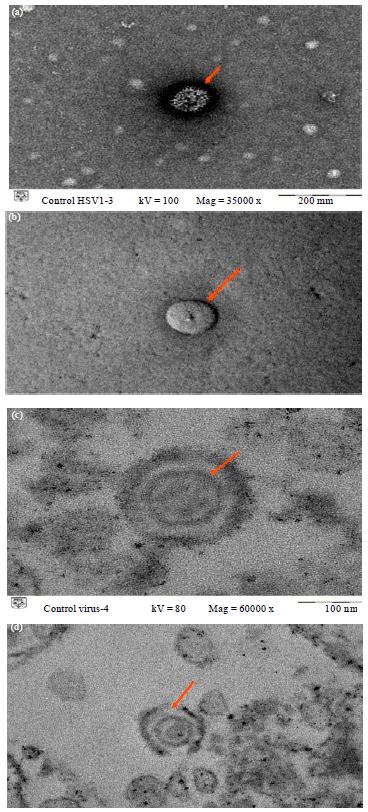
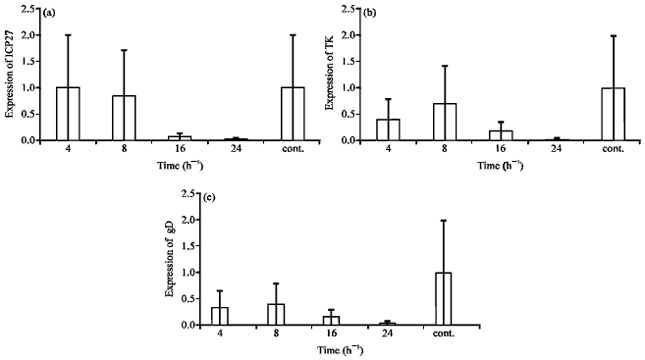
Research Article
In vitro Virucidal Activity of Hexane Fraction of Marrubium vulgare Against Type 1 Herpes simplex Virus
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600, Bangi Selangor, Malaysia
LiveDNA: 970.8274
Nazlina Ibrahim
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600, Bangi Selangor, Malaysia
Wan Ahmad Yaacob
School of Chemical Sciences and Food Technology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600, Bangi Selangor, Malaysia