Research Article
Spring Water Hydrochemistry along the North-South Profile in the Jordan Valley
Bir Zeit University, P.O. Box 14, Ramallah, Palestine
S. Ghannam
Bir Zeit University, P.O. Box 14, Ramallah, Palestine
The hydrochemical parameters are the main indicators of water quality for the springs. The hydrochemistry of the springs will indicate to which extent the spring water is suitable for drinking or agricultural purposes. Springs played a major role for localizing the majority of the Palestinian villages according to their water needs. Water springs were considered as the major source for drinking purposes in the rural Palestinian communities throughout the previous decades. The outcropping in the Eastern side of the Jordan Valley is sedimentary rocks composed of limestone, dolomite and marly limestone rock, while the others are composed of evaporates of Lisan formations. There are few studies about the hydrochemistry of the springs in the West bank. Hydrochemistry of Faria drainage basin/West bank was studied by Ghanem (1999) and revealed that the Faria springs were found to be of Calcium Bicarbonate type and not polluted. The hydrogeology and hydrochemistry of springs and groundwater dug wells of Hebron District were studied by Awadallah and Owaiwi (2005). The wide distribution of cesspools and septic tanks in the West bank cause rapid contamination of aquifer systems through karstic conduits in the area. Abadi (2006) assessed the water resources in Al Auja spring area in terms of rainfall-runoff relation, base flow and recession for the major springs and evaluated the sustainable yield of the lower aquifer of Ein Samia well field. Hydrogeological and hydrochemical studies were conducted for the North Western Auja Tamaseeh basin (Tulkarm area) by Samahan (2007) and represented a highly sensitive area for pollution and the study was used as an assessment tool for the evaluation of the human activities on groundwater quality. Springs in Natuf drainage basin are of good water quality, except those near populated areas such as Ein Musbah, Al Alaq and Ein Ayoub springs which have low water quality for agricultural and domestic uses based on SAR, SSP and EC values and considered as microbially contaminated (Shalash and Ghanem, 2008). The anthropogenic nitrate as well as the dissolved organic carbon in the springs water are with spatially and temporally variations. The chemical composition of the springs’ water as well as stable isotopes δ18O and δ2H was studied by Abu Hilou (2008). Water quality study of Al Qilt springs showed moderate levels of major ions and higher concentrations of lead and cadmium than allowable concentrations for drinking water in three spring samples, whereas, some springs were microbial contaminated, which indicate the infiltration of pollutants in the recharge area of the springs (Daghrah, 2009). This study analyzed the hydrochemical characteristics and water quality of twenty springs distributed along the North-South profile of the Jordan Rift Valley. The main objective of this research is to determine the pollution level of these springs in reference to physical and chemical parameters as well as the trends of the hydrochemical variability of the North-South directions. The water quality of these springs was determined through measuring the concentration of the major and minor ion concentrations. Their suitability for irrigation purposes and the anthropogenic pollution of these springs were also evaluated.
The sampling campaigns was carried out in wet seasons, starting from Nov-2008 to January 2009; a total of 50 samples were collected. The water samples were collected in one-liter p olyethylene bottles and refrigerated in the laboratory at 2°C. A Sharewood 4010 flame photometer was used to determine calcium, sodium and potassium. A HP 8453 Diode Array Spectrophotometer was used to determine nitrate and sulfate concentrations. A Metrohm 716 titrator used to determine chloride and bicarbonate concentrations. Concentration of trace elements such as iron, copper, zinc, cadmium, lead and arsenic will be determined using Graphite furnace Atomic Spectrophotometer.
Study Area
The study area lies within the Eastern flank of the West Bank Aquifers. Its groundwater generally flows towards the Jordan Valley. The available potential resource of this Basin is estimated to be 1 50 MCM/year (Tahal Consulting Engineers Ltd., 1990) supporting over 79 large flowing springs of an average discharge greater than 0.1 L sec-1. These springs constitute around 90% of the total annual spring discharge in the West bank. Sixteen of the springs are used for domestic and agricultural purposes. The study area consists of 8 surface catchments including: Malih Shubash (4 springs), Malih (2 springs), Faria catchment (8 springs), Al-Ahmer (1 spring), Al Auja (1 spring), Nuweima (3 springs) and Qilt (1 spring), (Fig. 1).
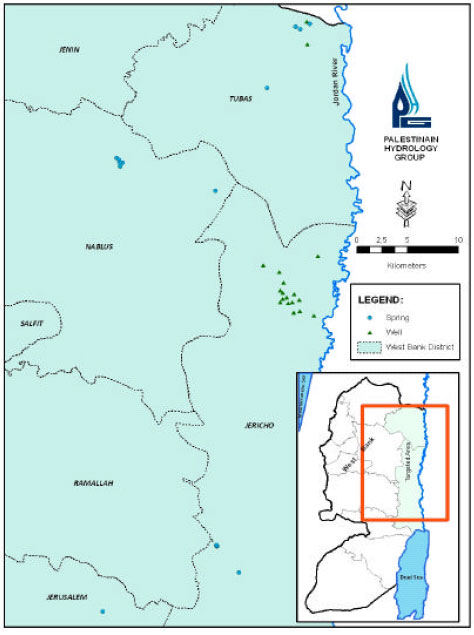 | |
| Fig. 1: | Location area with the sampled spring sites |
RESULTS
Fifty water spring samples were analyzed for their hydrochemical parameters of Ca2+, Mg2+, Na+, K+, Cl¯, SO42¯, NO3¯ and HCO3¯ as well as for heavy metals of Fe, Zn, Li, Cr, Co, Ni, Cu, Cu, Mn, Al, Cd, Ba, Pb and Bi in order to understand the hydrochemical characteristics of the water springs as well as to understand their variability trends in the north-south and east-west directions. Three types of water springs were found: Na-Cl, Na-HCO3 and Ca-HCO3. Piper diagrams were plotted for all the water samples analyzed in terms of major ions using AquaChem 5.1 software. It is clear from the graph that most of the samples fall into the normal earth alkaline water group with prevailing bicarbonate and sulfate or chloride (Fig. 2). Samples taken from Al Jesir and Shosah springs are classified under normal earth alkaline water with prevailing bicarbonate.
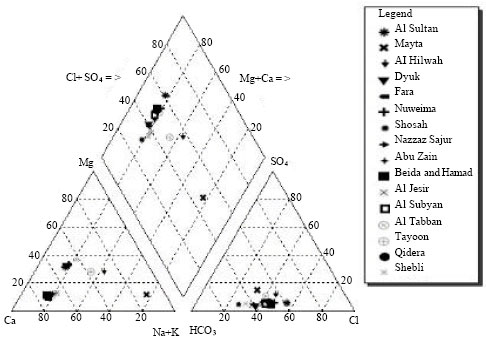 | |
| Fig. 2: | Piper diagram for the springs |
The sample taken from Al Hilwah spring is earth alkaline water with increased portions of alkalis (prevailing sulfate and chloride), this water type is characterized by its high TDS and Cl¯ which may be an indication of sever contamination with wastewater derived from the cesspits in the nearby villages. The sample taken from Al Sultan spring was normal earth alkaline water with prevailing sulfate while the one taken from Mayta spring was alkaline water with prevailing bicarbonate. However, that from Tayoon spring was normal earth alkaline water with prevailing bicarbonate. This type of water originates through natural processes by the dissolution of carbon dioxide (CO2) from the atmosphere and from the soil horizon which causes the dissolution of the carbonate minerals, calcite CaCO3 and dolomite (Ca, Mg) (CO3)2 of the aquifer (Suk and Lee, 1999).
The Durov diagram is used to display some possible geochemical processes that could affect the water genesis (Domenico and Schwards 1990) (Fig. 3). Samples from Mayta spring corresponds to Field 6, Shosah spring lies in Field 2, those from Tayoon, Nuweima, Al Sultan, Dyuk, Al Hilwah and Shebli springs lie within the Field 5. The plotted water spring samples in Durov Diagram agree with their corresponding water types of the springs.
The water of the spring samples has the following ionic strengths for major ions:
The analyzed parameters were subjected to product linear correlation analysis (Mendenhall et al., 2003). Correlation coefficients were calculated for all possible pairs of variables. These numerically express the extent to which two variables are perfectly correlated, a value of 0 indicates that the variables have no correlation at all (i.e., they are completely independent) and a value of -1 indicates that they are perfectly negatively correlated (an increase in one corresponds to a decrease in the other). A good correlation in the spring water analyses were found between Fe and Al with all cations as well as SO4 and Cl from the anions in addition to the Zn, Mn, Bi.
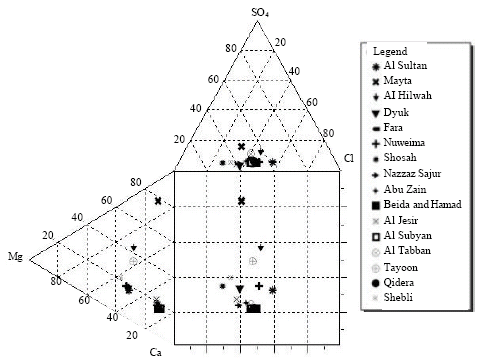 | |
| Fig. 3: | Durov diagram for the spring samples |
Ca and Mg show good correlation to Bi, Al, Zn. Cr shows a negative correlation to Na and positive correlation to Co and Ni. Barium shows a positive correlation to Mn and negative correlation to Cr. In the well matrix correlation, the major cations show good correlation to Al and Ba. The following correlation pairs were found: (Fe to Mn and Pb), (Cr to Na and Mg), (Zn to Ni and Al), (Ni to Li), (SO4 to Ba) and (Bi to Al). T he quality of the recharge water and its interaction with soil and rocks during its percolation and its storage in the aquifers are key factors in the chemistry of groundwater (Todd, 1980). These interactions involve mainly dissolution and precipitation processes, which are controlled by the solubility products of the different involved mineral phases. One of the procedures to determine the equilibrium state of water with a mineral phase in the matrix of the aquifer is by calculating the saturation index. The results of the saturation index show that all the spring water samples were found to be undersaturated with respect to all the minerals. These relationships may be comparatively simple and uncomplicated as in the case of an aquifer that receives direct recharge by rainfall and from which water is discharged without contacting any other aquifer or other water.
Water Quality Evaluation
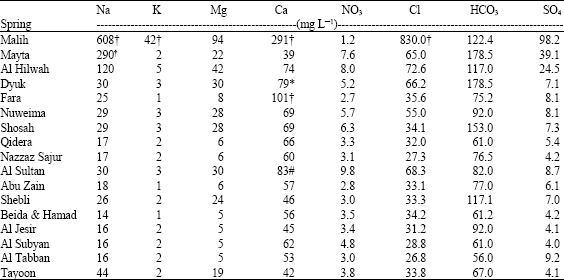
Accordingly most of the samples collected from the springs are considered to be of fresh water type with the exception of that taken from Al Maleh which is brackish. Calcium concentration in the majority of the springs are in the range that reflect the limestone origin, with the exception of the northern springs that reflects the Na-Cl type (Lloyd, 1999). From the results it is noticeable that high concentrations of Mg2+ usually accompany high concentrations of Ca2+ which may be an indication of dolomitization, especially for the northern springs. The low Mg2+ concentrations reflect the carbonate origin. All sampled springs are within the WHO range with the exception of the Malih spring, where an increase reaching a maximum limit of 608 mg L-1 and chloride concentration of 830 mg L-1 and has the Na-Cl type. The Na concentrations of the sampled wells are higher than the WHO standard which reflects the Na-Cl type. The K concentrations are lower than its limit, with the exception of the northern springs and the sampled groundwater wells. The results show that the sulfate concentrations are under the range of the WHO limits. No health-based guideline value is proposed for chloride in drinking water. However, chloride concentrations in excess of about 250 mg L-1 can rise to detectable taste in water. The chloride concentrations of the sampled springs are within the WHO range, with the exception to Malih spring. The HCO3 concentration results in the sampled springs reflect the Ca-HCO3 water type. The nitrate results of the water samples are within the range of the WHO limits. The results of the trace elements show that all springs, with the exception of Malih springs have a concentration within the WHO standards with the exception of Pb and partially Al. Table 1 shows the average hydrochemical parameters of the analyzed water spring samples highlighting those that exceed different water quality standards.
By comparing the results with the hardness classification, all the water samples range from being hard to very hard with the exception of that taken from Al Jesir which is moderately hard.
Trends
Increasing concentration trends of the North-South profile are shown for Mg, Ca, Na, K, HCO3, SO4, Pb and Bi; while a decreasing trend is shown for the parameters of NO3, Cl, Ba and Al. The southern increasing trends for Na and HCO3 and the northern increasing trends of Pb, Zn, Ba and NO3 are shown in Fig. 4a-f.
| Table 1: | Major ion concentrations compared to standards of the sampled springs |
 | |
| *: Exceeding palestinian standards, #: Exceeding WHO standards _: Exceeding both palestinian and WHO standards | |
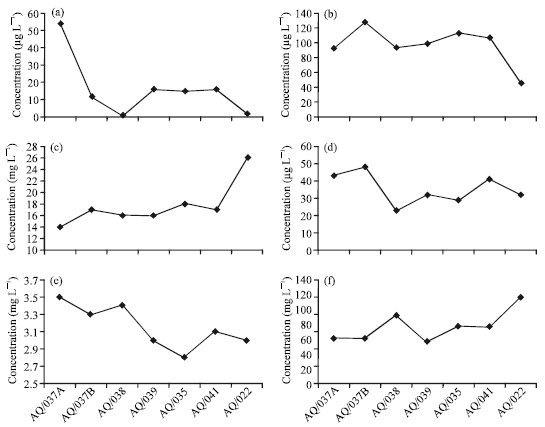 | |
| Fig. 4: | N-S trends of major ions and trace elements; (a) Lead N-S trend, (b) Barium N-S trend, (c) Sodium N-S trend, (d) Zinc N-S trend, (e) Nitrate N-S trend and (f) Bicarbonate N-S trend |
The hydrochemical characteristics of twenty springs, distributed North-South in the Upper Jordan Rift Valley indicate that the increasing concentration trends of Na+, SO42-, NO3-, as well as of Fe, Co, Bi towards the south is due to natural and manmade pollution sources. The industrial wastewater discharge in the study area increases the concentrations of Co, Bi, Li and threatens the quality of the groundwater. The majority of the spring water samples fall into the normal earth alkaline water group with prevailing bicarbonate and sulfate or chloride. The Southern springs show high TDS and are of Na-Cl water type.