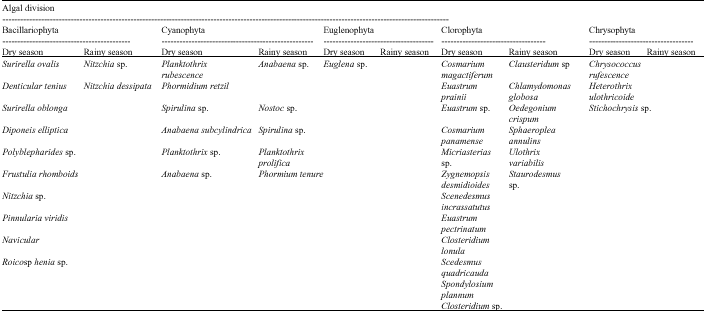
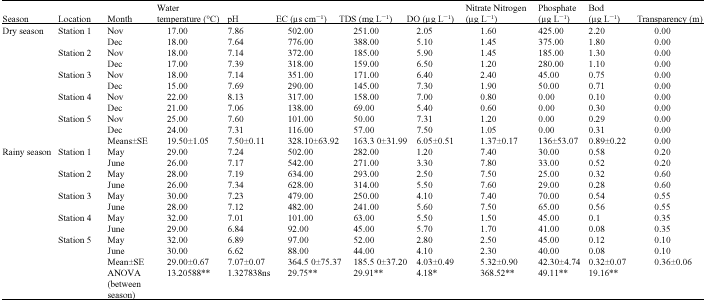
Research Article
Seasonal Occurrence of Algae and Physicochemical Parameters of Samaru Stream, Zaria, Nigeria
Department of Biological Sciences, Ahmadu Bello University (ABU) Zaria, Nigeria
Y. Tanimu
Department of Biological Sciences, Ahmadu Bello University (ABU) Zaria, Nigeria
A.M. Chia
Department of Biological Sciences, Ahmadu Bello University (ABU) Zaria, Nigeria