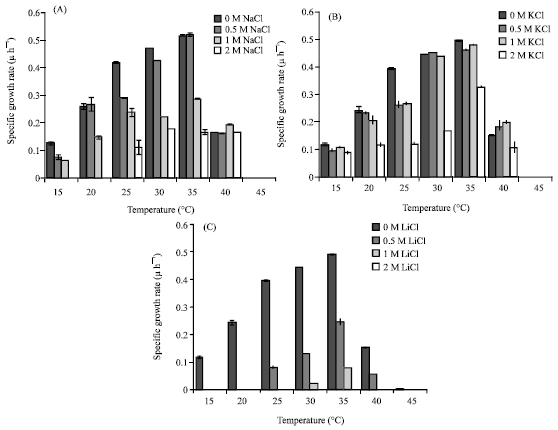
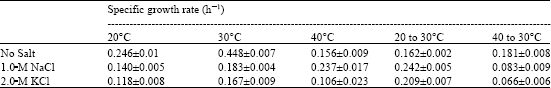
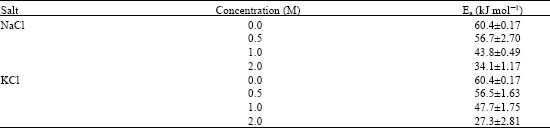
Research Article
Effects of Temperature and Salts on Growth of Halotolerant Debaryomyces nepalensis NCYC 3413
Applied and Industrial Microbiology Laboratory, Department of Biotechnology, Indian Institute of Technology-Madras, Chennai 600 036, India
LiveDNA: 91.30544
Sawan Kumar
Applied and Industrial Microbiology Laboratory, Department of Biotechnology, Indian Institute of Technology-Madras, Chennai 600 036, India