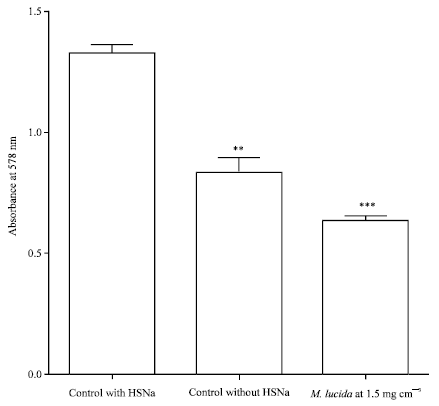
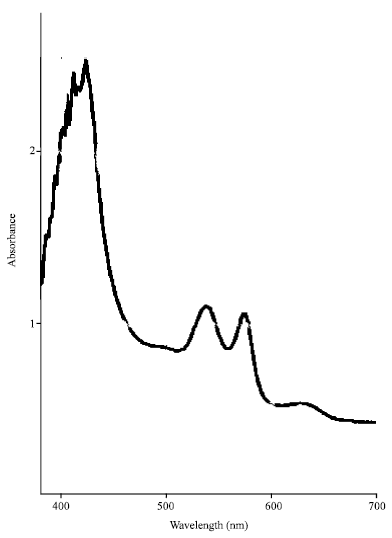
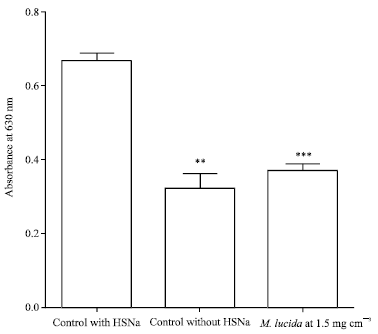
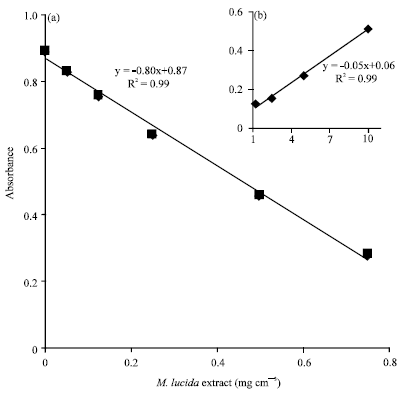
Research Article
Study of the Inhibition of Deoxyhemoglobin S Polymerization by the Hydroalcoholic Extract of Morinda lucida
Laboratory of Physiology and Pharmacology, Faculty of Medicine and Pharmacy, University of Lome, P.O. Box 1515, Togo
LiveDNA: 228.31884
Kossi Metowogo
Laboratory of Physiology and Pharmacology, Faculty of Medicine and Pharmacy, University of Lome, P.O. Box 1515, Togo
Irenee M. Kueviakoe
Laboratory Service of CHU Campus, Faculty of Medicine and Pharmacy, University of Lome, P.O. Box 1515, Togo
Povi Lawson-Evi
Laboratory of Physiology and Pharmacology, Faculty of Medicine and Pharmacy, University of Lome, P.O. Box 1515, Togo
Ahoefa Vovor
Laboratory of Hematology of CHU Sylvanus Olympio, Faculty of Medicine and Pharmacy, University of Lome, P.O. Box 1515, Togo
Kodjo Aklikokou
Laboratory of Physiology and Pharmacology, Faculty of Medicine and Pharmacy, University of Lome, P.O. Box 1515, Togo
Messanvi Gbeassor
Laboratory of Physiology and Pharmacology, Faculty of Medicine and Pharmacy, University of Lome, P.O. Box 1515, Togo